Which Of The Following Is A Single Replacement Reaction
Muz Play
Apr 03, 2025 · 6 min read
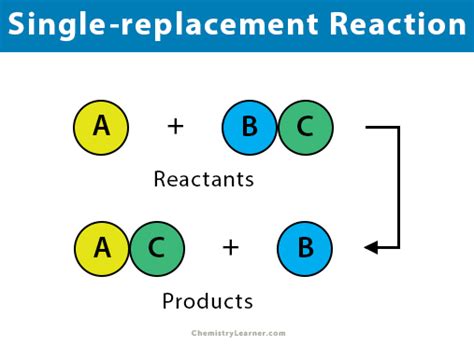
Table of Contents
Which of the Following is a Single Replacement Reaction? A Deep Dive into Chemical Reactions
Understanding different types of chemical reactions is fundamental to grasping chemistry. One crucial reaction type is the single replacement reaction, also known as a single displacement reaction. This article will thoroughly explore single replacement reactions, differentiating them from other reaction types, providing numerous examples, and clarifying common misconceptions. We'll delve into the underlying principles, explore the conditions that favor these reactions, and even examine some real-world applications. By the end, you’ll be able to confidently identify a single replacement reaction from a variety of chemical equations.
What is a Single Replacement Reaction?
A single replacement reaction, in its simplest form, involves one element replacing another element in a compound. This results in the formation of a new element and a new compound. The general form of a single replacement reaction is:
A + BC → AC + B
Where:
- A is a more reactive element.
- B is a less reactive element.
- BC is a compound.
- AC is a new compound formed.
The key here is that one element (A) is replacing another (B) within the compound (BC). This reaction hinges on the relative reactivity of the elements involved. The more reactive element will displace the less reactive element from its compound.
Identifying the Characteristics of Single Replacement Reactions
Several key characteristics help distinguish single replacement reactions from other reaction types:
- One element and one compound react: This is a defining feature. You will always have one element reacting with one compound, not two elements or two compounds.
- An element replaces another in a compound: The more reactive element replaces a less reactive one within the existing compound.
- New element and new compound form: The products always involve a new element and a new compound, not just a rearrangement of the existing atoms.
- Reactivity Series is crucial: The reactivity of the elements plays a critical role in determining whether the reaction will occur. A more reactive element will displace a less reactive one; otherwise, no reaction will take place.
The Reactivity Series: A Decisive Factor
The reactivity series (also known as the activity series) is a list of elements ordered by their reactivity. Elements higher on the series are more reactive than those lower down. This series is vital in predicting whether a single replacement reaction will occur. A higher element on the series will replace a lower element in a compound; a lower element will not replace a higher one.
For example, in the series for metals, potassium (K) is highly reactive, while gold (Au) is relatively unreactive. Potassium can displace many other metals from their compounds, but gold cannot displace elements higher in the series.
Example:
Potassium (K) is higher than copper (Cu) in the reactivity series. Therefore, the following reaction will occur:
2K + CuCl₂ → 2KCl + Cu
However, the reverse reaction will not occur because copper is less reactive than potassium.
Types of Single Replacement Reactions
Single replacement reactions can be broadly categorized into two main types:
-
Metal replacing a metal: This involves a more reactive metal displacing a less reactive metal from its salt solution. These reactions are usually carried out in aqueous solutions. For example:
Zn(s) + CuSO₄(aq) → ZnSO₄(aq) + Cu(s)
Here, zinc (Zn) replaces copper (Cu) in copper sulfate (CuSO₄).
-
Non-metal replacing a non-metal: A more reactive non-metal can displace a less reactive non-metal from its compound. These reactions often involve halogens. For example:
Cl₂(g) + 2NaBr(aq) → 2NaCl(aq) + Br₂(l)
Here, chlorine (Cl₂) replaces bromine (Br₂) in sodium bromide (NaBr).
Examples of Single Replacement Reactions
Let's delve into several specific examples to solidify your understanding:
1. Reaction of Zinc with Hydrochloric Acid:
Zn(s) + 2HCl(aq) → ZnCl₂(aq) + H₂(g)
Zinc is more reactive than hydrogen, thus it displaces hydrogen from hydrochloric acid.
2. Reaction of Iron with Copper(II) Sulfate:
Fe(s) + CuSO₄(aq) → FeSO₄(aq) + Cu(s)
Iron displaces copper from copper(II) sulfate. You'll observe a reddish-brown copper deposit forming.
3. Reaction of Chlorine with Potassium Iodide:
Cl₂(g) + 2KI(aq) → 2KCl(aq) + I₂(s)
Chlorine is more reactive than iodine; therefore, it displaces iodine from potassium iodide.
Distinguishing Single Replacement Reactions from Other Reaction Types
It's crucial to distinguish single replacement reactions from other common reaction types:
-
Double Replacement Reactions: In these reactions, two compounds exchange ions, resulting in the formation of two new compounds. They lack the characteristic of one element replacing another in a compound. For example: AgNO₃(aq) + NaCl(aq) → AgCl(s) + NaNO₃(aq)
-
Synthesis Reactions: These reactions involve the combination of two or more substances to form a single, more complex substance. For instance: 2H₂(g) + O₂(g) → 2H₂O(l)
-
Decomposition Reactions: These are the opposite of synthesis reactions; a single compound breaks down into two or more simpler substances. For example: 2H₂O₂(l) → 2H₂O(l) + O₂(g)
-
Combustion Reactions: These are rapid reactions that produce heat and light, often involving the reaction of a substance with oxygen. For example: CH₄(g) + 2O₂(g) → CO₂(g) + 2H₂O(l)
Applications of Single Replacement Reactions
Single replacement reactions have numerous practical applications, including:
-
Extraction of Metals: Many metals are extracted from their ores using single replacement reactions. For example, iron is extracted from its ore using carbon.
-
Metal Plating: This process involves depositing a thin layer of a more reactive metal onto a less reactive metal. It protects the underlying metal from corrosion and enhances its appearance.
-
Water Purification: Single replacement reactions can be used to remove unwanted substances from water.
-
Production of Chemicals: Several important chemicals are produced through single replacement reactions in industrial settings.
Troubleshooting Common Mistakes
Several common mistakes can occur when identifying single replacement reactions:
-
Confusing it with double replacement: Remember, only one element is replaced in a single replacement reaction.
-
Ignoring the reactivity series: The reactivity series is critical; the reaction will only proceed if a more reactive element displaces a less reactive one.
-
Incorrectly balancing the equation: Ensure the equation is balanced correctly to accurately represent the stoichiometry of the reaction.
Conclusion
Single replacement reactions are a fundamental type of chemical reaction with far-reaching applications. By understanding their characteristics, the reactivity series, and the distinctions between them and other reaction types, you can confidently identify and predict these reactions. This knowledge forms the bedrock for further exploration in chemistry, enabling you to tackle more complex chemical concepts and reactions. Remember to always practice and carefully examine the chemical equations provided to solidify your understanding of this important topic.
Latest Posts
Latest Posts
-
Identify The Features Associated With Waxes
Apr 04, 2025
-
Rna Differs From Dna In That Rna
Apr 04, 2025
-
What Is The Net Change In Math
Apr 04, 2025
-
Why Cant The Subscripts Be Changed In A Chemical Equation
Apr 04, 2025
-
How Many Bonds Does Boron Form
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Is A Single Replacement Reaction . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
