Alkali Metals Alkaline Earth Metals Halogens And Noble Gases
Muz Play
Apr 03, 2025 · 6 min read
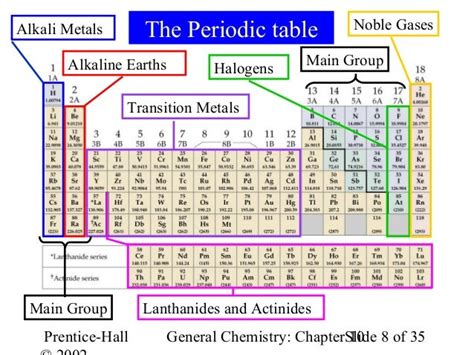
Table of Contents
Exploring the Periodic Table: Alkali Metals, Alkaline Earth Metals, Halogens, and Noble Gases
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. Understanding these properties allows us to predict how elements will behave and interact, leading to advancements in various fields, from medicine to materials science. This article delves into four key groups of elements: alkali metals, alkaline earth metals, halogens, and noble gases, exploring their unique characteristics, reactivity, and applications.
Alkali Metals: The Highly Reactive Group
Alkali metals, residing in Group 1 of the periodic table, are characterized by their extreme reactivity. This stems from their electronic configuration: they possess only one electron in their outermost shell (valence shell). This single electron is easily lost, forming a +1 ion. This tendency to readily lose an electron makes them highly reactive, especially with water and halogens.
Key Characteristics of Alkali Metals:
- Low ionization energy: The ease with which they lose their valence electron contributes to their low ionization energy.
- Low electronegativity: They have a weak attraction for electrons, further contributing to their reactivity.
- Soft and silvery-white appearance (except cesium, which has a golden hue): Their metallic bonding structure contributes to their malleability and ductility.
- Low density: They are significantly less dense than most other metals.
- Excellent conductors of heat and electricity: The loosely held valence electron facilitates the flow of charge.
Notable Alkali Metals and Their Applications:
- Lithium (Li): Used extensively in batteries (lithium-ion batteries), ceramics, and lubricating greases. Its lightweight nature makes it crucial in aerospace applications. Lithium compounds are also used in the treatment of bipolar disorder.
- Sodium (Na): Abundant in nature (sodium chloride, common salt), sodium is vital for biological processes. It's used in sodium-vapor lamps, producing a bright yellow light, and in the production of various chemicals.
- Potassium (K): Essential for plant growth and animal health, potassium is a crucial electrolyte in the human body. It's also used in fertilizers and in the manufacture of soaps and detergents.
- Rubidium (Rb) and Cesium (Cs): These less common alkali metals find applications in atomic clocks and specialized optical instruments, leveraging their unique atomic properties.
Alkaline Earth Metals: Reactivity and Applications
Located in Group 2 of the periodic table, alkaline earth metals possess two electrons in their valence shell. These electrons are also readily lost, but not as easily as those in alkali metals, resulting in a +2 ion. Consequently, they are less reactive than alkali metals but still exhibit significant reactivity, particularly at elevated temperatures.
Key Characteristics of Alkaline Earth Metals:
- Higher ionization energy than alkali metals: Losing two electrons requires more energy than losing one.
- Higher density than alkali metals: Their stronger metallic bonding contributes to their higher density.
- Relatively high melting and boiling points compared to alkali metals: Again, stronger metallic bonding plays a role.
- Stronger metallic character: This makes them more resistant to corrosion compared to alkali metals.
Notable Alkaline Earth Metals and Their Applications:
- Beryllium (Be): A lightweight yet incredibly strong metal, beryllium is used in aerospace applications, X-ray windows, and nuclear reactors, although its toxicity necessitates careful handling.
- Magnesium (Mg): A crucial element in chlorophyll, magnesium is used extensively in lightweight alloys (e.g., in automobiles and aircraft), as well as in flash photography and pyrotechnics.
- Calcium (Ca): Essential for strong bones and teeth, calcium is a vital nutrient. It's also used in cement, plaster, and as a reducing agent in metallurgy.
- Strontium (Sr): Used in fireworks, giving them a vibrant red color, strontium is also used in some specialized glass and ceramics.
- Barium (Ba): Used in drilling muds (oil and gas industry), barium compounds find applications in medical imaging (barium sulfate).
Halogens: The Salt-Formers
Group 17 elements, the halogens, are known for their high reactivity. They have seven electrons in their valence shell, meaning they readily gain one electron to achieve a stable octet, forming a -1 ion. This makes them strong oxidizing agents. Their reactivity decreases down the group as the atomic size increases.
Key Characteristics of Halogens:
- High electronegativity: Their strong attraction for electrons makes them highly reactive.
- Highly reactive nonmetals: They readily react with most metals and many nonmetals.
- Varying physical states at room temperature: Fluorine and chlorine are gases, bromine is a liquid, and iodine is a solid.
- Form diatomic molecules: They exist as diatomic molecules (e.g., F₂, Cl₂, Br₂, I₂) due to their strong tendency to form covalent bonds.
Notable Halogens and Their Applications:
- Fluorine (F): The most reactive halogen, fluorine is used in the production of fluorocarbons (Teflon, refrigerants), and its compounds are found in toothpaste (fluoride) for dental health.
- Chlorine (Cl): Widely used as a disinfectant (bleaching agent, water purification), chlorine is also crucial in the production of various chemicals, including PVC plastics.
- Bromine (Br): Used in flame retardants, photographic film, and certain dyes, bromine is less reactive than chlorine but still a significant industrial chemical.
- Iodine (I): Essential for thyroid hormone production, iodine is used in antiseptic solutions and in certain medications. It's also used in photography and some specialized lighting.
- Astatine (At): A radioactive and highly unstable element, astatine has limited practical applications.
Noble Gases: The Inert Elements
The noble gases, located in Group 18, are unique for their extreme inertness. They have a full valence shell (eight electrons, except for helium, which has two), making them exceptionally stable and unreactive. They rarely form chemical compounds.
Key Characteristics of Noble Gases:
- High ionization energy: They resist losing electrons.
- Very low electronegativity: They have virtually no tendency to gain electrons.
- Colorless, odorless, and tasteless gases: Their inert nature contributes to these characteristics.
- Low boiling points: Their weak interatomic forces result in low boiling points.
Notable Noble Gases and Their Applications:
- Helium (He): Used in balloons, cryogenics (low-temperature applications), and in MRI machines due to its inertness and low density.
- Neon (Ne): Known for its bright red-orange glow in neon lights, neon is used in various lighting applications.
- Argon (Ar): Used as an inert atmosphere in welding and metallurgy to prevent oxidation, argon is also found in incandescent light bulbs.
- Krypton (Kr): Used in some lasers and high-intensity lighting, krypton's unique spectral lines are used in precise measurement applications.
- Xenon (Xe): Used in some specialized lighting (high-intensity flash lamps) and in medical imaging, xenon's unique properties make it suitable for certain applications.
- Radon (Rn): A radioactive gas, radon is a health hazard and has no beneficial applications.
Conclusion: The Importance of Understanding Group Properties
Understanding the properties of alkali metals, alkaline earth metals, halogens, and noble gases is crucial for many scientific and technological advancements. Their diverse characteristics lead to a broad spectrum of applications, impacting various aspects of our daily lives. From the batteries in our phones to the lighting in our homes, the properties of these groups of elements are fundamental to modern technology and advancements in various scientific fields. Continued research into these elements promises further breakthroughs and innovations.
Latest Posts
Latest Posts
-
Wavelength Of A Helium Neon Laser
Apr 04, 2025
-
How Do You Write Complex Numbers In Standard Form
Apr 04, 2025
-
Shaft Of The Long Bone Is Called
Apr 04, 2025
-
Example Of Stoichiometry In Real Life
Apr 04, 2025
-
Dna Biology And Technology Dna And Rna Structure
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Alkali Metals Alkaline Earth Metals Halogens And Noble Gases . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
