An Organized Arrangement Of Elements According To Their Atomic Number
Muz Play
Apr 02, 2025 · 6 min read
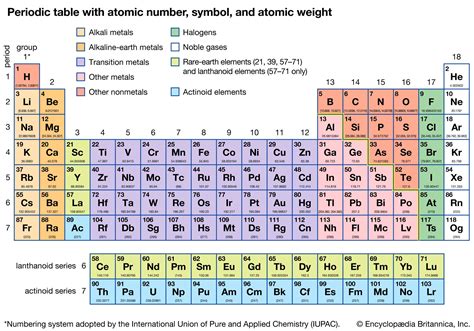
Table of Contents
The Periodic Table: An Organized Arrangement of Elements by Atomic Number
The periodic table, a cornerstone of chemistry and a testament to human understanding of the natural world, represents an organized arrangement of chemical elements based on their atomic number. This seemingly simple organization unlocks a wealth of information about the elements' properties, behaviors, and relationships, providing a powerful tool for predicting and understanding chemical reactions. This article will delve into the intricacies of the periodic table, exploring its structure, history, and the profound implications of its arrangement.
The Genesis of Order: Mendeleev's Vision
Before the modern periodic table, elements were scattered, lacking a unifying principle. Scientists recognized similarities in properties between certain elements, but no systematic organization existed. This changed dramatically with Dmitri Mendeleev in 1869. Mendeleev, a Russian chemist, arranged the known elements in order of increasing atomic weight (a close approximation to atomic number at the time), noticing a recurring pattern of properties. He observed that elements with similar properties appeared at regular intervals. This led him to create the first periodic table, arranging the elements in rows and columns based on their repeating chemical and physical properties.
Mendeleev's Bold Prediction: The Power of Gaps
Mendeleev's genius extended beyond mere organization. He noticed gaps in his table, representing undiscovered elements. Instead of ignoring these gaps, he boldly predicted the properties of these missing elements based on the properties of their neighbors in the table. These predictions, remarkably accurate, proved to be a powerful validation of his system. The subsequent discovery of elements like gallium, scandium, and germanium, with properties closely matching Mendeleev's predictions, solidified the acceptance and importance of his periodic table.
The Modern Periodic Table: Atomic Number Takes Center Stage
While Mendeleev's table was groundbreaking, the modern periodic table is based on atomic number, not atomic weight. The atomic number, representing the number of protons in an atom's nucleus, proved to be the fundamental organizing principle. This shift occurred with the development of modern atomic theory and the understanding of the nucleus. The atomic number uniquely identifies each element, explaining the underlying reason for the recurring properties observed by Mendeleev.
The Structure: Rows, Columns, and Blocks
The periodic table is structured in a grid format:
- Periods (Rows): Horizontal rows represent increasing energy levels of electrons. Elements within the same period have the same number of electron shells.
- Groups (Columns): Vertical columns represent elements with similar outer electron configurations. These similar configurations drive similar chemical behaviors. Elements in the same group often exhibit similar chemical properties and reactivity.
- Blocks: The periodic table is further divided into blocks based on the subshells in which the valence electrons are located (s, p, d, and f blocks). These blocks correlate with specific properties and bonding behaviors.
Understanding the Periodic Trends
The organization of the periodic table allows us to predict several periodic trends, which are systematic changes in element properties as you move across a period or down a group.
1. Atomic Radius: Size Matters
Atomic radius, the distance from the nucleus to the outermost electron, shows a clear trend. Atomic radius generally:
- Increases down a group: The addition of electron shells increases the distance from the nucleus to the outermost electrons.
- Decreases across a period: The increasing nuclear charge pulls the electrons closer to the nucleus, despite the addition of electrons to the same shell.
2. Ionization Energy: The Energy of Loss
Ionization energy is the energy required to remove an electron from an atom. This energy generally:
- Decreases down a group: Outer electrons are further from the nucleus and thus less strongly attracted.
- Increases across a period: The increasing nuclear charge holds outer electrons more tightly.
3. Electronegativity: The Tug of War
Electronegativity measures an atom's ability to attract electrons in a chemical bond. It generally:
- Decreases down a group: Outer electrons are further from the nucleus and less attracted.
- Increases across a period: The increasing nuclear charge increases the attraction for electrons.
4. Electron Affinity: Gaining an Electron
Electron affinity is the energy change when an atom gains an electron. While the trends are less consistent than others, generally:
- Generally decreases down a group.
- Generally increases across a period (with exceptions).
The Significance of Groups: Families of Elements
The groups of the periodic table represent families of elements with similar properties:
- Group 1 (Alkali Metals): Highly reactive metals, readily losing one electron to form +1 ions.
- Group 2 (Alkaline Earth Metals): Reactive metals, losing two electrons to form +2 ions.
- Group 17 (Halogens): Highly reactive nonmetals, readily gaining one electron to form -1 ions.
- Group 18 (Noble Gases): Inert gases with full electron shells, rarely forming compounds.
- Transition Metals (d-block): Exhibit variable oxidation states and form colored compounds.
- Lanthanides and Actinides (f-block): Inner transition metals with similar properties.
Each group’s unique properties stem directly from their electron configurations, highlighting the predictive power of the periodic table's arrangement.
Beyond the Basics: Isotopes and Beyond
While the periodic table primarily organizes elements by atomic number (and therefore number of protons), it's important to remember that elements can have isotopes. Isotopes are atoms of the same element with the same number of protons but a different number of neutrons. This variation in neutron number affects the atom's mass but not its chemical properties, which are primarily determined by the electron configuration.
The periodic table also doesn't explicitly display information about different allotropes of elements. Allotropes are different structural forms of the same element. For example, carbon exists as diamond, graphite, and fullerenes, each with different properties despite being composed solely of carbon atoms.
Applications of the Periodic Table
The periodic table is indispensable across various scientific disciplines:
- Chemistry: Predicting chemical reactions, understanding bonding, and determining reactivity.
- Materials Science: Designing new materials with specific properties.
- Nuclear Physics: Understanding nuclear reactions and radioactive decay.
- Engineering: Selecting materials for specific applications based on their properties.
- Medicine: Understanding the biological roles of elements and developing new drugs.
The table's ability to organize and predict elemental behavior is essential in fields ranging from the development of new technologies to the understanding of fundamental processes in the universe.
The Ongoing Evolution of Understanding
The periodic table, though seemingly static, is a constantly evolving representation of scientific knowledge. As our understanding of atomic structure and chemical behavior deepens, our interpretation and application of the periodic table continue to evolve. New isotopes are discovered, new compounds are synthesized, and our understanding of the relationships between elements is constantly refined. The periodic table serves not only as a summary of current knowledge but also as a dynamic roadmap guiding future scientific explorations.
Conclusion: A Timeless Tool for Discovery
The periodic table, an organized arrangement of elements by atomic number, represents a monumental achievement in scientific understanding. From Mendeleev's insightful vision to the sophisticated understanding of atomic structure that informs its modern form, the periodic table stands as a testament to the power of systematic observation, bold prediction, and the continuous pursuit of scientific knowledge. Its enduring utility across diverse scientific fields underscores its significance as a foundational tool for discovery and innovation, ensuring its continued relevance for generations to come. Its simple yet powerful organization continues to unlock the secrets of the chemical world, guiding us towards a deeper understanding of the matter that makes up our universe.
Latest Posts
Latest Posts
-
What Is A Disadvantage Of A Corporation
Apr 03, 2025
-
What Is Kf In Freezing Point Depression
Apr 03, 2025
-
What Are Some Disadvantages Of A Corporation
Apr 03, 2025
-
Describe How Atp Is Produced In The Light Reactions
Apr 03, 2025
-
What Are Characteristics Of Covalent Compounds
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about An Organized Arrangement Of Elements According To Their Atomic Number . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
