Which Subatomic Particle Determines The Identity Of The Atom
Muz Play
Apr 04, 2025 · 5 min read
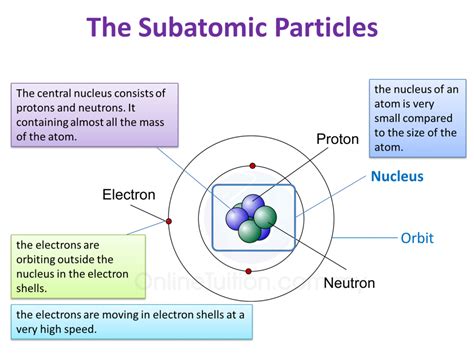
Table of Contents
Which Subatomic Particle Determines the Identity of an Atom?
The identity of an atom, its unique place on the periodic table, is fundamentally determined by a single subatomic particle: the proton. While electrons and neutrons play crucial roles in an atom's behavior and properties, it's the number of protons that definitively identifies an element. This article delves deep into the subatomic world, exploring the roles of protons, neutrons, and electrons, and solidifying the understanding of why the proton reigns supreme in defining atomic identity.
Understanding Subatomic Particles
Before diving into the specifics of atomic identity, let's establish a clear understanding of the three primary subatomic particles:
Protons: The Defining Factor
Protons are positively charged particles residing within the atom's nucleus. Their defining characteristic, beyond their positive charge, is their mass, approximately 1 atomic mass unit (amu). More importantly, the number of protons in an atom's nucleus determines its atomic number, and this atomic number uniquely identifies the element. For example, all hydrogen atoms have one proton, all helium atoms have two, all lithium atoms have three, and so on. This fundamental property is unchanging; it's what differentiates hydrogen from helium, oxygen from carbon, and every other element in the vast periodic table. Changing the number of protons fundamentally alters the element itself.
Neutrons: The Nuclear Stabilizers
Neutrons, as their name suggests, carry no electrical charge. Like protons, they reside within the atom's nucleus and contribute significantly to its mass (also approximately 1 amu). The number of neutrons in an atom's nucleus can vary, even within the same element. These variations lead to isotopes, atoms of the same element with different numbers of neutrons. While neutrons don't alter the element's identity (that's the proton's job), they significantly impact the atom's stability and properties. Some isotopes are stable, while others are radioactive, undergoing decay to achieve greater stability. The neutron-to-proton ratio plays a vital role in nuclear stability.
Electrons: The Reactive Players
Electrons are negatively charged particles that orbit the nucleus in specific energy levels or shells. Their mass is significantly smaller than that of protons and neutrons, approximately 1/1836 amu. While electrons don't define the element's identity, they play a crucial role in chemical reactions and bonding. The arrangement of electrons in the outermost shell, known as the valence electrons, determines the atom's reactivity and how it will interact with other atoms to form molecules and compounds. The number of electrons typically equals the number of protons in a neutral atom, maintaining an overall neutral charge. However, atoms can gain or lose electrons to form ions, which carry a net positive (cation) or negative (anion) charge.
The Atomic Number: The Proton's Reign
The atomic number is the cornerstone of an element's identity. It's defined as the number of protons in the nucleus of an atom. This number is unique to each element and is the basis for the organization of the periodic table. Elements are arranged in order of increasing atomic number, reflecting the increasing number of protons in their nuclei. This simple yet fundamental concept is the linchpin that distinguishes one element from another. No two elements share the same atomic number.
Isotopes: Variations on a Theme
While the number of protons defines the element, the number of neutrons can vary. Atoms of the same element with different numbers of neutrons are called isotopes. For example, carbon-12 (¹²C) has 6 protons and 6 neutrons, while carbon-14 (¹⁴C) has 6 protons and 8 neutrons. Both are carbon atoms because they both have 6 protons, but they differ in their mass and stability. Carbon-14 is radioactive, undergoing beta decay, while carbon-12 is stable. Isotopes have various applications, including carbon dating in archaeology and medicine.
The Role of Electrons in Chemical Behavior
Although electrons don't determine an atom's identity, their arrangement is crucial in determining the atom's chemical behavior. The outermost shell electrons, or valence electrons, are the primary participants in chemical bonds. Atoms strive to achieve a stable electron configuration, often by filling their outermost shell. This drive for stability leads to the formation of chemical bonds between atoms, resulting in the formation of molecules and compounds. The number of valence electrons dictates the bonding capacity of an atom, determining how many bonds it can form with other atoms. This aspect is vital in understanding chemical reactions and the properties of substances.
Beyond the Basics: Nuclear Physics and Isotope Applications
The relationship between protons, neutrons, and the atom's identity extends beyond simple chemistry into the realm of nuclear physics. Understanding isotopic variations allows us to explore nuclear reactions, radioactivity, and the applications of isotopes in various fields. For example:
- Nuclear Medicine: Radioactive isotopes are used in diagnostic imaging techniques like PET (positron emission tomography) scans and in radiotherapy for cancer treatment.
- Carbon Dating: The radioactive isotope carbon-14 is used to determine the age of ancient artifacts and organic materials.
- Nuclear Energy: Nuclear power plants harness the energy released during nuclear fission reactions, often involving specific isotopes of uranium.
- Scientific Research: Isotopes are used as tracers in various scientific experiments, helping scientists understand complex processes in biology, chemistry, and other fields.
Conclusion: The Proton's Undisputed Role
In conclusion, the identity of an atom is unequivocally determined by the number of protons in its nucleus. The atomic number, which directly represents the number of protons, is the defining characteristic that separates one element from another on the periodic table. While neutrons contribute to the atom's mass and stability, and electrons determine its chemical reactivity, it's the proton that holds the ultimate authority in defining the fundamental identity of an atom. Understanding this foundational concept is crucial for comprehending the organization of matter, chemical reactions, and the fascinating world of nuclear physics and its applications. The proton, a tiny particle within the atom's core, is the ultimate key to unlocking the secrets of the elements. Its simple yet profound influence on atomic identity underpins the entire structure and function of the universe as we know it. The study of protons and other subatomic particles continues to be a dynamic and ever-evolving field of scientific inquiry, constantly revealing new insights into the fundamental building blocks of matter and the universe. Further research into the behavior and properties of protons, neutrons, and electrons promises to unlock even deeper understanding of atomic structure and its implications for various scientific disciplines.
Latest Posts
Latest Posts
-
An Acid Is A Proton Donor
Apr 04, 2025
-
Calculate The Heat Capacity Of The Calorimeter
Apr 04, 2025
-
What Is The Correct Order Of Events In Mitosis
Apr 04, 2025
-
Writing A Chemical Equation From A Description Of The Reaction
Apr 04, 2025
-
Highness Or Lowness Of A Sound
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Which Subatomic Particle Determines The Identity Of The Atom . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
