Activation Energy For The Forward Reaction
Muz Play
Apr 02, 2025 · 6 min read
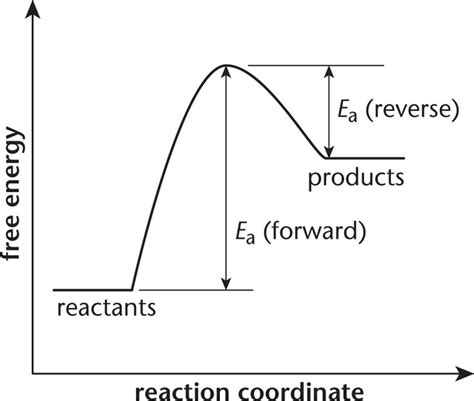
Table of Contents
Activation Energy for the Forward Reaction: A Deep Dive
Activation energy is a fundamental concept in chemistry, crucial for understanding reaction rates and the spontaneity of chemical processes. This article delves deep into the activation energy for the forward reaction, exploring its definition, significance, factors influencing it, and its applications in various fields. We will also examine the relationship between activation energy and the equilibrium constant, touching upon the Arrhenius equation and its implications.
Understanding Activation Energy
Activation energy (Ea) is the minimum amount of energy required for a chemical reaction to occur. It represents the energy barrier that reactant molecules must overcome to transform into products. Think of it like pushing a boulder up a hill; the energy needed to get the boulder to the top of the hill is analogous to the activation energy. Once the boulder reaches the top, it can roll down the other side (representing the formation of products), even if the overall process is energetically favorable (the bottom of the hill is lower than the starting point).
The Transition State
The process doesn't simply involve reactants transforming directly into products. Instead, reactants first form a high-energy, unstable intermediate called the transition state (or activated complex). This transition state is a fleeting structure, existing only for a brief moment before collapsing to form products or reverting back to reactants. The energy difference between the reactants and the transition state is the activation energy for the forward reaction.
Visualizing Activation Energy with Energy Diagrams
Energy diagrams, often depicted as graphs plotting potential energy against the reaction coordinate, provide a visual representation of activation energy. The reaction coordinate represents the progress of the reaction, from reactants to products. The activation energy is clearly visible as the energy difference between the reactants and the peak of the curve (representing the transition state).
A high activation energy indicates a slow reaction, as only a small fraction of reactant molecules possess enough energy to overcome the barrier. Conversely, a low activation energy signifies a fast reaction, as a larger fraction of molecules can overcome the energy barrier.
Factors Affecting Activation Energy
Several factors can significantly influence the activation energy for the forward reaction:
1. Nature of Reactants:
The chemical nature of the reactants plays a vital role. Reactions involving strong bonds (e.g., triple bonds) often have higher activation energies compared to reactions involving weaker bonds (e.g., single bonds). The inherent stability of the reactants directly affects the energy required to break bonds and form the transition state.
2. Reaction Mechanism:
The mechanism of a reaction, the step-by-step process through which reactants transform into products, profoundly impacts the activation energy. Reactions proceeding through multiple steps usually have lower overall activation energies compared to those proceeding through a single, high-energy step. Each step in a multi-step mechanism will have its own activation energy, and the rate-determining step (the slowest step) dictates the overall rate of the reaction.
3. Temperature:
Temperature is a crucial factor. Increasing the temperature boosts the kinetic energy of reactant molecules. This means a larger fraction of molecules possesses sufficient energy to surpass the activation energy barrier, thereby increasing the reaction rate. The relationship between temperature and rate is quantified by the Arrhenius equation (discussed later).
4. Catalysts:
Catalysts are substances that increase the rate of a reaction without being consumed in the process. They achieve this by lowering the activation energy. Catalysts provide an alternative reaction pathway with a lower energy barrier, thus allowing more reactant molecules to overcome the energy requirement and transform into products. Enzyme catalysis in biological systems is a prime example of this phenomenon. Enzymes act as biological catalysts, significantly accelerating the rates of biochemical reactions.
5. Presence of Solvents and Other Factors:
The solvent used in a reaction can also influence the activation energy. Polar solvents, for example, can stabilize charged transition states, thereby lowering the activation energy. Other factors such as pressure (particularly in gas-phase reactions) and surface area (in heterogeneous catalysis) also play a role in influencing the reaction rate indirectly by affecting the activation energy.
The Arrhenius Equation: Quantifying the Relationship Between Rate Constant and Activation Energy
The Arrhenius equation mathematically describes the relationship between the rate constant (k) of a reaction and its activation energy (Ea):
k = Ae^(-Ea/RT)
Where:
- k is the rate constant
- A is the pre-exponential factor (frequency factor) which represents the frequency of collisions between reactant molecules with correct orientation.
- Ea is the activation energy
- R is the ideal gas constant
- T is the absolute temperature
This equation reveals that the rate constant increases exponentially with temperature and decreases exponentially with activation energy. A higher activation energy results in a smaller rate constant at a given temperature, indicating a slower reaction.
Determining Activation Energy Experimentally
The Arrhenius equation can be manipulated into a linear form:
ln k = ln A – Ea/RT
By plotting ln k against 1/T, a straight line is obtained with a slope of -Ea/R. This allows for the experimental determination of the activation energy from the slope of the line.
Activation Energy and the Equilibrium Constant
While activation energy primarily governs the rate of a reaction, it also indirectly influences the equilibrium constant (K). For a reversible reaction, both the forward and reverse reactions have their own activation energies (Ea,forward and Ea,reverse). The relationship between these activation energies and the equilibrium constant is given by:
ΔG° = -RTlnK = Ea,forward – Ea,reverse
Where ΔG° is the standard Gibbs free energy change of the reaction. This equation highlights that the equilibrium constant is dependent not only on the difference in activation energies but also on the temperature. A larger difference between the forward and reverse activation energies favors the direction with the lower activation energy.
Applications of Activation Energy Concepts
The concept of activation energy has widespread applications in various scientific and engineering fields:
1. Catalysis:
Understanding activation energy is paramount in catalysis research and development. Scientists strive to design catalysts that effectively lower the activation energy of specific reactions, leading to enhanced reaction rates and increased efficiency in industrial processes.
2. Chemical Kinetics:
Activation energy is a cornerstone of chemical kinetics, the study of reaction rates. It allows scientists to predict and control reaction rates by manipulating factors that influence activation energy, such as temperature, catalysts, and reactant concentrations.
3. Material Science:
The activation energy concept is also vital in materials science, particularly in studying the kinetics of solid-state reactions such as diffusion and phase transformations. Understanding these processes is crucial for designing and developing new materials with desired properties.
4. Biochemistry:
In biochemistry, activation energy plays a central role in understanding enzyme-catalyzed reactions. The ability of enzymes to significantly lower the activation energy of biochemical processes is fundamental to life.
5. Environmental Science:
Activation energy is relevant in environmental chemistry, for instance, in predicting the rates of pollutant degradation or the kinetics of atmospheric reactions involving air pollutants.
Conclusion
Activation energy is a crucial concept in chemistry and related fields. It dictates the rate of a chemical reaction by defining the energy barrier that must be overcome for reactants to transform into products. Understanding factors influencing activation energy, its relationship with the Arrhenius equation, and its connections to the equilibrium constant are essential for controlling and predicting reaction rates. Its applications are far-reaching, extending from industrial catalysis to biological processes, making it a fundamental concept with significant practical implications. Further research continues to refine our understanding of activation energy and its diverse roles in various chemical and biological systems.
Latest Posts
Latest Posts
-
Where Is The Energy Stored In Glucose
Apr 03, 2025
-
Light Amplification By The Stimulated Emission Of Radiation
Apr 03, 2025
-
Octet Rule Violation Vs Wrong Electron Total
Apr 03, 2025
-
What Are The Three Points Of Cell Theory
Apr 03, 2025
-
How Many Atoms Are In A Simple Cubic Unit Cell
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Activation Energy For The Forward Reaction . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
