Which Group On The Periodic Table Is The Most Reactive
Muz Play
Apr 02, 2025 · 5 min read
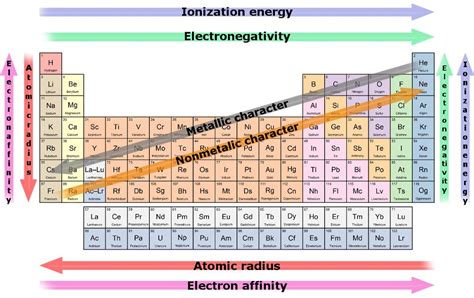
Table of Contents
Which Group on the Periodic Table is the Most Reactive?
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. One of the most striking trends observable is the variation in reactivity across groups (vertical columns). While reactivity is a complex concept influenced by multiple factors, a clear answer emerges when considering the most reactive groups: the alkali metals (Group 1) and the halogens (Group 17). However, a nuanced understanding requires exploring the underlying reasons for their high reactivity, comparing their behavior, and considering the exceptions and complexities within each group.
Understanding Reactivity
Before diving into specific groups, let's define reactivity. In chemistry, reactivity refers to the tendency of an element to undergo chemical changes. This involves forming or breaking chemical bonds with other elements or compounds. Highly reactive elements readily participate in chemical reactions, often exhibiting vigorous or even explosive behavior. Conversely, less reactive elements (like noble gases) are less inclined to form bonds.
Reactivity is fundamentally linked to an element's electronic configuration, specifically the number and arrangement of electrons in its outermost shell, also known as the valence shell. Elements strive for stability, often achieved by acquiring a full valence shell (typically eight electrons, following the octet rule). This drive towards a stable electronic configuration dictates their reactivity.
The Alkali Metals: Group 1 – Masters of Electron Donation
The alkali metals (lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr)) are located in Group 1 of the periodic table. They are characterized by having one valence electron. This single electron is relatively loosely held, making it easily lost in chemical reactions. This readily available electron makes alkali metals exceptionally reactive.
Why are Alkali Metals so Reactive?
-
Low Ionization Energy: The ionization energy is the energy required to remove an electron from an atom. Alkali metals possess extremely low ionization energies, meaning very little energy is needed to remove that single valence electron. This facilitates the formation of +1 cations (positively charged ions).
-
Electropositivity: Alkali metals are highly electropositive, meaning they have a strong tendency to lose electrons and form positive ions. This is a direct consequence of their low ionization energy and their desire to achieve a stable, noble gas electron configuration.
-
Increasing Reactivity Down the Group: As you move down Group 1, the atomic radius increases. This means the valence electron is further away from the nucleus and experiences weaker electrostatic attraction. Consequently, reactivity increases down the group. Cesium and francium are the most reactive alkali metals.
Reactions of Alkali Metals
Alkali metals react vigorously with water, producing hydrogen gas and a metal hydroxide. For example, sodium reacts with water according to the following equation:
2Na(s) + 2H₂O(l) → 2NaOH(aq) + H₂(g)
The reaction is highly exothermic (releases a significant amount of heat), often resulting in a flame. The reactivity increases dramatically down the group, with cesium reacting explosively with even traces of moisture.
Alkali metals also react readily with halogens (Group 17), forming ionic compounds known as alkali halides (e.g., NaCl – table salt). These reactions are typically very fast and exothermic.
The Halogens: Group 17 – Masters of Electron Acceptance
The halogens (fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At)) occupy Group 17. They are characterized by having seven valence electrons. Their high reactivity stems from their strong tendency to gain one electron to complete their octet and achieve a stable noble gas configuration.
Why are Halogens so Reactive?
-
High Electron Affinity: Electron affinity is the energy change that occurs when an atom gains an electron. Halogens exhibit high electron affinities, meaning they release a significant amount of energy when they accept an electron. This makes the process energetically favorable.
-
Electronegativity: Halogens are highly electronegative, indicating their strong attraction for electrons in a chemical bond. This enables them to readily pull electrons away from other atoms, forming negative ions (anions).
-
Decreasing Reactivity Down the Group: Unlike alkali metals, the reactivity of halogens decreases as you move down the group. This is due to the increasing atomic radius, which results in a weaker attraction for an additional electron. Fluorine is the most reactive halogen.
Reactions of Halogens
Halogens readily react with most metals to form ionic halides. For instance, chlorine reacts with sodium to form sodium chloride:
2Na(s) + Cl₂(g) → 2NaCl(s)
Halogens also react with hydrogen to form hydrogen halides (e.g., HCl – hydrochloric acid). These reactions can be vigorous, particularly with fluorine. The reactivity decreases down the group, with astatine being the least reactive. Furthermore, halogens can undergo displacement reactions, where a more reactive halogen displaces a less reactive one from its compound.
Comparing Alkali Metals and Halogens
Both alkali metals and halogens are exceptionally reactive, but their reactivity mechanisms differ significantly:
| Feature | Alkali Metals (Group 1) | Halogens (Group 17) |
|---|---|---|
| Valence electrons | 1 | 7 |
| Reactivity mechanism | Electron donation | Electron acceptance |
| Ion formation | Positive ions (+1) | Negative ions (-1) |
| Reactivity trend | Increases down the group | Decreases down the group |
Noble Gases: The Exception
The noble gases (Group 18) are exceptionally unreactive, hence their name. They have a complete valence shell (eight electrons, except for helium with two), rendering them very stable and less inclined to participate in chemical reactions. This stable electronic configuration explains their low reactivity. However, under extreme conditions, some noble gases can form compounds.
Conclusion: Nuance in Reactivity
While both alkali metals (Group 1) and halogens (Group 17) are considered the most reactive groups on the periodic table, their reactivity manifests in different ways. Alkali metals readily donate their single valence electron, while halogens readily accept an electron to complete their octet. The reactivity within each group shows a clear trend related to atomic size and its effect on electron attraction. Understanding these fundamental principles provides a deeper appreciation for the periodic table's organization and the predictable behavior of its elements. The apparent simplicity of this "most reactive" question highlights the rich complexity and intricate chemical behaviors of the elements and their interactions. Further exploration into other factors influencing reactivity, such as electronegativity, ionization energy, and atomic radius, will enhance the understanding of this crucial chemical concept.
Latest Posts
Latest Posts
-
Shortage On Supply And Demand Graph
Apr 03, 2025
-
Place The Products And Reactants Of The Citric Acid Cycle
Apr 03, 2025
-
Red Blood Cells In Hypertonic Solution
Apr 03, 2025
-
The Echelon Form Of A Matrix Is Unique
Apr 03, 2025
-
How To Write An Equation For A Vertical Line
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Group On The Periodic Table Is The Most Reactive . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
