What Is The Atomic Radius Of Hydrogen
Muz Play
Apr 04, 2025 · 6 min read
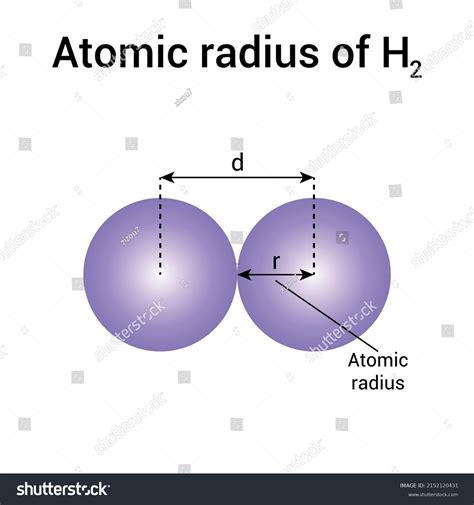
Table of Contents
What is the Atomic Radius of Hydrogen? A Deep Dive into the Quantum Realm
The seemingly simple question, "What is the atomic radius of hydrogen?" opens a fascinating window into the complexities of atomic structure and the challenges of defining size at the quantum level. Unlike macroscopic objects with clearly defined boundaries, atoms, and particularly their electron clouds, lack sharp edges. Therefore, the atomic radius isn't a single, universally agreed-upon value but rather a concept with several interpretations and resulting values depending on the method of measurement. This article will explore the various ways to determine hydrogen's atomic radius, the factors influencing its size, and the implications of this fundamental property.
Understanding Atomic Radius: A Quantum Conundrum
Before delving into the specifics of hydrogen, let's establish a foundational understanding of atomic radius. It's a measure of the size of an atom, but the difficulty arises from the probabilistic nature of electron location. Electrons don't orbit the nucleus in neat, predictable paths like planets around a sun. Instead, they exist in orbitals, regions of space where the probability of finding an electron is high. This means the atom's "edge" is fuzzy and depends on how we choose to define it.
Several methods attempt to quantify this fuzzy edge:
-
Covalent Radius: This measures half the distance between the nuclei of two identical atoms bonded together covalently. For hydrogen, this is a relatively straightforward measurement because hydrogen readily forms covalent bonds (e.g., in H₂).
-
Metallic Radius: This applies to metallic elements and represents half the distance between adjacent nuclei in a metallic crystal lattice. Hydrogen, being non-metallic, doesn't have a metallic radius in the traditional sense. However, under extreme pressure, hydrogen can exhibit metallic properties, leading to a metallic radius in these specific, high-pressure conditions.
-
Van der Waals Radius: This represents half the distance between the nuclei of two identical non-bonded atoms that are very close together. Van der Waals forces, weak intermolecular attractions, determine this distance. It's relevant to hydrogen in its interactions with other non-bonded atoms or molecules.
-
Ionic Radius: This pertains to ions – atoms that have gained or lost electrons, resulting in a net positive or negative charge. For hydrogen, this typically refers to the H⁺ ion (a proton), which has a significantly smaller radius than neutral hydrogen due to the absence of an electron.
The Challenges of Defining Hydrogen's Atomic Radius
Hydrogen, being the simplest atom with just one proton and one electron, presents unique challenges. Its single electron is highly susceptible to external influences, making its orbital size dynamic and context-dependent. Furthermore, the different methods for determining atomic radius yield different values:
-
Covalent Radius: The covalent radius of hydrogen is typically cited as around 37 pm (picometers, 1 pm = 10⁻¹² m). This is a widely accepted value, but even this "simple" measurement has inherent uncertainties due to the quantum nature of the electron cloud.
-
Van der Waals Radius: The Van der Waals radius for hydrogen is slightly larger than its covalent radius, often reported to be in the range of 120 pm. This larger value reflects the greater distance between non-bonded hydrogen atoms compared to those sharing electrons in a covalent bond.
-
Ionic Radius (H⁺): The ionic radius of the proton (H⁺) is extremely small, on the order of 1.5 pm. This tiny size is a result of the electron being removed, leaving only the positively charged proton.
Factors Influencing Hydrogen's Atomic Radius
Several factors contribute to the variability in reported atomic radii for hydrogen:
-
Quantum Mechanics: The inherent uncertainty principle in quantum mechanics prevents us from knowing both the electron's position and momentum with perfect precision. This probabilistic nature of electron location directly affects our ability to define a precise atomic radius.
-
Electron-Electron Repulsion (in multi-electron atoms): While hydrogen has only one electron, this concept is crucial when comparing it to other elements. In multi-electron atoms, electron-electron repulsion influences the size of the atom; the greater the repulsion, the larger the atom tends to be. Hydrogen, lacking this repulsive force, has a smaller size than other atoms.
-
Effective Nuclear Charge: The effective nuclear charge represents the net positive charge experienced by an electron, considering the shielding effect of other electrons. In hydrogen, with only one electron, the effective nuclear charge is simply the charge of the proton (+1). In larger atoms, the inner electrons shield outer electrons from the full positive charge of the nucleus, reducing the effective nuclear charge experienced by the outer electrons.
-
Bonding Environment: The environment in which hydrogen is found significantly impacts its apparent size. Whether it's in a covalent bond, a van der Waals interaction, or an ionic compound, the electron distribution changes, altering the measured radius.
-
Pressure and Temperature: Under extreme conditions of high pressure and temperature, as mentioned earlier, hydrogen can transition to a metallic state, dramatically altering its atomic radius. This transition is being actively studied in the search for metallic hydrogen, a hypothetical material with potentially revolutionary properties.
Applications and Implications
Understanding hydrogen's atomic radius, despite its inherent challenges, has broad implications across various scientific fields:
-
Chemistry: The size of hydrogen atoms significantly influences chemical bonding, reactivity, and the structure of molecules. Its small size allows it to readily form covalent bonds and participate in various chemical reactions.
-
Physics: The study of hydrogen's atomic structure is crucial for understanding fundamental principles of quantum mechanics, including the wave-particle duality of electrons and the uncertainty principle.
-
Materials Science: The research into metallic hydrogen and its potential properties depends on a detailed understanding of how hydrogen's atomic radius changes under high pressure.
-
Astrophysics: Hydrogen, the most abundant element in the universe, plays a crucial role in stellar processes. Understanding its properties, including its atomic radius, is fundamental to comprehending stellar evolution and the formation of elements heavier than hydrogen.
Conclusion: A Continuing Quest for Precision
The question of hydrogen's atomic radius highlights the limitations of classical models when applied to the quantum world. While there isn't a single definitive answer, various methods and considerations provide a range of values, each with its specific context and implications. Understanding these nuances and the factors influencing hydrogen's apparent size is critical for advancing our knowledge in chemistry, physics, materials science, and astrophysics. The ongoing quest for a more precise understanding of atomic radii, including hydrogen's, continues to push the boundaries of scientific inquiry and reveals the rich complexities of the atomic realm. Further research, particularly utilizing advanced computational methods and experimental techniques, promises to provide even more refined measurements and insights into this fundamental property of matter.
Latest Posts
Latest Posts
-
Color To Emphasize The Focal Point
Apr 05, 2025
-
Why Is Carbon A Special Element
Apr 05, 2025
-
Boundary Between The Crust And The Mantle
Apr 05, 2025
-
Acetic Acid Sodium Acetate Buffer Equation
Apr 05, 2025
-
What Is The Standard Electrode Potential
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about What Is The Atomic Radius Of Hydrogen . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
