Consider This Step In A Radical Reaction:
Muz Play
Apr 05, 2025 · 5 min read
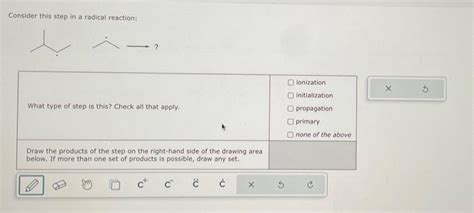
Table of Contents
Consider This Step in a Radical Reaction: A Deep Dive into Reaction Mechanisms and Selectivity
Radical reactions, characterized by the presence of unpaired electrons, are a cornerstone of organic chemistry, underpinning numerous industrial processes and synthetic strategies. Understanding the intricacies of these reactions is crucial for predicting outcomes and designing efficient synthetic pathways. This article delves into a critical aspect of radical reactions: the propagation step. We will explore its mechanism, influencing factors, and the implications for reaction selectivity, providing a comprehensive guide for both students and seasoned chemists.
Understanding Radical Reactions: Initiation, Propagation, and Termination
Before focusing on the propagation step, let's briefly review the fundamental stages of a radical chain reaction:
1. Initiation: The Genesis of Radicals
Initiation involves the generation of radical species, often through homolytic bond cleavage. This process usually requires an external energy source, such as heat, light (photochemical initiation), or a radical initiator (e.g., peroxides, azo compounds). A typical example is the homolytic cleavage of a peroxide bond:
(RO-OR) → 2 RO•
This generates two alkoxy radicals (RO•), each possessing an unpaired electron.
2. Propagation: The Chain Reaction
This is where the magic happens. The propagation step involves a series of reactions where radicals react with non-radical molecules to produce new radicals and continue the chain reaction. This step is crucial because it's responsible for the majority of product formation. It typically consists of two or more steps, each involving a radical reacting with a non-radical molecule and generating a new radical that can then participate in further propagation steps. The exact nature of these steps depends heavily on the specific reaction and the reactants involved.
The propagation step is where selectivity often comes into play. The relative rates of different propagation pathways determine the major and minor products formed.
3. Termination: Bringing the Chain to a Halt
Termination steps involve the combination of two radical species, resulting in the formation of a non-radical molecule and effectively stopping the chain reaction. These reactions are usually bimolecular, meaning they involve two radicals colliding. Examples include:
- Radical coupling: Two identical radicals combine to form a single molecule.
- Disproportionation: Two radicals react, transferring a hydrogen atom from one to the other, producing an alkane and an alkene.
The Propagation Step: A Detailed Analysis
The propagation step is the heart of a radical reaction, driving the overall transformation. It’s crucial to understand that this step isn't a single event, but a sequence of reactions, each involving the creation and consumption of a radical intermediate. Let's explore some common examples:
Example 1: Halogenation of Alkanes
Consider the free radical chlorination of methane:
Initiation: Cl₂ → 2 Cl• (UV light)
Propagation:
- Cl• + CH₄ → HCl + •CH₃ (A chlorine radical abstracts a hydrogen atom from methane, forming hydrogen chloride and a methyl radical.)
- •CH₃ + Cl₂ → CH₃Cl + Cl• (The methyl radical reacts with chlorine molecule, producing chloromethane and regenerating a chlorine radical.)
This is a classic example of a two-step propagation sequence. Notice how the chlorine radical is both consumed and regenerated, allowing the chain reaction to continue.
Example 2: Addition Polymerization
In addition polymerization, the propagation step involves the repeated addition of monomer units to a growing polymer chain. For instance, in the polymerization of ethylene:
Initiation: (Formation of a radical initiator, e.g., from a peroxide)
Propagation:
- R• + CH₂=CH₂ → R-CH₂-CH₂• (The radical initiator adds to the double bond of ethylene, creating a new radical.)
- R-CH₂-CH₂• + CH₂=CH₂ → R-CH₂-CH₂-CH₂-CH₂• (The new radical adds another ethylene molecule, extending the chain.)
This step repeats numerous times, building a long polymer chain.
Factors Influencing Propagation Step Selectivity
The selectivity of a radical reaction—its preference for forming one product over another—is strongly influenced by various factors:
-
Bond Strengths: Radicals preferentially abstract hydrogen atoms from weaker C-H bonds. Tertiary C-H bonds are weaker than secondary, which are weaker than primary. This explains the selectivity observed in halogenation reactions.
-
Steric Hindrance: Bulky substituents can hinder the approach of radicals, making the reaction slower at sterically hindered sites.
-
Polar Effects: Although less pronounced than in ionic reactions, polar effects can influence radical reactions. Electron-withdrawing groups can stabilize radicals, increasing their reactivity at that site.
-
Temperature: Higher temperatures generally lead to less selective reactions, as the activation energy barriers are overcome more easily, allowing less favorable pathways to compete.
Controlling Selectivity in Radical Reactions
Understanding the factors affecting selectivity is crucial for controlling the outcome of radical reactions. Several strategies can be employed to achieve desired selectivity:
-
Careful Choice of Initiator: Different initiators can lead to varying radical concentrations and reactivity, influencing selectivity.
-
Temperature Control: Lower temperatures often favor more selective reactions.
-
Solvent Effects: Solvents can influence the stability and reactivity of radicals, thereby affecting selectivity.
-
Catalyst Use: In certain cases, catalysts can be employed to enhance the selectivity of radical reactions.
Advanced Topics and Applications
The propagation step in radical reactions is a complex process with many nuances. Advanced concepts, such as:
- Radical clocks: Used to measure the lifetime of radical intermediates.
- Intramolecular radical reactions: Reactions where the radical center reacts with another part of the same molecule, leading to cyclic products.
- Radical rearrangements: Rearrangements of radicals to form more stable structures.
These advanced concepts greatly expand the applications of radical reactions, making them powerful tools in the synthesis of complex molecules. Applications include:
- Polymer synthesis: The vast majority of commercially important polymers are made via radical polymerization.
- Drug discovery: Radical reactions are employed in the synthesis of many pharmaceutical compounds.
- Materials science: Radical reactions are important in the synthesis of advanced materials, including conducting polymers and nano-materials.
Conclusion: Mastering the Propagation Step
The propagation step is the driving force behind radical chain reactions. A thorough understanding of its mechanism, the factors that influence its selectivity, and the strategies for controlling the outcome is essential for anyone working with these reactions. By meticulously considering the various parameters, chemists can harness the power of radical reactions to achieve desired synthetic outcomes, paving the way for innovations in various fields. Further exploration of advanced concepts and applications will continue to expand the utility and importance of radical reactions in chemical synthesis and beyond. The ongoing research into this area promises even more exciting discoveries and applications in the future. This deep dive into the propagation step allows for a more nuanced understanding and control over these powerful, yet often unpredictable, transformations. Remember that safety is paramount when dealing with radical reactions, and appropriate precautions should always be taken.
Latest Posts
Latest Posts
-
How Are The Elements Organized In The Periodic Table
Apr 06, 2025
-
Are Proteins Polymers Of Amino Acids
Apr 06, 2025
-
How To Find Boiling Point Of Solution
Apr 06, 2025
-
The Muscle Tissue That Shows No Striations Is
Apr 06, 2025
-
The Originals Classic Readings In Western Philosophy
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Consider This Step In A Radical Reaction: . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
