Any Substance That Occupies Space And Has Weight Is ______.
Muz Play
Apr 04, 2025 · 5 min read
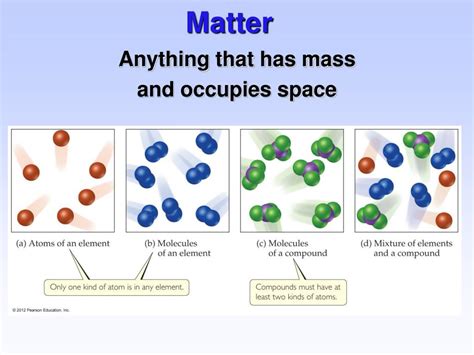
Table of Contents
Any Substance That Occupies Space and Has Weight Is ______: Understanding Matter
The answer to the question, "Any substance that occupies space and has weight is ______," is matter. This seemingly simple definition underpins our understanding of the physical world. From the air we breathe to the stars in the night sky, everything that exists is made up of matter. This article delves deep into the concept of matter, exploring its properties, classifications, and significance in the universe.
Defining Matter: More Than Just Space and Weight
While the definition "occupies space and has weight" provides a good starting point, it's crucial to understand the nuances of matter. Space refers to volume; matter takes up a three-dimensional area. Weight, on the other hand, is a measure of the gravitational force acting on the matter's mass. This means weight can vary depending on the gravitational field (you'd weigh less on the moon), while mass remains constant.
Therefore, a more precise definition of matter considers both its mass and its volume. Matter possesses inertia, meaning it resists changes in motion. This inertia is directly related to its mass. The combination of mass and volume allows us to calculate density, a crucial property distinguishing different types of matter.
Key Characteristics of Matter
- Mass: A fundamental property representing the amount of matter in an object. It's a measure of inertia and is independent of gravitational forces.
- Volume: The amount of three-dimensional space occupied by matter.
- Density: The mass per unit volume of a substance. Dense materials pack more mass into a given volume.
- Inertia: The resistance of an object to changes in its state of motion. More massive objects have greater inertia.
States of Matter: Solid, Liquid, Gas, and Beyond
Matter exists in various states, each characterized by its unique properties and arrangement of particles. The most common states are:
1. Solid
Solids have a definite shape and volume. Their particles are tightly packed in a fixed, ordered arrangement, resulting in strong intermolecular forces. This explains their rigidity and resistance to compression. Examples include ice, rocks, and wood.
Key Properties of Solids:
- Fixed shape: Maintains its shape regardless of container.
- Fixed volume: Doesn't change volume easily.
- Incompressible: Resistant to compression.
- High density: Particles are closely packed.
2. Liquid
Liquids have a definite volume but no definite shape. Their particles are closer together than in gases but less so than in solids. They can flow and take the shape of their container. Examples include water, oil, and mercury.
Key Properties of Liquids:
- Variable shape: Takes the shape of its container.
- Fixed volume: Maintains a constant volume.
- Relatively incompressible: Slightly compressible compared to solids.
- Moderate density: Particles are relatively close together.
3. Gas
Gases have neither a definite shape nor a definite volume. Their particles are widely dispersed and move freely, resulting in weak intermolecular forces. They are easily compressible and expand to fill their container. Examples include air, oxygen, and carbon dioxide.
Key Properties of Gases:
- Variable shape: Expands to fill its container.
- Variable volume: Changes volume easily with changes in pressure and temperature.
- Highly compressible: Easily compressed into smaller volumes.
- Low density: Particles are far apart.
4. Plasma
Plasma is often considered the fourth state of matter. It's a highly energized state where electrons are stripped from atoms, forming a mixture of ions and free electrons. Plasma is found in stars, lightning, and fluorescent lights.
Key Properties of Plasma:
- Highly ionized: Contains significant numbers of free ions and electrons.
- High conductivity: Conducts electricity very well.
- Variable shape and volume: Similar to gases but with unique electrical properties.
5. Bose-Einstein Condensate (BEC)
At extremely low temperatures, certain atoms can behave as a single quantum entity, forming a BEC. It's a state of matter where a large number of atoms occupy the same quantum state, exhibiting macroscopic quantum phenomena.
Key Properties of BEC:
- Superfluidity: Flows without viscosity.
- Macroscopic quantum phenomena: Exhibits properties typically only observed at the atomic level.
- Extremely low temperature: Exists only at temperatures near absolute zero.
Classifications of Matter: Pure Substances and Mixtures
Matter can also be classified into pure substances and mixtures.
1. Pure Substances
Pure substances have a fixed chemical composition and uniform properties throughout. They cannot be separated into simpler substances by physical methods. Pure substances can be further categorized into:
- Elements: Substances that cannot be broken down into simpler substances by chemical means. Examples include hydrogen, oxygen, and iron.
- Compounds: Substances formed by the chemical combination of two or more elements in fixed proportions. Examples include water (H₂O) and table salt (NaCl).
2. Mixtures
Mixtures are combinations of two or more substances that are not chemically bonded. They have variable compositions and properties that depend on the proportions of their components. Mixtures can be separated into their components by physical methods. Examples include air, seawater, and soil. Mixtures can be further classified into:
- Homogeneous mixtures: Uniform in composition throughout. Examples include saltwater and air.
- Heterogeneous mixtures: Not uniform in composition; different components are visibly distinct. Examples include sand and water, and a salad.
The Significance of Matter in the Universe
Matter is fundamental to the existence of everything we see and interact with in the universe. Its properties and transformations drive all natural processes. The study of matter, therefore, is crucial to understanding:
- Cosmology: Understanding the formation and evolution of the universe, from the Big Bang to the formation of stars and galaxies.
- Chemistry: The study of matter's composition, structure, properties, and reactions.
- Physics: The study of matter's interactions with energy and forces.
- Biology: The study of living organisms, which are complex systems composed of matter.
- Materials Science: The development and application of new materials with specific properties.
Conclusion: The Ever-Expanding World of Matter
In conclusion, the statement "Any substance that occupies space and has weight is matter" provides a foundational understanding of this fundamental concept. However, exploring the nuances of matter—its properties, states, and classifications—reveals a far richer and more complex picture. From the solid ground beneath our feet to the gaseous expanse of space, matter is the building block of our universe, and its continuous study remains essential to scientific progress and technological advancement. Further exploration into the subatomic particles that constitute matter opens even more intriguing possibilities and avenues of research. The journey of understanding matter is an ongoing one, with new discoveries continually reshaping our comprehension of the physical world.
Latest Posts
Latest Posts
-
Highness Or Lowness Of A Sound
Apr 04, 2025
-
What Is The Number Of Protons For Sulfur
Apr 04, 2025
-
What Is The Atomic Radius Of Hydrogen
Apr 04, 2025
-
What Are The Two Kinds Of Matter
Apr 04, 2025
-
How Is A Subsatnce Saruaryed On A Graph
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Any Substance That Occupies Space And Has Weight Is ______. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
