Example Of Weak Base And Strong Base
Muz Play
Apr 05, 2025 · 6 min read
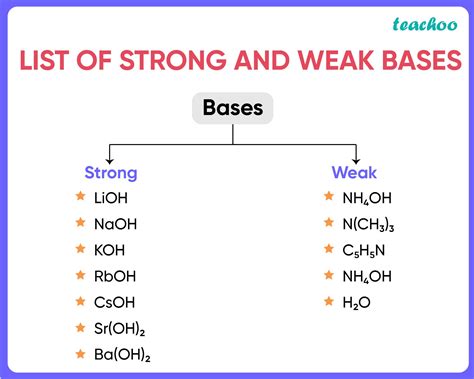
Table of Contents
Understanding Weak and Strong Bases: A Comprehensive Guide
Bases, a fundamental concept in chemistry, are substances that can accept protons (H⁺) or donate hydroxide ions (OH⁻) in a solution. They are crucial in numerous chemical reactions and industrial processes. However, not all bases are created equal. They are broadly classified into two categories: strong bases and weak bases, differing significantly in their ability to dissociate in water. This article delves deep into the characteristics, examples, and applications of both strong and weak bases.
What Makes a Base Strong or Weak?
The strength of a base is determined by its degree of dissociation in an aqueous solution. Strong bases completely dissociate into their constituent ions, while weak bases only partially dissociate. This difference has significant implications for their pH, reactivity, and applications.
Strong bases undergo complete ionization in water, meaning every molecule of the base releases hydroxide ions (OH⁻). This results in a high concentration of OH⁻ ions, leading to a highly alkaline solution with a high pH (typically above 10).
Weak bases, on the other hand, only partially ionize in water. Only a small fraction of the base molecules dissociate into hydroxide ions and their conjugate acid. This leads to a lower concentration of OH⁻ ions and a less alkaline solution with a lower pH compared to strong bases. The extent of dissociation is quantified by the base dissociation constant, Kb. A smaller Kb value indicates a weaker base.
Examples of Strong Bases
Strong bases typically involve group 1 (alkali metals) and group 2 (alkaline earth metals) hydroxides. Their complete dissociation in water makes them highly effective in neutralizing acids.
1. Sodium Hydroxide (NaOH) – Caustic Soda
Sodium hydroxide, commonly known as lye or caustic soda, is one of the most common and widely used strong bases. It's a highly reactive solid that readily dissolves in water, producing a strongly alkaline solution. Its applications are vast, including:
- Chemical industry: Used in the production of soaps, detergents, paper, textiles, and various chemicals.
- Drain cleaners: Its strong basicity effectively dissolves organic matter clogging drains.
- Food processing: Used in the processing of certain foods, though its use is strictly regulated due to its corrosive nature.
2. Potassium Hydroxide (KOH) – Caustic Potash
Similar to NaOH, potassium hydroxide or caustic potash is another potent strong base. It's also highly soluble in water and finds applications in:
- Soap making: Used in the production of liquid soaps and soft soaps.
- Fertilizers: Used in the production of potassium-based fertilizers.
- Electrolyte in batteries: Plays a crucial role in alkaline batteries.
3. Calcium Hydroxide (Ca(OH)₂) – Slaked Lime
Calcium hydroxide, or slaked lime, is a less soluble strong base compared to NaOH and KOH. It's still considered a strong base because the portion that dissolves completely dissociates. Its uses include:
- Construction: Used in mortar, plaster, and cement to bind materials together.
- Water treatment: Used to adjust the pH of water and remove impurities.
- Agriculture: Used to adjust soil pH for optimal plant growth.
4. Barium Hydroxide (Ba(OH)₂)
Barium hydroxide is another example of a strong base, though its solubility is relatively moderate. It's less commonly used compared to other strong bases but finds application in analytical chemistry and some specialized industrial processes.
Examples of Weak Bases
Weak bases don't fully dissociate in water. They establish an equilibrium between the undissociated base and its ions. Their relatively low pH compared to strong bases makes them suitable for specific applications where a milder alkaline environment is required.
1. Ammonia (NH₃)
Ammonia is a common weak base that dissolves in water to form ammonium hydroxide (NH₄OH), which partially dissociates into ammonium ions (NH₄⁺) and hydroxide ions (OH⁻). Its applications are diverse:
- Cleaning products: A key component in many household cleaners due to its ability to dissolve grease and grime.
- Fertilizers: A vital source of nitrogen for plant growth.
- Refrigerants: Used as a refrigerant in some industrial applications.
2. Pyridine (C₅H₅N)
Pyridine is an organic weak base containing a nitrogen atom in its aromatic ring. It's used as a:
- Solvent: In various chemical reactions and as a solvent in organic chemistry.
- Intermediate in chemical synthesis: Used in the production of pharmaceuticals, pesticides, and other chemicals.
3. Aniline (C₆H₅NH₂)
Aniline, also known as aminobenzene, is another organic weak base widely used in:
- Dye industry: A key ingredient in the production of various dyes and pigments.
- Pharmaceutical industry: Used as a starting material in the synthesis of many pharmaceuticals.
- Rubber industry: Used as an antioxidant in rubber production.
4. Methylamine (CH₃NH₂)
Methylamine is a simple organic weak base with applications in:
- Pharmaceutical industry: Used as a building block in the synthesis of certain pharmaceuticals.
- Pesticide industry: Used in the production of some pesticides.
Comparing Strong and Weak Bases
The following table summarizes the key differences between strong and weak bases:
| Feature | Strong Base | Weak Base |
|---|---|---|
| Dissociation | Complete in water | Partial in water |
| pH | High (typically above 10) | Lower than strong bases |
| Kb | Very large | Small |
| Examples | NaOH, KOH, Ca(OH)₂, Ba(OH)₂ | NH₃, Pyridine, Aniline, Methylamine |
| Conductivity | High electrical conductivity | Lower electrical conductivity |
| Reactivity | Highly reactive, readily neutralizes acids | Less reactive, slower neutralization of acids |
Practical Applications and Considerations
The choice between a strong or weak base depends heavily on the specific application. Strong bases are preferred when complete neutralization of an acid is required, or when a highly alkaline environment is necessary. However, their corrosive nature necessitates careful handling and safety precautions. Weak bases are suitable for applications where a milder alkaline environment is required, or when reactivity needs to be controlled.
Safety Precautions
Strong bases are corrosive and can cause severe burns to skin and eyes. Always handle them with appropriate safety equipment, including gloves, goggles, and lab coats. In case of accidental contact, immediately flush the affected area with plenty of water and seek medical attention. Weak bases are generally less hazardous, but appropriate safety measures should still be followed.
Environmental Considerations
The disposal of both strong and weak bases needs careful consideration. Improper disposal can lead to environmental pollution. Follow local regulations and guidelines for the safe disposal of chemical waste.
Conclusion
Understanding the difference between strong and weak bases is crucial in various fields, including chemistry, biology, and environmental science. Their diverse properties and applications make them essential components in numerous industrial processes and everyday products. However, it's equally important to be aware of their potential hazards and to handle them safely and responsibly. This comprehensive guide provides a solid foundation for appreciating the intricacies of these fundamental chemical substances. Further exploration into specific base reactions and their mechanisms will deepen this understanding even further, facilitating effective and safe applications across diverse scientific disciplines.
Latest Posts
Latest Posts
-
Study Of The Evolutionary Relationships Among Organisms
Apr 05, 2025
-
What Is The Disadvantage Of A Corporation
Apr 05, 2025
-
How To Find Average Molecular Speed
Apr 05, 2025
-
How To Divide Monomials And Polynomials
Apr 05, 2025
-
Give The Iupac Name Of The Branched Alkane
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Example Of Weak Base And Strong Base . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
