Where Are The Alkaline Earth Metals On The Periodic Table
Muz Play
Apr 02, 2025 · 6 min read
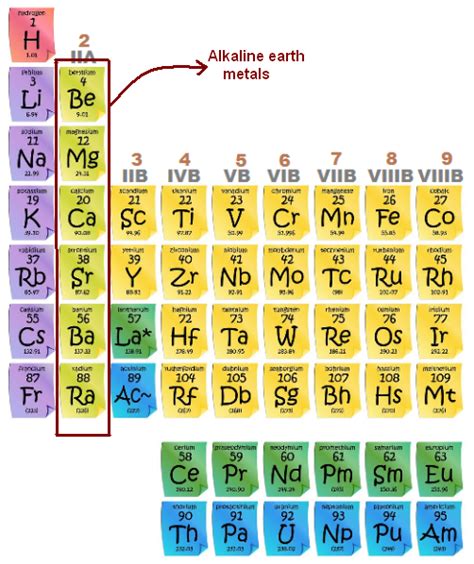
Table of Contents
Where Are the Alkaline Earth Metals on the Periodic Table? A Deep Dive into Group 2
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. Understanding its layout is crucial to comprehending the behavior of individual elements and their relationships with each other. This article delves into the location and characteristics of a specific group: the alkaline earth metals. We'll explore not only where they reside on the periodic table but also their unique properties, reactivity, and applications, making this a comprehensive guide for students and enthusiasts alike.
Identifying the Alkaline Earth Metals on the Periodic Table
The alkaline earth metals are found in Group 2 of the periodic table. This is the second column, located directly to the right of the alkali metals (Group 1). They are all s-block elements, meaning their valence electrons (the electrons involved in chemical bonding) occupy the s orbital. This shared characteristic contributes significantly to their similar chemical behavior.
Here's a visual representation:
Group 1 Group 2 ...
Alkali Alkaline Earth
Metals Metals
Li Be
Na Mg
K Ca
Rb Sr
Cs Ba
Fr Ra
The elements included in the alkaline earth metals group are:
- Beryllium (Be)
- Magnesium (Mg)
- Calcium (Ca)
- Strontium (Sr)
- Barium (Ba)
- Radium (Ra)
Notice that radium is radioactive; this highlights the trend of increasing reactivity and radioactivity as you move down the group.
Properties of Alkaline Earth Metals: A Closer Look
The alkaline earth metals share several key properties, stemming from their similar electronic configurations. These properties dictate their reactivity and applications.
1. Electronic Configuration and Valence Electrons:
All alkaline earth metals have two electrons in their outermost s orbital. This configuration makes them relatively reactive, as they readily lose these two electrons to achieve a stable, noble gas electron configuration. This tendency to lose electrons is what defines them as metals.
2. Metallic Properties:
They exhibit typical metallic properties such as:
- Good electrical conductivity: The free movement of electrons allows for efficient electrical conduction.
- Good thermal conductivity: Similarly, the mobile electrons facilitate the transfer of heat.
- Malleability and ductility: They can be hammered into sheets (malleability) and drawn into wires (ductility). However, this property is less pronounced compared to some other metals.
- Metallic luster: They possess a shiny, metallic appearance.
3. Reactivity:
While less reactive than the alkali metals (Group 1), alkaline earth metals are still quite reactive, especially when reacting with water and oxygen. Their reactivity increases down the group as the atomic radius increases, making it easier to lose the valence electrons.
- Reaction with oxygen: They readily react with oxygen to form oxides (e.g., MgO, CaO). These oxides are typically ionic compounds.
- Reaction with water: The reaction with water is more vigorous further down the group. Beryllium does not react readily with water, magnesium reacts slowly, while calcium, strontium, and barium react more vigorously, producing hydrogen gas and the corresponding hydroxide.
- Reaction with acids: They react with acids to form salts and hydrogen gas.
4. Ionization Energy:
Alkaline earth metals have relatively low ionization energies, meaning it takes relatively little energy to remove their two valence electrons. This low ionization energy directly contributes to their reactivity.
5. Electronegativity:
They have low electronegativity values, indicating a lower tendency to attract electrons in a chemical bond. This further reinforces their tendency to lose electrons and form positive ions.
Applications of Alkaline Earth Metals: From Everyday Life to High-Tech Industries
The unique properties of alkaline earth metals lead to a wide range of applications in various industries.
1. Magnesium (Mg):
- Lightweight alloys: Magnesium's low density makes it ideal for creating lightweight alloys used in automobiles, aircraft, and other transportation applications.
- Grignard reagents: In organic chemistry, Grignard reagents (organomagnesium compounds) are crucial for forming carbon-carbon bonds, used extensively in the synthesis of organic molecules.
- Photography: Magnesium is used in flash photography due to its ability to burn brightly and quickly.
2. Calcium (Ca):
- Construction materials: Calcium carbonate (CaCO3), a major component of limestone, is vital in cement production. Gypsum (calcium sulfate) is also used in construction.
- Biological roles: Calcium plays a crucial role in biological systems, including bone formation and muscle contraction. Calcium supplements are widely used.
- Metallurgy: Calcium is used as a reducing agent in the extraction of some metals from their ores.
3. Strontium (Sr):
- Pyrotechnics: Strontium compounds produce a vibrant red color in fireworks.
- Medical applications: Some strontium isotopes are used in medical imaging and treatments.
4. Beryllium (Be):
- Aerospace industry: Beryllium alloys are used in aerospace applications due to their high strength-to-weight ratio and stiffness.
- X-ray windows: Beryllium's transparency to X-rays makes it suitable for X-ray equipment.
5. Barium (Ba):
- Medical imaging: Barium sulfate (BaSO4) is used as a contrast agent in medical imaging, specifically for X-rays of the digestive system.
- Ceramics and glass: Barium compounds are used in the manufacture of certain ceramics and glasses.
6. Radium (Ra):
Due to its radioactivity, radium has limited modern applications. Historically, it was used in luminous paints, but this is no longer considered safe.
The Trend of Reactivity Down the Group
As we move down Group 2 from beryllium to radium, the reactivity of the alkaline earth metals increases significantly. This trend is primarily due to two factors:
- Increasing atomic radius: The atomic radius increases down the group, meaning the outermost electrons are further from the nucleus. This reduces the attractive force of the nucleus on these valence electrons, making them easier to lose.
- Decreasing ionization energy: Consequently, the ionization energy (the energy required to remove an electron) decreases down the group. The lower the ionization energy, the easier it is for the atom to lose its valence electrons and participate in chemical reactions.
This increase in reactivity is clearly seen in the reactions of the alkaline earth metals with water. Beryllium reacts negligibly with water, while barium reacts very vigorously.
Alkaline Earth Metals and Their Compounds: A Chemical Perspective
The chemical behavior of alkaline earth metals is largely governed by their tendency to form +2 ions (cations) by losing their two valence electrons. These ions readily form ionic compounds with nonmetals. The properties of these compounds are often significantly different from the elemental metals themselves. For example, magnesium metal is flammable, but magnesium oxide (MgO) is a stable, high-melting-point solid.
The study of alkaline earth metal compounds is vast and spans numerous fields, including inorganic chemistry, materials science, and biochemistry. Their diverse applications and unique properties continue to be the subject of ongoing research and development.
Conclusion: The Significance of Group 2
The alkaline earth metals, residing in Group 2 of the periodic table, represent a fascinating group of elements with distinctive properties and widespread applications. Their consistent electronic configuration, coupled with the predictable trend of increasing reactivity down the group, makes them a crucial element in our understanding of chemical periodicity and reactivity. From lightweight alloys to medical imaging, their impact on various aspects of modern life is undeniable, highlighting the importance of comprehending their unique place on the periodic table. This in-depth exploration serves as a foundation for further study of these elements and their crucial roles in various scientific and industrial contexts.
Latest Posts
Latest Posts
-
Evaluating Functions Linear And Quadratic Or Cubic
Apr 03, 2025
-
What Are The Reactants In The Calvin Cycle
Apr 03, 2025
-
What Happened To Islam After The Death Of Muhammad
Apr 03, 2025
-
Which Of These Diagrams Is A Convex Mirror
Apr 03, 2025
-
Which State Of Matter Has A Definite Shape
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Where Are The Alkaline Earth Metals On The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
