Where Are Nonmetals Located In The Periodic Table
Muz Play
Apr 02, 2025 · 6 min read
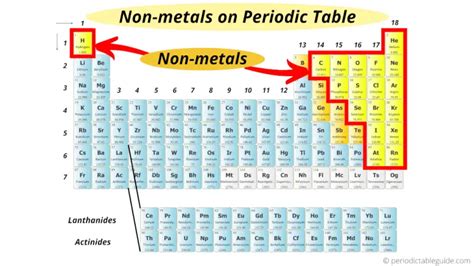
Table of Contents
Where are Nonmetals Located in the Periodic Table? A Comprehensive Guide
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. Understanding the arrangement of elements allows us to predict their behavior and reactivity. One crucial aspect of this organization is the classification of elements into metals, nonmetals, and metalloids. This article delves into the specific location of nonmetals on the periodic table, exploring their characteristics and providing a detailed explanation for their placement.
Understanding the Periodic Table's Organization
Before pinpointing the location of nonmetals, it's crucial to grasp the fundamental structure of the periodic table. Elements are arranged in rows (periods) and columns (groups or families). Periods represent the energy levels of electrons, while groups reflect the number of valence electrons—electrons in the outermost shell—which significantly influence an element's chemical properties. The periodic table is not a random arrangement; it's a carefully designed structure reflecting the underlying principles of atomic structure.
The Significance of Valence Electrons
The number of valence electrons directly impacts an element's reactivity. Elements in the same group possess the same number of valence electrons and, consequently, share similar chemical properties. For instance, elements in Group 18 (noble gases) have a full outer electron shell, making them exceptionally unreactive. This stable electron configuration is the driving force behind their inert nature. Conversely, elements with incomplete outer shells tend to be more reactive, readily gaining, losing, or sharing electrons to achieve a stable configuration.
Identifying Nonmetals: Characteristics and Properties
Nonmetals constitute a diverse group of elements exhibiting a range of properties that distinguish them from metals. Their physical and chemical characteristics are key to understanding their location on the periodic table.
Physical Properties of Nonmetals
-
State at Room Temperature: Many nonmetals exist as gases (e.g., oxygen, nitrogen, chlorine) at room temperature, while others are solids (e.g., carbon, sulfur, phosphorus). Only bromine is a liquid at room temperature. This varied physical state contrasts with the predominantly solid nature of metals.
-
Appearance: Nonmetals lack the characteristic metallic luster. They often appear dull or have various colors, depending on the element. For instance, sulfur is yellow, while phosphorus can be white or red.
-
Conductivity: Nonmetals are generally poor conductors of heat and electricity. This is a direct consequence of their electronic structure; electrons are tightly bound to their atoms, limiting their movement and inhibiting the flow of electricity.
-
Malleability and Ductility: Unlike metals, nonmetals are brittle and not easily shaped into wires or sheets. Their crystalline structures are less conducive to deformation compared to the closely packed metallic structures.
-
Density: Nonmetals typically have lower densities than metals.
Chemical Properties of Nonmetals
-
Electronegativity: Nonmetals have high electronegativity, meaning they strongly attract electrons in a chemical bond. This tendency contributes to their ability to gain electrons to achieve a stable electron configuration.
-
Ionization Energy: Nonmetals generally have high ionization energies. This reflects the energy required to remove an electron from a nonmetal atom, which is greater than that required for metals.
-
Oxidation States: Nonmetals can exhibit multiple oxidation states, meaning they can exist in various charged forms depending on the chemical context.
-
Formation of Covalent Bonds: Nonmetals readily form covalent bonds by sharing electrons with other atoms, including other nonmetals or even some metals.
Locating Nonmetals on the Periodic Table: A Visual Guide
Nonmetals are not clustered in a single, continuous block like metals. Instead, they occupy a distinct region of the periodic table. Their position is largely determined by their electronic configuration and associated properties.
The Staircase Line: A Defining Boundary
A diagonal line, often referred to as the "staircase," roughly separates metals from nonmetals. Elements bordering this line are metalloids, exhibiting properties intermediate between metals and nonmetals. Nonmetals are primarily located to the right of the staircase line, with the exception of hydrogen, which sits uniquely at the top left.
Specific Groups of Nonmetals
-
Group 17 (Halogens): This group contains highly reactive nonmetals (fluorine, chlorine, bromine, iodine, and astatine). Their high electronegativity makes them eager to gain an electron to form a stable -1 ion.
-
Group 16 (Chalcogens): This group includes oxygen, sulfur, selenium, tellurium, and polonium. They are involved in many biological and geological processes, exhibiting varying degrees of reactivity.
-
Group 15 (Pnictogens): This group contains nitrogen, phosphorus, arsenic, antimony, and bismuth. These elements exhibit a wider range of properties, with nitrogen being a gas and bismuth being a solid.
-
Group 14 (Tetrels): Carbon is the quintessential nonmetal in this group, while silicon and germanium exhibit metalloid characteristics. Carbon forms the basis of all organic life.
-
Group 18 (Noble Gases): Although unreactive, noble gases are classified as nonmetals due to their lack of metallic properties. Their full valence shells explain their inertness.
-
Hydrogen (Group 1): Hydrogen is an unusual element, possessing properties that differ from both metals and nonmetals. However, its nonmetallic properties are dominant, often classifying it within the nonmetal category, although its placement at the top left of the table makes it unique.
Metalloids: Bridging the Gap
Metalloids, also known as semimetals, are positioned along the staircase line separating metals and nonmetals. Their properties lie between those of metals and nonmetals, exhibiting a blend of characteristics. For example, silicon is a metalloid, acting as a semiconductor; this property makes it crucial in electronic devices.
The Importance of Understanding Nonmetal Location
The placement of nonmetals on the periodic table is not arbitrary; it directly reflects their atomic structure and resulting chemical behavior. Understanding this location allows us to predict:
-
Reactivity: The position indicates the likelihood of a nonmetal participating in chemical reactions.
-
Bonding: The placement helps predict the type of bonds a nonmetal will form (covalent bonds).
-
Properties: The location provides clues about a nonmetal's physical and chemical properties.
-
Applications: Knowledge of a nonmetal's characteristics helps in designing its applications in various fields such as medicine, electronics, and industry.
Conclusion: A Periodic Table Perspective on Nonmetals
The periodic table is a powerful tool for understanding the relationships between elements. The location of nonmetals, primarily to the right of the staircase line, is not accidental; it highlights their unique atomic structure, resulting in properties distinct from metals. Grasping the organization and characteristics of nonmetals provides a deeper appreciation of their chemical behavior and their indispensable role in various aspects of our world. Their diverse properties and significant applications underscore their vital contribution to science and technology. From the gaseous nitrogen in the air we breathe to the solid carbon in the pencils we write with, the nonmetals play a crucial role in shaping our environment and advancing our understanding of the natural world.
Latest Posts
Latest Posts
-
What Are The Two Major Groups Of Minerals
Apr 03, 2025
-
What Is A Word Equation In Chemistry
Apr 03, 2025
-
How Many A Columns Are On The Periodic Table
Apr 03, 2025
-
Kelvin Celsius And Fahrenheit Are Three Types Of Scales
Apr 03, 2025
-
Difference Between Applied And Basic Science
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Where Are Nonmetals Located In The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
