Why Does The Atomic Radii Increase Down A Group
Muz Play
Apr 05, 2025 · 5 min read
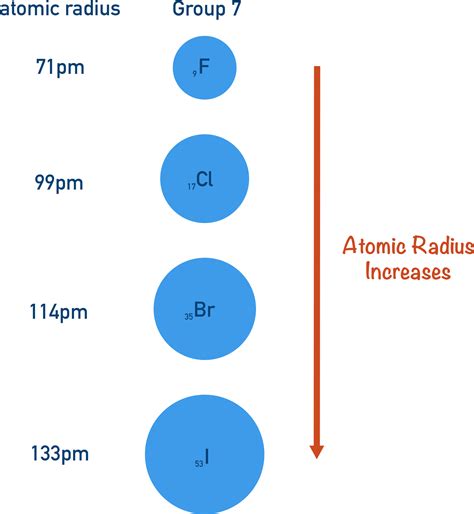
Table of Contents
Why Does Atomic Radius Increase Down a Group? A Deep Dive into Periodic Trends
Understanding periodic trends is fundamental to grasping the behavior of elements and their compounds. One such crucial trend is the increase in atomic radius as you move down a group in the periodic table. This seemingly simple observation belies a complex interplay of fundamental forces within the atom, specifically the electrostatic attraction between protons and electrons and the shielding effect of inner electrons. This article will delve into the detailed reasons behind this increase, exploring the concepts of effective nuclear charge, electron shielding, and the addition of principal energy levels.
The Role of Electron Shells and Principal Energy Levels
The atomic radius is essentially the distance between the nucleus and the outermost electron in an atom. As we move down a group, we're adding electrons to new principal energy levels (or shells). Each principal energy level represents a region of space further away from the nucleus. Think of it like adding layers to an onion; each layer is further from the center.
Principal Energy Levels and Distance:
- n=1 (First shell): This shell is closest to the nucleus, holding a maximum of two electrons.
- n=2 (Second shell): This shell is further away, holding a maximum of eight electrons.
- n=3 (Third shell): Even further away, holding a maximum of eighteen electrons.
And so on. The higher the principal quantum number (n), the greater the distance of that electron shell from the nucleus. This inherent increase in distance is the primary reason for the larger atomic radius down a group.
Effective Nuclear Charge: The Tug-of-War Between Protons and Electrons
The nucleus, containing positively charged protons, exerts an attractive force on the negatively charged electrons. This attraction is not uniform across all electrons. The effective nuclear charge (Z<sub>eff</sub>) represents the net positive charge experienced by an electron, considering both the attraction from the protons and the repulsion from other electrons.
Shielding Effect: Inner Electrons' Protective Role:
Inner electrons shield the outer valence electrons from the full positive charge of the nucleus. Imagine the inner electrons forming a "shield" that partially blocks the attractive force of the protons on the outer electrons. This shielding effect is crucial in understanding atomic radius trends.
Z<sub>eff</sub> and Atomic Radius:
A higher effective nuclear charge means a stronger attraction between the nucleus and the outer electrons, resulting in a smaller atomic radius. Conversely, a lower effective nuclear charge leads to a weaker attraction and a larger atomic radius.
As we move down a group, while the number of protons increases, the shielding effect from the added inner electrons becomes increasingly significant. The increase in the number of shielding electrons outweighs the increase in nuclear charge, leading to a decrease in the effective nuclear charge experienced by the outermost electrons. This reduced effective nuclear charge causes the outermost electrons to be less tightly bound to the nucleus, leading to a larger atomic radius.
Understanding Shielding and Penetration
Shielding isn't a uniform effect; different electron subshells (s, p, d, f) have different abilities to penetrate the inner shells and experience the nuclear charge. S orbitals, for example, have a higher probability density near the nucleus than p orbitals, meaning s electrons are less effectively shielded and experience a higher effective nuclear charge.
Penetration and Shielding:
- High Penetration: Electrons in s orbitals penetrate closer to the nucleus, experiencing less shielding and a greater effective nuclear charge.
- Low Penetration: Electrons in d and f orbitals penetrate less, experiencing more shielding and a lower effective nuclear charge.
The interplay between penetration and shielding across different subshells significantly influences the overall trend of atomic radius down a group, though the overall trend of increasing atomic radius remains dominant.
The Dominant Role of Added Energy Levels
While the effective nuclear charge and shielding play a role, the dominant factor contributing to the increase in atomic radius down a group is the addition of principal energy levels. The outermost electrons are found in progressively higher energy levels, further away from the nucleus. This increase in distance overwhelms any slight increase in effective nuclear charge.
Comparing Across Periods and Groups
It's important to contrast the trends in atomic radius down a group with those across a period (horizontally). Across a period, the atomic radius generally decreases. This is because the number of protons increases, but the electrons are added to the same principal energy level. The increased nuclear charge outweighs the shielding effect, leading to a stronger attraction and smaller radius.
Exceptions and Subtleties
While the general trend of increasing atomic radius down a group is well-established, some exceptions and subtleties exist. These deviations are often due to the complex interplay of electron-electron repulsions and the specific electronic configurations of elements. For instance, some anomalies might be observed due to the irregular filling of d and f orbitals, which can affect the shielding effect in unpredictable ways.
Real-World Applications of Understanding Atomic Radius
Understanding atomic radii and periodic trends has profound implications across various scientific disciplines:
- Chemistry: Predicting reactivity, bond lengths, and the properties of compounds. Larger atomic radii often correlate with lower ionization energies and increased reactivity.
- Material Science: Designing materials with specific properties. Atomic size influences the crystal structure and the physical properties of materials.
- Nuclear Physics: Understanding nuclear reactions and interactions. The size of the atom's electron cloud affects the likelihood of nuclear collisions.
Conclusion: A Comprehensive Overview
The increase in atomic radius down a group is a fundamental periodic trend resulting from the combined effects of increasing principal energy levels, effective nuclear charge, and electron shielding. While the addition of principal energy levels is the dominant factor, the interplay between shielding and penetration of different subshells adds complexity. This understanding forms a crucial foundation for predicting and explaining the properties of elements and their compounds, spanning diverse fields of science and technology. By comprehending these underlying mechanisms, we gain a deeper appreciation for the intricate and fascinating world of atomic structure and behavior.
Latest Posts
Latest Posts
-
Formulas With Polyatomic Ions Worksheet Answers
Apr 06, 2025
-
Practicing Dna Transcription And Translation Answer Key
Apr 06, 2025
-
How To Prove A Transformation Is Linear
Apr 06, 2025
-
Is The Mutant Allele Dominant Or Recessive
Apr 06, 2025
-
Why Do Covalent Compounds Have Low Melting Points
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Why Does The Atomic Radii Increase Down A Group . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
