Classify The Complex Ions As High Spin Or Low Spin
Muz Play
Apr 02, 2025 · 7 min read
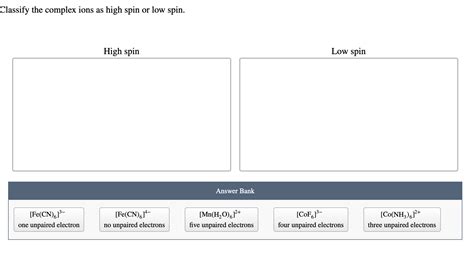
Table of Contents
Classifying Complex Ions as High Spin or Low Spin: A Comprehensive Guide
Understanding the electronic configuration of complex ions is crucial in inorganic chemistry. A key aspect of this understanding lies in classifying them as either high spin or low spin. This classification directly impacts the magnetic properties, color, and reactivity of the complex. This comprehensive guide will delve into the factors determining high spin versus low spin configurations, providing a clear and detailed explanation with examples.
The Crystal Field Theory: A Foundation for Understanding Spin States
The behavior of transition metal complexes is often explained using crystal field theory (CFT). CFT simplifies the interaction between the metal ion and the ligands by considering the ligands as point charges that repel the d electrons of the central metal ion. This repulsion causes the degeneracy of the d orbitals to be lifted, splitting them into different energy levels. The magnitude of this splitting, denoted as Δo (octahedral splitting) or Δt (tetrahedral splitting), is crucial in determining whether a complex will be high spin or low spin.
The Importance of Δo and Δt: The Energy Gap
The energy difference, Δo or Δt, between the split d orbitals dictates how the electrons will fill these orbitals. This energy difference is influenced by several factors:
-
The nature of the ligand: Strong field ligands (e.g., CN⁻, CO, NO₂) create a large Δo/Δt, leading to a large energy gap between the d orbitals. Weak field ligands (e.g., I⁻, Br⁻, Cl⁻) create a small Δo/Δt, resulting in a smaller energy gap. This is often summarized in the spectrochemical series, which arranges ligands in order of increasing field strength.
-
The oxidation state of the metal ion: A higher oxidation state generally leads to a larger Δo/Δt because the increased positive charge on the metal ion attracts the electrons from the ligands more strongly.
-
The geometry of the complex: Octahedral complexes typically have a larger splitting than tetrahedral complexes (Δo > Δt). Other geometries have their own characteristic splitting patterns.
High Spin vs. Low Spin: Electron Configuration and Hund's Rule
The filling of the split d orbitals is governed by two competing factors:
-
Hund's rule: Electrons will individually occupy each orbital within a subshell before pairing up. This minimizes electron-electron repulsion and maximizes spin multiplicity.
-
The magnitude of Δo/Δt: If the energy gap is large (strong field ligands), it is energetically more favorable for electrons to pair up in the lower energy orbitals, even if it violates Hund's rule. If the energy gap is small (weak field ligands), it's more favorable to fill each orbital singly, following Hund's rule.
High Spin Complexes: Maximizing Spin Multiplicity
In high spin complexes, the energy gap (Δo/Δt) is smaller than the pairing energy (P), the energy required to force two electrons into the same orbital. Therefore, electrons will individually occupy all available orbitals before pairing up, maximizing the total spin. This results in a greater number of unpaired electrons and a higher overall magnetic moment.
Example: Consider a tetrahedral [FeCl₄]²⁻ complex. Fe²⁺ has 6 d electrons. Since Cl⁻ is a weak field ligand, Δt is small. The electrons will fill the orbitals according to Hund's rule, resulting in four unpaired electrons. This is a high spin complex.
Low Spin Complexes: Minimizing Energy
In low spin complexes, the energy gap (Δo/Δt) is larger than the pairing energy (P). In this case, it's energetically more favorable for electrons to pair up in the lower energy orbitals, even if it means violating Hund's rule. This minimizes the overall energy of the complex, resulting in fewer unpaired electrons and a lower overall magnetic moment.
Example: Consider an octahedral [Fe(CN)₆]⁴⁻ complex. Fe²⁺ again has 6 d electrons. However, CN⁻ is a strong field ligand, resulting in a large Δo. The electrons will fill the lower energy orbitals first, pairing up before occupying the higher energy orbitals. This results in only two unpaired electrons, making it a low spin complex.
Factors Influencing High Spin vs. Low Spin Behavior: A Deeper Dive
The classification of a complex ion as high spin or low spin is a delicate balance between several factors. Let's examine these factors in more detail:
1. Ligand Field Strength: The Spectrochemical Series
The spectrochemical series is a critical tool for predicting the spin state of a complex. It orders ligands according to their ability to cause d-orbital splitting. The series is not absolute and can vary slightly depending on the metal ion and other factors. A simplified version of the spectrochemical series is:
I⁻ < Br⁻ < S²⁻ < SCN⁻ < Cl⁻ < NO₃⁻ < N₃⁻ < F⁻ < OH⁻ < C₂O₄²⁻ < H₂O < NCS⁻ < CH₃CN < py < NH₃ < en < bipy < phen < NO₂⁻ < PPh₃ < CN⁻ < CO
Strong field ligands (right side of the series): These ligands create a large Δo/Δt, favoring low spin complexes.
Weak field ligands (left side of the series): These ligands create a small Δo/Δt, favoring high spin complexes.
2. Metal Ion and its Oxidation State
The metal ion and its oxidation state play a significant role in determining the spin state. Generally:
-
Higher oxidation states: Higher oxidation states lead to stronger metal-ligand interactions and a larger Δo/Δt, favoring low spin complexes.
-
d⁴-d⁷ metal ions: These ions are particularly susceptible to exhibiting both high and low spin configurations, depending on the ligand field strength. d¹, d², d³, d⁸, d⁹, and d¹⁰ ions typically exhibit only high spin or low spin depending on ligand strength.
3. Geometry of the Complex
The geometry of the complex influences the magnitude of the d-orbital splitting. Octahedral complexes generally have a larger Δo than tetrahedral complexes (Δo > Δt). Square planar complexes have a more complex splitting pattern. This difference in splitting can affect the spin state.
4. Pairing Energy (P)
The pairing energy (P) represents the energy required to force two electrons into the same orbital. This energy is influenced by electron-electron repulsion. A larger pairing energy makes high spin configurations more likely.
Predicting Spin States: A Practical Approach
Predicting the spin state of a complex ion involves a combination of understanding the factors discussed above. Here’s a step-by-step approach:
-
Identify the metal ion and its oxidation state. This determines the number of d electrons.
-
Identify the ligands and their positions in the spectrochemical series. Determine whether they are strong or weak field ligands.
-
Determine the geometry of the complex. This influences the magnitude of the splitting.
-
Consider the relative magnitudes of Δo/Δt and P. For octahedral complexes, if the ligand is strong field (large Δo), it is low spin; if the ligand is weak field (small Δo), it is high spin. For tetrahedral complexes, generally, weak-field ligands dominate, leading to high spin configurations.
-
Fill the d orbitals based on Hund's rule and the relative magnitudes of Δo/Δt and P. This will give you the number of unpaired electrons and thus the spin state.
Applications and Importance of Spin State Classification
The classification of complex ions as high spin or low spin has significant implications in various fields:
-
Magnetic properties: High spin complexes have a higher magnetic moment due to a greater number of unpaired electrons. This is important in applications like MRI contrast agents and magnetic materials.
-
Catalysis: The spin state of a metal complex can influence its catalytic activity. Different spin states can lead to different reaction mechanisms and selectivities.
-
Spectroscopy: The electronic transitions between d orbitals are affected by the spin state. This allows for the determination of spin state through techniques like UV-Vis spectroscopy.
-
Color: The color of transition metal complexes is directly related to the electronic transitions between d orbitals, which is influenced by the spin state.
-
Biological systems: Many biological molecules involve transition metal complexes, and their spin state plays a vital role in their function. For example, the spin state of iron in hemoglobin is crucial for oxygen transport.
Conclusion
Classifying complex ions as high spin or low spin is a fundamental concept in inorganic chemistry. Understanding the factors that govern this classification—ligand field strength, metal ion properties, geometry, and pairing energy—allows for the prediction of magnetic properties, color, and reactivity. This knowledge is crucial for applications ranging from catalysis to biological systems and material science. By carefully considering the interplay of these factors, we can gain a deeper understanding of the rich and varied chemistry of transition metal complexes.
Latest Posts
Latest Posts
-
How To Find Base Of Parallelogram
Apr 03, 2025
-
Chloroplast In Plant Cell Under Microscope
Apr 03, 2025
-
Contribution Of John Newlands In Periodic Table
Apr 03, 2025
-
Measure Of The Amount Of Matter
Apr 03, 2025
-
Bases Produce Which Ions In Aqueous Solution
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Classify The Complex Ions As High Spin Or Low Spin . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
