An Acid Is A Proton Donor
Muz Play
Apr 04, 2025 · 6 min read
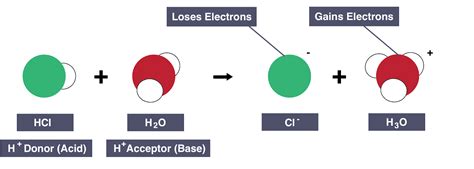
Table of Contents
An Acid is a Proton Donor: A Deep Dive into Acid-Base Chemistry
The statement, "an acid is a proton donor," is a cornerstone of modern chemistry, specifically within the context of the Brønsted-Lowry acid-base theory. Understanding this definition requires exploring various aspects of acid-base chemistry, from the fundamental nature of protons to the implications of this definition in diverse chemical reactions and applications. This comprehensive article will delve into this concept, exploring its nuances and practical significance.
Understanding the Brønsted-Lowry Theory
Before diving into the intricacies of proton donation, it's crucial to establish the theoretical framework within which this definition holds. The Brønsted-Lowry theory, proposed independently by Johannes Nicolaus Brønsted and Thomas Martin Lowry in 1923, offers a broader perspective on acid-base reactions than the earlier Arrhenius theory. The Arrhenius definition, while useful for simple aqueous solutions, restricts acids to substances that produce hydrogen ions (H⁺) and bases to substances that produce hydroxide ions (OH⁻) in water. The Brønsted-Lowry theory expands this significantly.
Key tenets of the Brønsted-Lowry theory:
- Acid: A Brønsted-Lowry acid is defined as a substance that donates a proton (H⁺) to another substance.
- Base: A Brønsted-Lowry base is defined as a substance that accepts a proton (H⁺) from another substance.
- Conjugate Acid-Base Pairs: When an acid donates a proton, it forms its conjugate base. Similarly, when a base accepts a proton, it forms its conjugate acid. These pairs are related by the difference of a single proton.
This theory elegantly explains acid-base reactions in non-aqueous solvents and expands the range of substances considered acidic or basic. For instance, ammonia (NH₃) acts as a base by accepting a proton, forming the ammonium ion (NH₄⁺), even though it doesn't produce hydroxide ions in water.
The Nature of the Proton (H⁺)
The proton, a hydrogen ion (H⁺), is a bare nucleus, essentially a single positively charged particle. Due to its small size and high charge density, it doesn't exist independently in solution. Instead, it strongly interacts with other molecules, typically through covalent or coordinate bonding. In aqueous solutions, protons are almost always associated with water molecules, forming hydronium ions (H₃O⁺). This is why we often represent acid dissociation in water as:
HA(aq) + H₂O(l) ⇌ H₃O⁺(aq) + A⁻(aq)
Where HA represents the acid, and A⁻ is its conjugate base. While the H⁺ notation is frequently used for simplicity, it's crucial to remember that the proton is always solvated (bound to solvent molecules).
Examples of Proton Donors: Acids in Action
Numerous substances qualify as Brønsted-Lowry acids based on their ability to donate protons. The strength of an acid is determined by its tendency to donate a proton. Strong acids completely dissociate in solution, while weak acids only partially dissociate.
Strong Acids: Complete Dissociation
Strong acids, such as hydrochloric acid (HCl), sulfuric acid (H₂SO₄), and nitric acid (HNO₃), readily donate their protons in aqueous solution. Their dissociation is essentially complete, meaning that virtually all of the acid molecules donate their protons to water molecules. This results in a high concentration of hydronium ions (H₃O⁺) and a low pH value.
Example: HCl(aq) + H₂O(l) → H₃O⁺(aq) + Cl⁻(aq)
Weak Acids: Partial Dissociation
Weak acids, such as acetic acid (CH₃COOH), formic acid (HCOOH), and carbonic acid (H₂CO₃), only partially dissociate in solution. An equilibrium is established between the undissociated acid and its conjugate base and hydronium ions. This results in a lower concentration of hydronium ions and a higher pH value compared to strong acids.
Example: CH₃COOH(aq) + H₂O(l) ⇌ H₃O⁺(aq) + CH₃COO⁻(aq)
The equilibrium constant, Ka, quantifies the extent of dissociation for weak acids. A smaller Ka value indicates a weaker acid.
Factors Affecting Acid Strength
Several factors influence the strength of an acid:
- Bond Strength: Weaker bonds between the proton and the rest of the molecule lead to stronger acids. The proton is more easily released.
- Electronegativity: The electronegativity of the atom bonded to the proton plays a crucial role. More electronegative atoms withdraw electron density, weakening the bond and making proton donation easier.
- Size and Structure: The size and structure of the molecule also affect the stability of the conjugate base. Larger, more stable conjugate bases lead to stronger acids.
- Resonance Stabilization: If the conjugate base is resonance-stabilized, the negative charge is delocalized over multiple atoms, making it more stable, and hence the acid stronger.
Beyond Aqueous Solutions: Proton Donation in Other Media
The Brønsted-Lowry theory transcends aqueous systems. Proton donation can occur in various solvents, including non-aqueous solvents like liquid ammonia or organic solvents. In these media, the proton is solvated by the solvent molecules instead of water. The acidity and basicity of a substance can vary depending on the solvent.
For example, acetic acid is a weak acid in water, but it behaves as a relatively strong acid in liquid ammonia. This is because liquid ammonia is a stronger base than water, making it more effective at accepting protons from acetic acid.
Applications of Proton Donation
The ability of acids to donate protons is crucial in countless chemical processes and applications. Some important examples include:
- Catalysis: Many acid-catalyzed reactions rely on the proton donating ability of acids. They often act as catalysts by donating protons to intermediates, lowering the activation energy of the reaction. Examples include esterification, dehydration, and many organic reactions.
- pH Regulation: Controlling pH is critical in many industrial processes and biological systems. Acids are frequently used to adjust pH to the desired level.
- Acid-Base Titrations: Acid-base titrations use the proton donation properties of acids to determine the concentration of unknown solutions.
- Electrochemistry: Acids play a vital role in electrochemical processes, such as batteries and fuel cells. The proton transfer is involved in the generation and transport of charge.
- Medicine: Many medications and biological processes involve acids and bases. The proper pH balance in the body is crucial for health.
Expanding the Definition: Lewis Acids
While the Brønsted-Lowry theory is extremely useful, it doesn't encompass all acid-base reactions. The Lewis theory provides a more general definition: A Lewis acid is an electron pair acceptor, while a Lewis base is an electron pair donor. A Brønsted-Lowry acid is a special case of a Lewis acid, where the electron pair acceptor is a proton.
Many substances that don't contain protons can still act as acids according to the Lewis definition. For example, boron trifluoride (BF₃) is a Lewis acid because it can accept an electron pair from a Lewis base. This expands the scope of acid-base chemistry beyond proton transfer.
Conclusion: The Significance of Proton Donation
The statement "an acid is a proton donor" is a fundamental concept in chemistry with wide-ranging implications. The Brønsted-Lowry theory provides a powerful framework for understanding acid-base reactions, explaining the behavior of acids and bases in various solvents and contexts. The ability of acids to donate protons is essential in numerous chemical processes, industrial applications, and biological systems, highlighting its central role in chemistry and beyond. While the Lewis definition provides a broader perspective, the proton donation aspect remains a key characteristic of Brønsted-Lowry acids and a crucial element in understanding their behavior and reactivity. Understanding this fundamental concept lays the groundwork for a deeper appreciation of the intricate world of acid-base chemistry.
Latest Posts
Latest Posts
-
Is Hbr An Acid Or Base
Apr 05, 2025
-
Example Of Weak Base And Strong Base
Apr 05, 2025
-
Which Quantity Will Always Have A Negative Value
Apr 05, 2025
-
An Oxidizing Agent Is A Substance That
Apr 05, 2025
-
F Test For Two Sample Variances
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about An Acid Is A Proton Donor . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
