Density Of Water At Different Temperatures Chart
Muz Play
Apr 02, 2025 · 5 min read
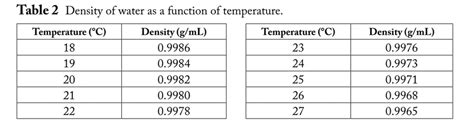
Table of Contents
Density of Water at Different Temperatures: A Comprehensive Chart and Guide
Water, the elixir of life, is a substance so ubiquitous that we often take its properties for granted. Yet, the seemingly simple molecule H₂O exhibits a fascinating array of behaviors, especially regarding its density at varying temperatures. Understanding this relationship is crucial across various scientific disciplines, from oceanography and meteorology to chemistry and engineering. This article delves into the intricacies of water density at different temperatures, providing a detailed chart, explanations of the underlying physics, and practical applications of this knowledge.
The Anomalous Behavior of Water Density
Unlike most substances, water doesn't exhibit a linear relationship between density and temperature. Its density increases as it cools, reaching a maximum at 4°C (39.2°F). Below this temperature, a remarkable anomaly occurs: water's density decreases as it continues to cool towards its freezing point (0°C or 32°F). This unusual behavior is attributed to the unique hydrogen bonding structure of water molecules.
Hydrogen Bonding: The Key to Water's Quirks
Water molecules are polar, meaning they possess a slightly positive end and a slightly negative end. This polarity allows them to form hydrogen bonds – relatively weak but significant attractions between the positive hydrogen atom of one molecule and the negative oxygen atom of another. These bonds create a complex, three-dimensional network.
At temperatures above 4°C, the kinetic energy of the molecules is high enough to overcome some of these hydrogen bonds, allowing the molecules to pack more closely together, thus increasing density. However, as the temperature drops below 4°C, the hydrogen bonds begin to dominate, forcing the molecules into a more open, crystalline structure – the hexagonal lattice of ice. This open structure is less dense than the liquid water at higher temperatures, leading to the decrease in density as water approaches its freezing point.
Implications of Water's Anomalous Density
This anomalous behavior has profound implications for aquatic life and the Earth's climate. The fact that ice is less dense than liquid water means that ice floats. This prevents lakes and oceans from freezing solid from the bottom up, allowing aquatic organisms to survive even during harsh winters. If ice were denser, it would sink, leading to a build-up of ice at the bottom, potentially killing off all aquatic life.
Furthermore, the density variations in water play a significant role in ocean currents and global heat distribution. The density differences drive thermohaline circulation, a global system of ocean currents that transports heat around the planet, influencing weather patterns and climate regulation.
Density of Water at Different Temperatures: A Detailed Chart
The following chart provides an approximation of water density at various temperatures. Note that precise values can vary slightly depending on the pressure and purity of the water. These values are typically measured at standard atmospheric pressure (1 atm).
| Temperature (°C) | Density (kg/m³) | Temperature (°F) | Density (lb/ft³) |
|---|---|---|---|
| 0 | 999.84 | 32 | 62.41 |
| 4 | 1000.00 | 39.2 | 62.43 |
| 10 | 999.70 | 50 | 62.40 |
| 15 | 999.10 | 59 | 62.36 |
| 20 | 998.21 | 68 | 62.30 |
| 25 | 997.05 | 77 | 62.21 |
| 30 | 995.65 | 86 | 62.10 |
| 40 | 992.2 | 104 | 61.90 |
| 50 | 988.04 | 122 | 61.61 |
| 60 | 983.2 | 140 | 61.31 |
| 70 | 977.8 | 158 | 60.97 |
| 80 | 971.8 | 176 | 60.60 |
| 90 | 965.3 | 194 | 60.20 |
| 100 | 958.4 | 212 | 59.76 |
Note: This chart represents pure water. The presence of dissolved salts or other substances will alter the density. For example, seawater has a higher density than freshwater due to the dissolved salts.
Factors Affecting Water Density
While temperature is the primary factor influencing water density, other factors play a secondary role:
Pressure:
Increasing pressure compresses water molecules, leading to a slight increase in density. This effect is more pronounced at higher pressures.
Salinity:
The presence of dissolved salts significantly increases water density. Ocean water, with its higher salt content, is denser than freshwater.
Isotopic Composition:
The relative abundance of different isotopes of hydrogen and oxygen in water molecules can slightly affect density. Heavy water (containing deuterium instead of protium) is denser than ordinary water.
Applications of Water Density Knowledge
Understanding the density of water at different temperatures is crucial in numerous fields:
Oceanography:
Oceanographers use density measurements to study ocean currents, mixing processes, and the formation of water masses. Density profiles provide insights into the stratification of the ocean and its role in marine ecosystems.
Meteorology:
Meteorologists consider water density variations to understand cloud formation, precipitation patterns, and atmospheric stability. Density differences drive convective currents that influence weather systems.
Chemical Engineering:
In chemical processes involving water, understanding its density variations is crucial for accurate calculations and efficient process design. This includes applications such as heat transfer, fluid dynamics, and material handling.
Environmental Science:
Water density is essential for assessing water quality, studying pollutant transport in aquatic systems, and managing water resources.
Aquatic Biology:
The density variations influence the distribution and behavior of aquatic organisms, impacting their survival and reproduction.
Conclusion
The seemingly simple relationship between water density and temperature is actually a complex interplay of molecular forces and physical phenomena. The anomalous behavior of water near its freezing point is a testament to the unique properties of this essential molecule. Understanding this relationship is crucial for a wide range of scientific disciplines and has practical applications in many areas of human activity. By continuing to study and refine our understanding of water density at different temperatures, we can improve our ability to predict and manage various environmental and industrial processes, ultimately contributing to a more sustainable and informed future. Further research into the precise density variations under different pressure and salinity conditions will further enhance our comprehensive understanding of this fundamental property of water.
Latest Posts
Latest Posts
-
What Is The Difference Between Intermolecular And Intramolecular Forces
Apr 03, 2025
-
Where Is The Energy Stored In Glucose
Apr 03, 2025
-
Light Amplification By The Stimulated Emission Of Radiation
Apr 03, 2025
-
Octet Rule Violation Vs Wrong Electron Total
Apr 03, 2025
-
What Are The Three Points Of Cell Theory
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Density Of Water At Different Temperatures Chart . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
