Give The Iupac Name Of The Branched Alkane
Muz Play
Apr 05, 2025 · 5 min read
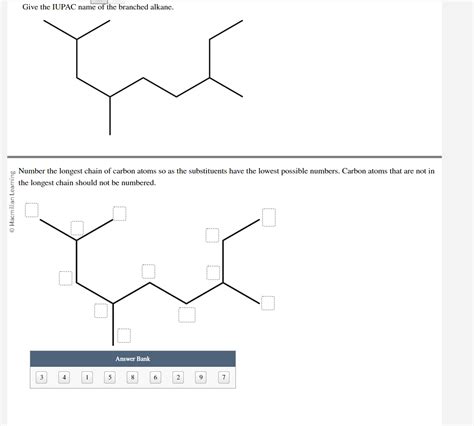
Table of Contents
Giving the IUPAC Name of Branched Alkanes: A Comprehensive Guide
Naming branched alkanes can seem daunting at first, but with a systematic approach, it becomes straightforward. This comprehensive guide will walk you through the process, equipping you with the knowledge to confidently name any branched alkane you encounter. We'll cover the fundamental rules of IUPAC nomenclature, providing clear examples and addressing common challenges.
Understanding IUPAC Nomenclature
The International Union of Pure and Applied Chemistry (IUPAC) established a standardized system for naming organic compounds, ensuring consistency and clarity in communication among scientists worldwide. This system is crucial for branched alkanes, as their structural complexity necessitates a rigorous naming convention. The core principles revolve around identifying the longest continuous carbon chain, numbering the carbons, identifying and naming substituents (branches), and combining these elements into a single, unambiguous name.
Key Steps in Naming Branched Alkanes
The process can be broken down into these key steps:
-
Identify the Longest Carbon Chain: This forms the parent alkane, determining the base name. It's crucial to find the longest continuous chain, even if it's not drawn in a straight line. Sometimes, you might need to rotate the molecule mentally to find the longest possible chain.
-
Number the Carbon Chain: Begin numbering from the end closest to the first substituent (branch). If substituents are equidistant from both ends, number to prioritize the lowest numbers for the substituents overall.
-
Identify and Name Substituents: Branches are alkyl groups, formed by removing a hydrogen atom from an alkane. Methyl (CH₃), ethyl (CH₂CH₃), propyl (CH₂CH₂CH₃), and butyl (CH₂CH₂CH₂CH₃) are common examples. More complex branches follow the same principles, with the longest continuous chain within the branch forming the base name.
-
Number the Substituents: Assign a number to each substituent based on its position on the parent chain. This number becomes a prefix in the name.
-
List Substituents Alphabetically: This is crucial for unambiguous naming, especially with multiple substituents. Numbers are ignored when alphabetizing (e.g., ethyl comes before methyl). However, prefixes such as di-, tri-, tetra- are considered only after the alphabetization.
-
Combine the Elements: Assemble the name by combining the substituent names (with their numbers) and the parent alkane name. Hyphens separate numbers and words, and commas separate numbers.
Examples: From Structure to IUPAC Name
Let's work through several examples, illustrating the application of these rules:
Example 1:
CH₃
|
CH₃-CH-CH₂-CH₃
- Longest Chain: Four carbons, hence butane.
- Numbering: Number from left to right, placing the methyl group at position 2.
- Substituent: One methyl group.
- Name: 2-methylbutane
Example 2:
CH₃ CH₃
| |
CH₃-CH-CH-CH₃
- Longest Chain: Four carbons, butane.
- Numbering: Number from left or right (both are equivalent); methyl groups are at positions 2 and 3.
- Substituents: Two methyl groups.
- Name: 2,3-dimethylbutane
Example 3: A more complex example:
CH₃
|
CH₃-CH-CH₂-CH-CH₂-CH₃
|
CH₂-CH₃
- Longest Chain: Six carbons, hexane.
- Numbering: Number from left to right to give the substituents the lowest numbers (2,4-dimethyl will have lower overall numbers than 3,5-dimethyl).
- Substituents: One methyl group on carbon 2, one ethyl group on carbon 4, and another methyl group on carbon 4.
- Alphabetical Ordering: Ethyl precedes methyl.
- Name: 4-ethyl-2,4-dimethylhexane
Example 4: Dealing with complex substituents
CH₃
|
CH₃-CH₂-CH-CH₂-CH₃
|
CH₂-CH₂-CH₃
- Longest Chain: Five carbons, pentane.
- Numbering: Number from left to right to provide the lowest numbers for the substituents.
- Substituents: One propyl group at position 3.
- Name: 3-propylhexane
Advanced Scenarios and Considerations
The examples above cover the basics. Let's address some more complex scenarios:
-
Multiple identical substituents: Use prefixes like di-, tri-, tetra-, penta-, etc., to indicate the number of identical substituents. These prefixes are not considered when alphabetizing the substituents. For example, 2,2,4-trimethylpentane.
-
Different substituents: List substituents alphabetically, regardless of their position on the chain. The numbers indicating the position are then used before each substituent's name. For instance, 3-ethyl-2-methylhexane.
-
Complex substituents: These are named as alkyl groups using the same rules as the main chain but with the suffix "-yl". For example, a substituent with three carbons attached to the main chain would be a propyl group, which would be named based on its position on the main chain, i.e., 2-propyl.
Isomers and Their IUPAC Names
Isomers are molecules with the same molecular formula but different structural arrangements. IUPAC nomenclature is crucial for distinguishing between isomers. Consider, for example, butane and methylpropane (isobutane). They both have the formula C₄H₁₀, but their structures and therefore their IUPAC names are distinct.
Understanding isomerism is vital for correctly naming branched alkanes. Different arrangements of the carbon chain lead to different IUPAC names, accurately reflecting the molecule's structure.
Practical Applications and Further Learning
Mastering IUPAC nomenclature for branched alkanes is essential in various fields, including:
-
Organic Chemistry: Accurate naming is fundamental in research, synthesis, and characterization of organic molecules.
-
Chemical Engineering: It's vital for understanding the properties and reactions of alkanes in industrial processes.
-
Biochemistry: Naming branched alkanes is relevant in understanding biological molecules and pathways.
This detailed guide provides a solid foundation in naming branched alkanes. Further learning can involve working through more complex examples, using online resources and interactive tutorials to reinforce your understanding. Remember, practice is key to mastering this crucial skill. Consistent application of the IUPAC rules will lead to accurate and unambiguous naming of even the most complex branched alkanes.
Latest Posts
Latest Posts
-
How To Do U Substitution With Definite Integrals
Apr 06, 2025
-
Membrane That Holds The Coils Of The Small Intestine Together
Apr 06, 2025
-
Differences Between Voltaic And Electrolytic Cells
Apr 06, 2025
-
Evaluate The Representation Of Ionic Bonding
Apr 06, 2025
-
Change In Allele Frequency Over Time
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Give The Iupac Name Of The Branched Alkane . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
