Write The Rate Law For The Iodine-clock Reaction.
Muz Play
Apr 02, 2025 · 6 min read
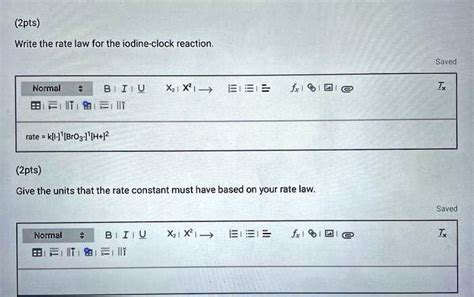
Table of Contents
Deciphering the Rate Law for the Iodine Clock Reaction: A Comprehensive Guide
The iodine clock reaction is a classic chemistry experiment demonstrating the principles of chemical kinetics. Its dramatic visual change – a sudden appearance of a deep blue-black color – makes it an engaging way to study reaction rates and determine rate laws. This article will delve into the intricacies of the iodine clock reaction, providing a comprehensive understanding of its mechanism and the derivation of its rate law. We'll explore the experimental procedure, data analysis techniques, and potential sources of error, equipping you with the knowledge to confidently conduct and interpret your own iodine clock experiments.
Understanding the Iodine Clock Reaction
The iodine clock reaction typically involves the oxidation of iodide ions (I⁻) by hydrogen peroxide (H₂O₂) in an acidic solution. This reaction is slow and doesn't produce a readily observable change. However, the cleverly designed experiment incorporates a second, much faster reaction that consumes the initially produced iodine (I₂). This second reaction involves the reduction of iodine by thiosulfate ions (S₂O₃²⁻):
I₂ + 2S₂O₃²⁻ → 2I⁻ + S₄O₆²⁻
The thiosulfate ions react rapidly with any iodine produced in the first reaction, preventing the formation of a visible iodine-starch complex. However, once all the thiosulfate ions are consumed, the iodine concentration begins to increase rapidly, and it immediately reacts with starch, forming a deep blue-black complex. This abrupt color change marks the "clock" moment, giving the reaction its name.
The Reaction Mechanism and Rate Determining Step
The overall reaction can be represented as:
H₂O₂ + 2I⁻ + 2H⁺ → I₂ + 2H₂O
However, this equation doesn't reveal the detailed mechanism. The actual reaction proceeds through several elementary steps. While the exact mechanism can be complex and debated, a simplified mechanism often used is:
Step 1 (Slow): H₂O₂ + I⁻ + H⁺ → H₂O + HOI
Step 2 (Fast): HOI + I⁻ + H⁺ → I₂ + H₂O
Step 3 (Fast): I₂ + 2S₂O₃²⁻ → 2I⁻ + S₄O₆²⁻
Step 4 (Fast): I₂ + Starch → I₂-Starch Complex (Blue-Black)
Identifying the Rate-Determining Step: The rate of the overall reaction is determined by the slowest step, which in this simplified mechanism is Step 1. This is because the subsequent steps are significantly faster and don't significantly impede the overall reaction rate.
Deriving the Rate Law
Since Step 1 is the rate-determining step, the rate law is determined by the concentrations of the reactants in this step:
Rate = k[H₂O₂][I⁻][H⁺]
Where:
- Rate is the rate of the reaction (typically expressed as the change in concentration per unit time).
- k is the rate constant (a temperature-dependent proportionality constant).
- [H₂O₂], [I⁻], and [H⁺] represent the concentrations of hydrogen peroxide, iodide ions, and hydrogen ions, respectively.
This rate law indicates that the reaction is first-order with respect to each of the reactants: hydrogen peroxide, iodide ions, and hydrogen ions. A doubling of the concentration of any of these reactants will result in a doubling of the reaction rate, assuming the other concentrations remain constant.
Experimental Determination of the Rate Law
To experimentally determine the rate law, you need to systematically vary the concentrations of the reactants while keeping others constant. By observing the time it takes for the color change to occur (the reaction time, t), you can indirectly measure the reaction rate. A shorter reaction time indicates a faster rate.
Procedure Outline:
-
Prepare Solutions: Prepare stock solutions of hydrogen peroxide, potassium iodide (KI), sulfuric acid (H₂SO₄), sodium thiosulfate (Na₂S₂O₃), and starch.
-
Mix Reactants: In separate beakers, mix appropriate volumes of hydrogen peroxide, potassium iodide, sulfuric acid, and sodium thiosulfate. Ensure the total volume of the mixture is consistent throughout your experiments.
-
Add Starch: Add a small amount of starch solution to the mixture.
-
Record Time: Start a timer immediately after mixing, and record the time (t) it takes for the blue-black color to appear.
-
Vary Concentrations: Repeat steps 2-4, systematically varying the concentration of one reactant at a time while keeping the others constant. This allows you to determine the order of the reaction with respect to each reactant.
-
Data Analysis: Analyze your data using the initial rates method or other appropriate kinetic analysis techniques. Plot the data appropriately (e.g., log(rate) vs. log(concentration) to determine the reaction order.
Data Analysis and Interpretation
The data collected from the experiment can be analyzed using several methods. The most common is the initial rates method. This method focuses on the initial rate of the reaction, where the concentrations of reactants have not changed significantly. By comparing the initial rates at different concentrations, you can determine the order of reaction with respect to each reactant.
For instance, if doubling the concentration of hydrogen peroxide doubles the initial rate, the reaction is first order with respect to hydrogen peroxide. If doubling the concentration quadruples the rate, the reaction is second order, and so on.
Another approach is to analyze the reaction time (t). Since the reaction time is inversely proportional to the rate, you can also analyze the reciprocal of the reaction time (1/t) to determine the reaction orders.
Potential Sources of Error and Mitigation Strategies
Several factors can affect the accuracy and precision of your experimental results. These include:
-
Temperature fluctuations: Temperature significantly influences the rate constant (k). Maintaining a constant temperature throughout the experiment is crucial. Use a water bath to control temperature.
-
Impurities: Impurities in the reactants can affect the reaction rate. Use high-purity chemicals.
-
Mixing inconsistencies: Ensure thorough and consistent mixing of the reactants. Incomplete mixing can lead to variations in reaction time.
-
Human error: Accurate timing and precise measurement of volumes are essential. Practice careful technique.
-
Starch concentration: The concentration of starch used can influence the sharpness of the color change, potentially affecting the accuracy of timing. Maintaining a consistent starch concentration is vital.
By carefully controlling experimental conditions and using appropriate data analysis techniques, these sources of error can be minimized.
Conclusion
The iodine clock reaction is a powerful demonstration of chemical kinetics principles. By understanding its mechanism, deriving its rate law, and conducting experiments carefully, you gain valuable insights into reaction rates and the factors influencing them. The seemingly simple experiment reveals the complexities of reaction dynamics and the importance of careful experimental design and data analysis in scientific investigation. This detailed guide provides a comprehensive understanding of the iodine clock reaction, empowering you to conduct successful experiments and interpret your results confidently. Remember to always prioritize safety when handling chemicals in the laboratory.
Latest Posts
Latest Posts
-
What Are The Two Major Groups Of Minerals
Apr 03, 2025
-
What Is A Word Equation In Chemistry
Apr 03, 2025
-
How Many A Columns Are On The Periodic Table
Apr 03, 2025
-
Kelvin Celsius And Fahrenheit Are Three Types Of Scales
Apr 03, 2025
-
Difference Between Applied And Basic Science
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Write The Rate Law For The Iodine-clock Reaction. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
