Which Of The Following Can Exist As A Meso Isomer
Muz Play
Apr 05, 2025 · 5 min read
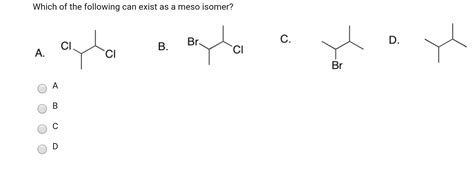
Table of Contents
Which of the Following Can Exist as a Meso Isomer?
Meso compounds are a fascinating subset of stereoisomers that often trip up students of organic chemistry. Understanding them requires a grasp of chirality, enantiomers, diastereomers, and internal planes of symmetry. This article will delve deep into the concept of meso compounds, providing a clear explanation of what they are, how to identify them, and clarifying the conditions necessary for their existence. We'll also explore several examples to solidify your understanding.
Understanding Stereoisomers: A Quick Recap
Before diving into meso compounds, let's briefly review the fundamental concepts of stereoisomerism. Stereoisomers are molecules with the same molecular formula and connectivity but differ in the three-dimensional arrangement of their atoms in space. There are two main types of stereoisomers:
-
Enantiomers: These are non-superimposable mirror images of each other. They possess chirality centers (carbon atoms with four different substituents) and rotate plane-polarized light in opposite directions.
-
Diastereomers: These are stereoisomers that are not mirror images of each other. They can have different physical and chemical properties. Diastereomers include meso compounds.
What is a Meso Compound?
A meso compound is a type of stereoisomer that contains chiral centers but possesses an internal plane of symmetry. This internal plane of symmetry makes the molecule superimposable on its mirror image, rendering it achiral despite the presence of chiral centers. Crucially, this means a meso compound is optically inactive; it does not rotate plane-polarized light.
Think of it this way: a meso compound is a special kind of diastereomer that is achiral due to its intrinsic symmetry. It's a molecule with a Jekyll and Hyde personality – it has chiral centers (Jekyll), but overall achiral symmetry cancels out their optical activity (Hyde).
Identifying Meso Compounds: The Key is Symmetry
The key to identifying a meso compound lies in the presence of an internal plane of symmetry. This plane divides the molecule into two halves that are mirror images of each other. If you can draw a plane through the molecule that perfectly bisects it, reflecting one half onto the other, you've likely found a meso compound.
Let's look at some common examples to illustrate this concept.
Examples of Meso Compounds
1. Meso-Tartaric Acid
Tartaric acid is a classic example used to illustrate the concept of meso compounds. It has two chiral centers, but the meso-tartaric acid isomer has an internal plane of symmetry. This plane passes through the molecule, dividing it into two halves that are mirror images of each other. As a result, meso-tartaric acid is optically inactive.
(Insert image of meso-tartaric acid with internal plane of symmetry clearly indicated)
The image would ideally show the molecule and a dashed line representing the plane of symmetry.
2. 2,3-Dibromobutane
Another frequently used example is 2,3-dibromobutane. This molecule also possesses two chiral centers. However, one stereoisomer of 2,3-dibromobutane is a meso compound. Again, the crucial factor is the presence of an internal plane of symmetry that renders the molecule achiral.
(Insert image of meso-2,3-dibromobutane with internal plane of symmetry clearly indicated)
Similar to the previous example, the image should showcase the molecule and the plane of symmetry.
3. Meso-1,2-Dibromocyclohexane
Cyclic molecules can also form meso compounds. Consider 1,2-dibromocyclohexane. Certain configurations of this molecule possess an internal plane of symmetry passing through the carbons that are not bonded to bromines, making it a meso compound.
(Insert image of meso-1,2-dibromocyclohexane with internal plane of symmetry clearly indicated)
The image should again clearly show the molecule and its plane of symmetry.
Distinguishing Meso Compounds from Other Stereoisomers
It's crucial to understand how meso compounds differ from other stereoisomers.
-
Meso compounds vs. Enantiomers: Meso compounds are achiral and optically inactive, unlike enantiomers, which are chiral and rotate plane-polarized light in opposite directions.
-
Meso compounds vs. other Diastereomers: While both are stereoisomers that are not mirror images, meso compounds are unique due to their internal plane of symmetry and consequent optical inactivity. Other diastereomers lack this symmetry and are typically optically active.
Predicting the Existence of Meso Isomers
The presence of multiple chiral centers does not automatically guarantee the existence of a meso isomer. The key requirement remains the presence of an internal plane of symmetry.
For example, a molecule with two chiral centers might have four possible stereoisomers (2<sup>n</sup>, where 'n' is the number of chiral centers). However, if the molecule is symmetrically substituted, one of these stereoisomers will be a meso compound, resulting in only three stereoisomers that are distinct (two pairs of enantiomers and one meso compound).
Applications of Meso Compounds
While not as widely discussed as other stereoisomers, meso compounds have implications in various fields:
-
Organic Synthesis: Understanding meso compounds is crucial in designing and predicting the outcome of stereoselective organic reactions.
-
Drug Design: The chirality of molecules is a critical factor in their biological activity. The presence or absence of a meso isomer can significantly influence a drug's effectiveness and potential side effects.
-
Material Science: The symmetry of molecules, including meso compounds, can influence the properties of materials such as crystals and polymers.
Advanced Considerations: More Complex Molecules
Identifying meso compounds becomes more challenging as molecular complexity increases. In such cases, visualizing the molecule in three dimensions and systematically checking for internal planes of symmetry becomes crucial. Molecular modeling software can be incredibly helpful in these scenarios.
Conclusion: Mastering Meso Compounds
Meso compounds represent a fascinating aspect of stereochemistry. Their existence highlights the importance of considering three-dimensional structure when analyzing molecules. Understanding the concept of internal planes of symmetry is the key to identifying these unique stereoisomers. By mastering this concept, you gain a deeper understanding of chirality, optical activity, and the subtle yet profound differences between seemingly similar molecules. This understanding is crucial for success in organic chemistry and related fields. Remember to practice identifying meso compounds through various examples, and don't hesitate to utilize molecular modeling tools for more complex structures. With practice and a solid understanding of the fundamental principles, you will confidently navigate the intricacies of meso isomers.
Latest Posts
Latest Posts
-
Accessory Structures Of The Integumentary System
Apr 06, 2025
-
How Do Water Particles Move In A Wave
Apr 06, 2025
-
Why Is Density An Intensive Property
Apr 06, 2025
-
Which Of The Following Bones Belong To The Axial Skeleton
Apr 06, 2025
-
What Are The Properties Of Solids
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Can Exist As A Meso Isomer . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
