Bases Produce Which Ions In Aqueous Solution
Muz Play
Apr 03, 2025 · 6 min read
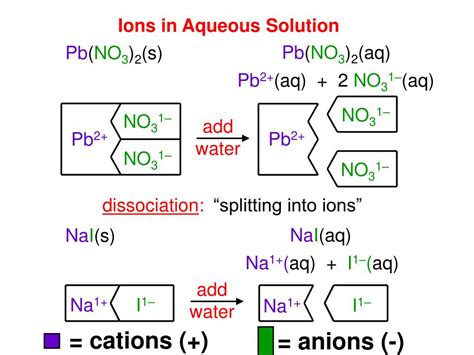
Table of Contents
Bases Produce Which Ions in Aqueous Solution? A Deep Dive into Arrhenius, Brønsted-Lowry, and Lewis Definitions
Understanding which ions bases produce in aqueous solution is fundamental to grasping the concepts of acidity and basicity in chemistry. While the simple answer often points to hydroxide ions (OH⁻), the reality is more nuanced, depending on the definition of a base used. This article will explore the different definitions of bases – Arrhenius, Brønsted-Lowry, and Lewis – and delve into the ions they produce in aqueous solution, clarifying common misconceptions and providing a comprehensive understanding of this crucial chemical concept.
The Arrhenius Definition: A Foundation for Understanding
The Arrhenius definition, one of the earliest and simplest models, defines a base as a substance that increases the concentration of hydroxide ions (OH⁻) when dissolved in water. This definition is straightforward and easily applicable to many common bases.
Examples of Arrhenius Bases and Their Ion Production
Strong Arrhenius bases, like sodium hydroxide (NaOH) and potassium hydroxide (KOH), completely dissociate in water, producing a high concentration of hydroxide ions:
- NaOH(s) → Na⁺(aq) + OH⁻(aq)
- KOH(s) → K⁺(aq) + OH⁻(aq)
These reactions illustrate the key feature of Arrhenius bases: they directly release hydroxide ions into the solution, thereby increasing its pH.
Weak Arrhenius bases, such as ammonia (NH₃), only partially dissociate, resulting in a lower concentration of hydroxide ions:
- NH₃(aq) + H₂O(l) ⇌ NH₄⁺(aq) + OH⁻(aq)
Notice the equilibrium arrow (⇌) indicating that the reaction is reversible and doesn't proceed to completion. This partial dissociation is why weak bases have a lower pH change compared to strong bases.
Key takeaway: According to the Arrhenius definition, the key ion produced by a base in aqueous solution is the hydroxide ion (OH⁻).
Expanding the Horizons: The Brønsted-Lowry Definition
The Brønsted-Lowry definition offers a broader perspective on basicity. It defines a base as a proton acceptor, meaning a substance that accepts a hydrogen ion (H⁺) or a proton. This definition expands the scope of bases beyond those that directly produce hydroxide ions.
Brønsted-Lowry Bases and Ion Production: Beyond Hydroxide
Many Brønsted-Lowry bases don't directly produce hydroxide ions but instead react with water to form them. Consider ammonia again:
- NH₃(aq) + H₂O(l) ⇌ NH₄⁺(aq) + OH⁻(aq)
In this reaction, ammonia accepts a proton from water, acting as a Brønsted-Lowry base. Water, in turn, donates a proton and acts as a Brønsted-Lowry acid. The hydroxide ion is formed as a product of this proton transfer.
Other examples include carbonate ions (CO₃²⁻) and bicarbonate ions (HCO₃⁻). These ions accept protons from water, generating hydroxide ions:
- CO₃²⁻(aq) + H₂O(l) ⇌ HCO₃⁻(aq) + OH⁻(aq)
- HCO₃⁻(aq) + H₂O(l) ⇌ H₂CO₃(aq) + OH⁻(aq)
Crucially, some Brønsted-Lowry bases don't produce hydroxide ions directly. Instead, they produce their conjugate acid. The production of hydroxide ions is a consequence of their reaction with water.
Key takeaway: The Brønsted-Lowry definition broadens the understanding of bases. While hydroxide ions are often produced in aqueous solution through the reaction with water, the fundamental characteristic of a Brønsted-Lowry base is its ability to accept a proton.
The Most Comprehensive View: The Lewis Definition
The Lewis definition provides the most inclusive understanding of basicity. A Lewis base is defined as an electron-pair donor. This means a Lewis base provides a lone pair of electrons to form a coordinate covalent bond with an electron-deficient species, often called a Lewis acid.
Lewis Bases and Ion Production: The Electron Pair Perspective
This definition encompasses a much wider range of substances than the Arrhenius and Brønsted-Lowry definitions. Many Lewis bases, in aqueous solution, can still indirectly lead to hydroxide ion production by reacting with water or other species that can subsequently produce hydroxide ions. However, some Lewis bases do not produce hydroxide ions at all.
For instance, consider ammonia again. Ammonia acts as a Lewis base because it has a lone pair of electrons that can be donated to a Lewis acid:
- NH₃(aq) + H⁺(aq) → NH₄⁺(aq)
In this reaction, ammonia donates its lone pair to a proton (H⁺), forming the ammonium ion (NH₄⁺). While this doesn't directly produce hydroxide ions, the subsequent reaction of the ammonium ion with water can lead to the formation of hydroxide ions. However, it's essential to understand that the ammonia acts as a Lewis base regardless of whether hydroxide ions are produced.
Other examples include fluoride ions (F⁻), chloride ions (Cl⁻), and even neutral molecules like water (H₂O) that possess lone pairs and can act as Lewis bases in various reactions.
Key takeaway: The Lewis definition is the most general, considering the electron pair donation capacity. While hydroxide ions might be indirectly produced in some cases, the defining characteristic of a Lewis base is its ability to donate an electron pair.
Comparing the Three Definitions: A Summary Table
| Definition | Defining Characteristic | Ions Produced in Aqueous Solution (Directly or Indirectly) | Examples |
|---|---|---|---|
| Arrhenius | Increases [OH⁻] | Primarily OH⁻ | NaOH, KOH, NH₃ (indirectly) |
| Brønsted-Lowry | Proton acceptor | Often OH⁻ (indirectly through reaction with water), but not always | NH₃, CO₃²⁻, HCO₃⁻ |
| Lewis | Electron-pair donor | May or may not produce OH⁻; Depends on the specific reaction and the solvent. | NH₃, F⁻, Cl⁻, H₂O, many others |
Practical Implications and Applications
Understanding the different definitions and the resulting ion production is crucial in various applications:
- pH calculations: Knowing which ions a base produces is essential for accurately calculating the pH of a solution.
- Titrations: Acid-base titrations rely on the reaction between acids and bases, and understanding the ion production helps in selecting appropriate indicators and interpreting results.
- Buffer solutions: Buffer solutions are prepared by mixing weak acids and their conjugate bases (or weak bases and their conjugate acids). The ion production characteristics of the base are crucial for determining buffer capacity and pH range.
- Industrial processes: Many industrial processes, such as the production of soaps and detergents, involve base-catalyzed reactions, and understanding the ion production is crucial for optimizing the process.
- Environmental chemistry: The effects of bases on the environment often depend on the ions they produce. For instance, hydroxide ions can impact soil pH and water quality.
Conclusion: A Holistic Understanding of Bases
In conclusion, while the simple answer to "bases produce which ions in aqueous solution?" often points to hydroxide ions, a deeper understanding requires considering the different definitions of bases. The Arrhenius definition focuses on hydroxide ion production, while the Brønsted-Lowry definition highlights proton acceptance, and the Lewis definition emphasizes electron pair donation. The ions produced can vary depending on the base and its reaction with the solvent and other species present. A comprehensive understanding of these nuances is essential for accurate interpretation of chemical reactions and various applications across different scientific disciplines. This broad understanding goes beyond rote memorization and encourages a deeper engagement with fundamental chemical principles.
Latest Posts
Latest Posts
-
Ionic Compound For Sodium And Sulfur
Apr 04, 2025
-
Cells Are Basic Unit Of Life
Apr 04, 2025
-
Two Bones That Form The Nasal Septum
Apr 04, 2025
-
How To Determine The Age Of A Fossil
Apr 04, 2025
-
The Nucleus Of An Atom Consists Of
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Bases Produce Which Ions In Aqueous Solution . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
