Different Conformations Of The Same Compound
Muz Play
Apr 02, 2025 · 7 min read
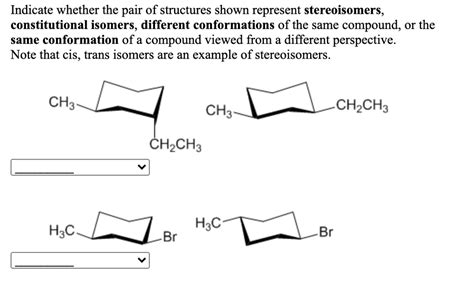
Table of Contents
Different Conformations of the Same Compound: A Deep Dive into Molecular Shapes
Understanding the different conformations of a single compound is crucial in chemistry, particularly in organic chemistry and biochemistry. While a molecule's connectivity (its constitution) remains fixed, the spatial arrangement of its atoms can vary significantly, leading to a range of conformations. This article will explore the intricacies of molecular conformations, focusing on the factors that influence their formation, the methods used to represent them, and their impact on a molecule's properties and reactivity.
What are Molecular Conformations?
Molecular conformations are different spatial arrangements of atoms in a molecule that can be interconverted by rotation about single bonds. Unlike isomers, which have different connectivities, conformers share the same molecular formula and connectivity but differ only in their three-dimensional arrangement. This difference in arrangement arises due to the free rotation around single bonds (sigma bonds). However, this rotation isn't entirely free; steric hindrance and other factors can create energy barriers, leading to certain conformations being more stable than others.
The Energy Landscape of Conformations
Conformations exist on a potential energy surface. Some conformations represent energy minima (more stable), while others represent energy maxima (less stable). The energy difference between conformations is crucial; a small energy difference allows for easy interconversion, while a large energy difference results in conformers that are effectively distinct at room temperature. This difference is often described in terms of the energy barrier separating them. This energy barrier is typically overcome through thermal energy, allowing for the interconversion of conformers.
Factors Influencing Conformation
Several factors play a vital role in determining the preferred conformations of a molecule:
1. Steric Hindrance:
Steric hindrance is a major factor influencing conformation. This refers to the repulsion between atoms or groups of atoms that are close in space. Bulky groups prefer to be as far apart as possible to minimize steric strain. For example, in butane, the anti conformation (methyl groups 180° apart) is more stable than the gauche conformation (methyl groups 60° apart) due to reduced steric repulsion between the methyl groups.
2. Electrostatic Interactions:
Electrostatic interactions between atoms or groups within a molecule also affect conformations. Polar groups tend to adopt arrangements that minimize dipole-dipole repulsions and maximize dipole-dipole attractions. For instance, in 1,2-dichloroethane, the anti conformation is favored because it places the polar C-Cl bonds as far apart as possible, minimizing dipole-dipole repulsion.
3. Hydrogen Bonding:
Intramolecular hydrogen bonding can significantly influence the conformation of a molecule. The formation of a hydrogen bond stabilizes the conformation where the hydrogen bond can form. This is especially important in biological molecules, where hydrogen bonding plays a critical role in maintaining protein structure and function.
4. Ring Strain:
In cyclic molecules, the ring size and the bond angles significantly impact the conformation. Smaller rings (e.g., three- and four-membered rings) experience significant ring strain due to deviation from ideal bond angles. Larger rings have more conformational flexibility. Cyclohexane, for example, adopts a chair conformation to minimize ring strain and achieve a stable structure.
Representing Molecular Conformations
Several methods are employed to represent and visualize molecular conformations:
1. Newman Projections:
Newman projections are a powerful tool for visualizing conformations. They are created by viewing the molecule down a specific C-C bond. The front carbon is represented by a dot, and the back carbon is represented by a circle. The substituents are then drawn radiating from these points. Newman projections effectively illustrate the torsional angle between substituents.
2. Sawhorse Projections:
Sawhorse projections offer an alternative representation of conformations. In this representation, the C-C bond is depicted as a slightly inclined line, with substituents drawn on either side. Similar to Newman projections, sawhorse projections help visualize the spatial relationship between substituents.
3. Fischer Projections:
Fischer projections are typically used for representing molecules with multiple chiral centers. They are two-dimensional representations where the molecule is shown as a straight line, with the vertical lines representing bonds projecting away from the viewer, and horizontal lines representing bonds projecting towards the viewer. While not as directly illustrative of three-dimensional structure as Newman or Sawhorse projections, they are useful for analyzing stereochemistry.
4. Molecular Models:
Three-dimensional molecular models (ball-and-stick or space-filling) provide a tangible representation of conformations. They allow for a more intuitive understanding of steric interactions and spatial relationships between atoms and groups within a molecule. These models are particularly valuable for visualizing complex molecules.
Conformations and Molecular Properties
The conformation of a molecule significantly influences its various properties:
1. Reactivity:
The reactivity of a molecule is often dictated by its conformation. Specific conformations may be required for a reaction to proceed. For example, certain enzyme-substrate interactions rely on specific conformations of both the enzyme and substrate. This conformational selectivity is critical in biochemistry.
2. Physical Properties:
Conformation impacts a molecule's physical properties, such as melting point, boiling point, and density. Different conformations can lead to different intermolecular interactions, affecting these physical parameters. For instance, the more compact conformations generally have higher melting and boiling points due to increased van der Waals interactions.
3. Spectroscopic Properties:
Conformations can influence a molecule's spectroscopic properties, such as NMR and IR spectra. Different conformations can lead to distinct chemical shifts in NMR or altered vibrational frequencies in IR, providing insights into the conformational distribution. Conformational analysis using spectroscopy is a powerful tool for studying molecular structure.
4. Biological Activity:
In biological systems, the conformation of a molecule is paramount to its biological activity. Proteins, for example, fold into specific three-dimensional structures that dictate their function. The precise conformation is essential for interactions with other molecules, leading to specific biological effects. Changes in conformation can lead to loss of function or altered activity.
Advanced Conformation Analysis Techniques
Beyond basic representations, advanced techniques are used to analyze and predict molecular conformations:
1. Computational Chemistry:
Computational methods such as molecular mechanics and quantum mechanics are employed to predict stable conformations and their relative energies. These methods allow researchers to explore the conformational landscape of a molecule, even for complex systems, and predict which conformations are most likely to exist under specific conditions.
2. X-ray Crystallography and NMR Spectroscopy:
Experimental techniques like X-ray crystallography and NMR spectroscopy provide valuable information about the conformation of a molecule in the solid state or in solution, respectively. X-ray crystallography can determine the exact atomic positions in a crystal, providing a definitive picture of the molecular conformation. NMR spectroscopy can reveal information about the relative orientations of atoms and groups within a molecule, helping to determine the conformational distribution.
Examples of Conformational Isomerism
Let's examine specific examples to illustrate the concept of different conformations:
1. Butane:
Butane, with its freely rotating C-C bond, exhibits various conformations, including the anti, gauche, and fully eclipsed forms. The anti conformation is the most stable due to minimal steric hindrance, whereas the fully eclipsed is the least stable due to maximum steric hindrance.
2. Cyclohexane:
Cyclohexane, a six-membered ring, famously exists in two major chair conformations, which are interconvertible through a process called ring-flipping. These chair conformations differ in the axial and equatorial positions of substituents. Substituents prefer to occupy equatorial positions to minimize 1,3-diaxial interactions.
3. Proteins:
Proteins are complex molecules with a vast array of conformations. The overall three-dimensional structure of a protein is essential for its function, and even subtle changes in conformation can alter its activity. The process of protein folding involves the exploration of a wide range of conformations until the most stable and functional structure is reached.
Conclusion
The study of molecular conformations is a fundamental aspect of chemistry and related fields. Understanding how different conformations arise, their relative stabilities, and their impact on molecular properties is crucial for interpreting chemical reactivity, predicting physical properties, and explaining biological function. Advanced experimental and computational techniques continue to improve our ability to study and understand this fascinating aspect of molecular structure. As we delve deeper into the intricacies of molecular behavior, the importance of considering conformational analysis in various scientific disciplines will only continue to grow. The detailed understanding of conformations is indispensable in drug design, materials science, and various other fields, highlighting the significance of continued exploration in this area. Further research into novel methods and techniques will undoubtedly provide even more detailed insights into the world of molecular conformations.
Latest Posts
Latest Posts
-
What Is Shared In A Covalent Bond
Apr 03, 2025
-
Chemical Reactions And Equations Report Sheet
Apr 03, 2025
-
Cis 1 3 Dimethylcyclohexane Chair Conformation
Apr 03, 2025
-
What Does The Bacterial Chromosome Do
Apr 03, 2025
-
What Is The Difference Between Dehydration And Hydrolysis
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Different Conformations Of The Same Compound . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
