Gaining Or Losing Electrons Is Called
Muz Play
Apr 05, 2025 · 6 min read
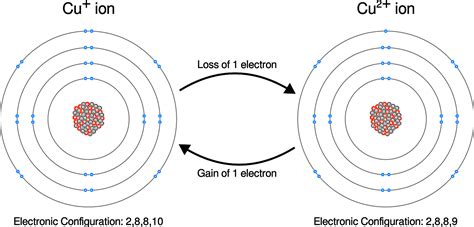
Table of Contents
Gaining or Losing Electrons is Called Ionization: A Deep Dive into Atomic Interactions
Gaining or losing electrons is a fundamental process in chemistry called ionization. It's a cornerstone concept that explains a vast array of phenomena, from the formation of ionic compounds to the behavior of electricity. Understanding ionization is crucial for grasping the intricacies of atomic structure, chemical bonding, and numerous other aspects of the physical world. This comprehensive guide delves into the process of ionization, exploring its mechanisms, consequences, and significance across various scientific disciplines.
What is Ionization?
Ionization is the process by which an atom or molecule acquires a net electrical charge by gaining or losing electrons. When an atom loses one or more electrons, it becomes positively charged, forming a cation. Conversely, when an atom gains one or more electrons, it becomes negatively charged, forming an anion. This charge imbalance profoundly impacts the atom's or molecule's properties and its interactions with other atoms and molecules.
The Role of Electrons in Ionization
Electrons, negatively charged subatomic particles, orbit the nucleus of an atom. The number of electrons in a neutral atom equals the number of protons in its nucleus, resulting in a net charge of zero. Ionization disrupts this balance. The outermost electrons, known as valence electrons, are the most loosely bound and are typically the first to be involved in ionization processes.
Factors Affecting Ionization
Several factors influence the ease with which an atom can be ionized:
-
Atomic Radius: Atoms with larger atomic radii have their valence electrons further from the nucleus, experiencing weaker electrostatic attraction. These atoms are generally easier to ionize.
-
Effective Nuclear Charge: The effective nuclear charge represents the net positive charge experienced by valence electrons. A higher effective nuclear charge leads to stronger attraction to the nucleus, making ionization more difficult.
-
Electron Shielding: Inner electrons shield the outer electrons from the full positive charge of the nucleus. Increased shielding reduces the effective nuclear charge and makes ionization easier.
-
Ionization Energy: Ionization energy is the energy required to remove an electron from a neutral atom in its gaseous state. Higher ionization energy indicates a greater difficulty in removing an electron. It's important to note that successive ionization energies increase progressively as more electrons are removed, as each subsequent electron is held more tightly by the increasingly positive ion.
Types of Ionization
Ionization can occur through various mechanisms:
1. Collisional Ionization:
This process involves the collision of an atom or molecule with a high-energy particle, such as an electron or another atom. The kinetic energy from the collision is transferred to an electron in the target atom, providing enough energy to overcome the electrostatic attraction and escape the atom. This is a common mechanism in plasma, gases subjected to high temperatures, and in particle accelerators.
2. Photoionization:
In photoionization, an atom absorbs a photon (a particle of light) with sufficient energy to eject an electron. The energy of the photon must exceed the atom's ionization energy. This process is crucial in astrophysics, where the ionization of atoms in stellar atmospheres is driven by intense ultraviolet radiation. It also plays a vital role in atmospheric chemistry and the formation of the ionosphere.
3. Chemical Ionization:
Chemical ionization involves the transfer of an electron from one atom or molecule to another during a chemical reaction. This type of ionization is crucial in many chemical processes, particularly in the formation of ionic compounds. For example, the reaction between sodium (Na) and chlorine (Cl) involves the transfer of an electron from sodium to chlorine, forming Na+ and Cl- ions, which then attract each other to form sodium chloride (NaCl).
4. Field Ionization:
Field ionization occurs when a strong electric field removes an electron from an atom. The intense electric field distorts the atom's electron cloud, allowing an electron to tunnel through the potential barrier and escape. This is commonly observed in techniques like field emission microscopy and mass spectrometry.
Consequences of Ionization
The consequences of ionization are far-reaching and have significant implications across various scientific domains:
1. Formation of Ions and Ionic Compounds:
Ionization is the primary mechanism behind the formation of ions, which are atoms or molecules with a net electrical charge. These ions can then interact through electrostatic forces, forming ionic compounds. These compounds have unique properties different from their constituent elements, like high melting points and solubility in water.
2. Electrical Conductivity:
Ions are responsible for the electrical conductivity of many substances. In ionic solutions and molten salts, the mobility of ions enables the flow of electric current. In gases, ionization produces charged particles that can carry electric current, leading to phenomena like electrical discharges and lightning.
3. Chemical Reactivity:
Ions have different chemical reactivities compared to their neutral counterparts. The presence of a net charge significantly alters their ability to participate in chemical reactions, influencing reaction rates and products. This is especially important in biological systems where ionic interactions are essential for enzyme activity and other biochemical processes.
4. Spectroscopic Analysis:
The energy levels of ions are different from those of neutral atoms, leading to distinct spectral lines. This allows scientists to use spectroscopy to identify and quantify ions in various samples, providing insights into the composition and properties of materials. Techniques like atomic absorption spectroscopy and inductively coupled plasma mass spectrometry rely heavily on ionization principles.
Ionization in Different Fields
Ionization plays a crucial role in a wide range of scientific and technological applications:
1. Medicine:
Ionizing radiation, such as X-rays and gamma rays, is used extensively in medical imaging and radiation therapy. These high-energy photons ionize atoms in biological tissues, leading to the generation of signals used for imaging or the destruction of cancerous cells.
2. Astrophysics:
Ionization processes are fundamental in astrophysics. The ionization of atoms in stars and interstellar gas clouds influences their temperature, density, and spectral properties. The study of ionized gas reveals information about the composition, evolution, and dynamics of celestial objects.
3. Environmental Science:
Ionization plays a role in atmospheric chemistry, influencing the formation of ozone and other atmospheric constituents. The ionization of air molecules by cosmic rays and solar radiation forms the ionosphere, which affects radio wave propagation.
4. Materials Science:
Ionization is used in various materials processing techniques, such as ion implantation, to modify the properties of materials. Ion beams can be used to dope semiconductors, creating devices with tailored electrical characteristics.
5. Analytical Chemistry:
Various analytical techniques rely on ionization for the detection and quantification of analytes. Techniques such as mass spectrometry and flame ionization detection use ionization to separate and measure different components in a sample.
Conclusion: The Ubiquity of Ionization
In conclusion, gaining or losing electrons – ionization – is a fundamental process with profound implications across a vast array of scientific disciplines. From the formation of ionic compounds to the workings of sophisticated analytical techniques and the vastness of astrophysical phenomena, ionization is a ubiquitous process that shapes our understanding of the physical world. A thorough understanding of ionization is essential for advancements in various fields, from medicine and materials science to environmental monitoring and astrophysics. The ongoing research into ionization continues to unlock deeper insights into the intricate interplay of atoms and molecules, furthering our knowledge of the universe and its fundamental building blocks.
Latest Posts
Latest Posts
-
Through Which Microscope Were Cells First Observed
Apr 05, 2025
-
Where Does Gas Exchange Take Place In Plants
Apr 05, 2025
-
How To Calculate Surplus And Shortage
Apr 05, 2025
-
How Many Valence Electrons Does Sulfer Have
Apr 05, 2025
-
Power Of A Quotient Property Examples
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Gaining Or Losing Electrons Is Called . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
