What Is The Standard Electrode Potential
Muz Play
Apr 05, 2025 · 6 min read
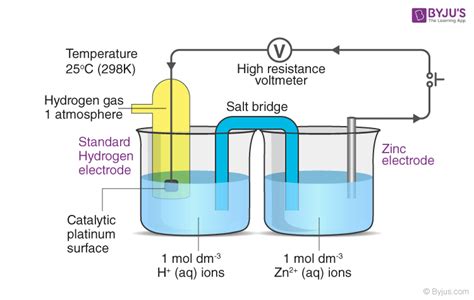
Table of Contents
What is Standard Electrode Potential? A Comprehensive Guide
Standard electrode potential, often denoted as E⁰, is a crucial concept in electrochemistry. It represents the measure of the individual potential of a reversible electrode at standard conditions relative to a standard hydrogen electrode (SHE). Understanding standard electrode potential is key to predicting the spontaneity of redox reactions, designing electrochemical cells, and interpreting electrochemical phenomena. This comprehensive guide delves into the intricacies of standard electrode potential, its determination, applications, and limitations.
Understanding the Basics: Redox Reactions and Electrode Potentials
Before diving into standard electrode potential, it's essential to grasp the fundamentals of redox reactions. Redox reactions, short for reduction-oxidation reactions, involve the transfer of electrons between species. One species undergoes oxidation (loss of electrons), while another undergoes reduction (gain of electrons). These reactions are fundamental to numerous processes, from corrosion to biological energy production.
Electrodes play a vital role in these reactions. An electrode is a conductive material that facilitates electron transfer between the electrode and the solution. The potential difference between an electrode and its solution is termed the electrode potential. This potential arises from the tendency of the electrode material to lose or gain electrons.
What Makes it "Standard"? Defining Standard Conditions
The term "standard" in standard electrode potential specifies the precise conditions under which the potential is measured. These standard conditions ensure reproducibility and allow comparison between different electrode potentials. These conditions are:
- Temperature: 298 K (25°C)
- Concentration: 1 M (molar) for all ionic species involved. For gases, the partial pressure is 1 atm (atmosphere).
- Pressure: 1 atm (atmosphere).
- Activity: Ideally, activity (effective concentration) should be used instead of concentration, but at low concentrations, concentration is a reasonable approximation.
It's crucial to remember that deviations from standard conditions will alter the electrode potential. The Nernst equation, discussed later, helps quantify this effect.
The Standard Hydrogen Electrode (SHE): The Reference Point
The standard hydrogen electrode (SHE), also known as the normal hydrogen electrode (NHE), serves as the reference point for measuring standard electrode potentials. It consists of a platinum electrode immersed in a 1 M solution of H⁺ ions, with hydrogen gas at 1 atm bubbling over the platinum surface. The half-cell reaction for the SHE is:
2H⁺(aq) + 2e⁻ ⇌ H₂(g)
By convention, the standard electrode potential of the SHE is assigned a value of 0 volts. All other electrode potentials are then measured relative to this reference.
Measuring Standard Electrode Potential: The Electrochemical Cell
Standard electrode potential is determined experimentally using an electrochemical cell. An electrochemical cell consists of two half-cells: one containing the electrode of interest and another containing the SHE. The two half-cells are connected by a salt bridge, which allows ion flow to maintain electrical neutrality. A voltmeter measures the potential difference between the two electrodes.
The potential difference measured is the difference in standard electrode potentials between the two half-cells. Since the SHE has a potential of 0 V, the measured potential directly represents the standard electrode potential of the electrode of interest.
For example, if a cell is constructed with a copper electrode and the SHE, and a positive potential is measured, it indicates that the copper electrode has a positive standard electrode potential compared to the SHE. This implies that copper ions have a stronger tendency to gain electrons (reduction) than hydrogen ions.
Interpreting Standard Electrode Potentials: Predicting Spontaneity
Standard electrode potentials provide valuable information for predicting the spontaneity of redox reactions. A positive standard electrode potential indicates that the reduction half-reaction is favored under standard conditions. Conversely, a negative standard electrode potential signifies that the oxidation half-reaction is favored.
To determine the spontaneity of a complete redox reaction, consider the difference in standard electrode potentials between the two half-reactions. A positive cell potential (E°cell) indicates that the reaction is spontaneous under standard conditions, while a negative cell potential indicates a non-spontaneous reaction. The cell potential is calculated as:
E°cell = E°reduction (cathode) - E°reduction (anode)
The cathode is the electrode where reduction occurs, while the anode is the electrode where oxidation occurs.
The Nernst Equation: Accounting for Non-Standard Conditions
The Nernst equation is a crucial tool that extends the concept of standard electrode potential to non-standard conditions. It allows for the calculation of the electrode potential when the concentrations or pressures deviate from standard values. The equation is:
E = E⁰ - (RT/nF)lnQ
Where:
- E is the electrode potential under non-standard conditions.
- E⁰ is the standard electrode potential.
- R is the ideal gas constant (8.314 J/mol·K).
- T is the temperature in Kelvin.
- n is the number of electrons transferred in the half-reaction.
- F is the Faraday constant (96485 C/mol).
- Q is the reaction quotient, which is the ratio of the concentrations (or partial pressures) of products to reactants raised to the power of their stoichiometric coefficients.
The Nernst equation is essential for understanding the behavior of electrochemical cells under various conditions, such as those found in batteries and fuel cells.
Applications of Standard Electrode Potential: Beyond the Basics
Standard electrode potentials have widespread applications across various scientific and technological fields. Some key applications include:
- Predicting the spontaneity of redox reactions: As discussed earlier, this is fundamental to understanding electrochemical processes.
- Designing electrochemical cells: Standard electrode potentials guide the selection of appropriate electrodes for constructing batteries, fuel cells, and other electrochemical devices. Choosing electrodes with a significant potential difference ensures sufficient voltage output.
- Corrosion prediction: Standard electrode potentials help predict the susceptibility of materials to corrosion. Metals with more negative standard electrode potentials are more prone to corrosion.
- Electroplating and electrorefining: These processes utilize the principles of standard electrode potential to deposit or purify metals.
- Analytical chemistry: Electrochemical techniques, such as potentiometry, rely heavily on standard electrode potentials for quantitative analysis.
Limitations of Standard Electrode Potential: Addressing the Caveats
While standard electrode potential is a powerful tool, it's essential to acknowledge its limitations:
- Standard conditions are idealized: Real-world conditions rarely match the strict standard conditions. The Nernst equation helps account for deviations, but it also introduces approximations.
- Kinetic factors are neglected: Standard electrode potential only provides thermodynamic information about the spontaneity of a reaction, not its rate. A reaction may be thermodynamically favorable but kinetically slow.
- Complex systems: The application of standard electrode potential to complex systems, such as biological systems, can be challenging. The presence of multiple redox couples and other interfering species can complicate the analysis.
- Overpotential: Overpotential is the extra voltage needed to drive a reaction beyond its thermodynamic potential. This arises from factors such as slow electron transfer kinetics or the formation of gas bubbles on the electrode surface. Overpotential isn't accounted for by standard electrode potential.
Conclusion: A Powerful Tool in Electrochemistry
Standard electrode potential is a cornerstone concept in electrochemistry, offering valuable insights into the behavior of redox reactions and electrochemical cells. Its applications are vast, ranging from predicting the spontaneity of reactions to designing electrochemical devices. While limitations exist, especially when moving away from ideal conditions, the understanding and application of standard electrode potential remain indispensable for comprehending and manipulating the intricate world of electrochemical processes. Further exploration into electrochemical kinetics and the intricacies of electrode reactions will provide a more complete understanding of the complexities of electrochemical systems. The Nernst equation, in tandem with a grasp of standard electrode potential, offers a powerful framework for comprehending and applying these crucial electrochemical principles.
Latest Posts
Latest Posts
-
How Do I Multiply Rational Expressions
Apr 05, 2025
-
How Does A Competitive Inhibitor Slow Enzyme Catalysis
Apr 05, 2025
-
What Is The Difference Between An Equation And Inequality
Apr 05, 2025
-
Does Electric Field Go From Positive To Negative
Apr 05, 2025
-
What Tools Are Used To Measure Humidity
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about What Is The Standard Electrode Potential . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
