Graph Of Atomic Number Vs Atomic Radius
Muz Play
Apr 02, 2025 · 5 min read
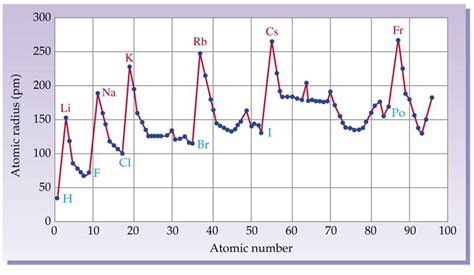
Table of Contents
Graph of Atomic Number vs. Atomic Radius: A Comprehensive Exploration
The relationship between an atom's atomic number and its atomic radius provides crucial insights into the periodic trends of elements and their chemical behavior. This article delves deep into this relationship, exploring the underlying principles, analyzing the graphical representation, and examining the exceptions and nuances that enrich our understanding of atomic structure.
Understanding the Fundamentals: Atomic Number and Atomic Radius
Before analyzing the graph, let's solidify our understanding of the key terms:
Atomic Number (Z):
The atomic number represents the number of protons in an atom's nucleus. It's a fundamental property that uniquely identifies each element. Since atoms are electrically neutral, the atomic number also equals the number of electrons in a neutral atom. This is crucial because electron configuration directly influences the atom's size.
Atomic Radius:
Defining atomic radius isn't straightforward, as atoms don't have sharply defined boundaries. Several methods exist for measuring atomic radius, but they generally represent the average distance between the nucleus and the outermost electron. Commonly used methods include covalent radius (half the distance between the nuclei of two identical atoms bonded covalently), metallic radius (half the distance between the nuclei of adjacent atoms in a metallic crystal), and van der Waals radius (half the distance between the nuclei of two identical atoms that are not bonded but are close enough to interact weakly). While different methods yield slightly different values, the overall trends remain consistent.
The Graph: Visualizing the Trend
A graph plotting atomic number against atomic radius reveals a clear periodic trend. The graph generally shows an oscillatory decrease in atomic radius across a period (row) and an overall increase down a group (column) of the periodic table.
Periodicity Across a Period:
As we move from left to right across a period, the atomic number increases, meaning more protons are added to the nucleus. Simultaneously, electrons are added to the same principal energy level (shell). The increased positive charge of the nucleus attracts the electrons more strongly, pulling them closer to the nucleus. This results in a decrease in atomic radius across the period.
However, this trend is not perfectly linear. The slight increases observed in some cases are due to electron-electron repulsions within the same subshell. As electrons are added to the same subshell, the repulsion between them partially offsets the attractive force from the nucleus, resulting in a slightly larger atomic radius than might be expected based solely on nuclear charge.
Periodicity Down a Group:
Moving down a group, both the atomic number and the number of electron shells increase. While the nuclear charge increases, the added electrons occupy new, higher energy levels farther from the nucleus. The effect of adding an electron shell far outweighs the increased nuclear attraction, leading to a significant increase in atomic radius as you descend a group. The increased distance of the outermost electrons from the nucleus significantly diminishes the attractive force, resulting in a larger atom.
Exceptions and Nuances: A Deeper Dive
While the general trends are clear, certain exceptions exist, adding complexity and providing valuable insights into atomic interactions.
Transition Metals:
Transition metals show a relatively smaller decrease in atomic radius across a period compared to the main group elements. This is attributed to the gradual filling of the d-orbitals, which are shielded from the full effect of the increasing nuclear charge by the inner-core electrons. The shielding effect mitigates the decrease in atomic radius.
Lanthanides and Actinides:
The lanthanides and actinides exhibit a unique trend of decreasing atomic radius across the series (known as the lanthanide and actinide contraction). This is due to the poor shielding effect of the f-orbitals. The addition of electrons to the f-subshell doesn't significantly increase the shielding effect, leading to a stronger effective nuclear charge that pulls the electrons closer to the nucleus, resulting in a smaller atomic radius than expected.
Anomalous Behavior of Certain Elements:
Certain elements demonstrate deviations from the general trends due to specific electron configurations or inter-electronic repulsions. For example, some elements might have slightly larger atomic radii than their neighbors due to electron pairing within a subshell, resulting in increased electron-electron repulsion.
The Importance of the Atomic Radius Trend
Understanding the relationship between atomic number and atomic radius is fundamental to comprehending various chemical and physical properties:
- Ionization energy: Smaller atoms with a greater effective nuclear charge require more energy to remove an electron, leading to higher ionization energies.
- Electronegativity: Smaller atoms with a high effective nuclear charge strongly attract electrons, resulting in higher electronegativity.
- Reactivity: Atomic radius influences the ease with which an atom can gain or lose electrons, directly impacting its reactivity.
- Metallic character: Larger atomic radii generally correlate with increased metallic character.
- Density: Atomic radius plays a role in determining the density of an element, as it influences how closely atoms pack together in the solid state.
- Crystal structure: The atomic radius determines the coordination number and crystal structure of elements and compounds.
Applications and Further Exploration
The knowledge gained from studying the atomic number vs. atomic radius graph has significant applications in various scientific fields:
- Materials science: Understanding atomic radii is essential for designing and predicting the properties of new materials.
- Chemistry: Predicting chemical reactions and bonding behavior relies heavily on the understanding of atomic size and electron configuration.
- Physics: Atomic radius is crucial in studies involving atomic interactions and properties.
Further explorations could involve:
- Detailed analysis of specific element groups and their deviations from general trends.
- Comparative studies of atomic radii determined using different methods.
- Incorporating other atomic properties, such as effective nuclear charge and shielding effect, into the analysis.
- Exploring the application of computational methods for predicting atomic radii.
Conclusion
The graph of atomic number vs. atomic radius showcases a fascinating periodic trend that underpins much of our understanding of chemical behavior and material properties. Although the general trends are predictable, exceptions and nuances add layers of complexity, highlighting the intricate interplay between nuclear charge, electron configuration, and inter-electronic repulsions. A deep understanding of this relationship is crucial for various scientific disciplines, and continued research promises to further refine our knowledge of this fundamental aspect of atomic structure. By analyzing the graph and understanding the underlying principles, we gain a powerful tool for predicting and interpreting the properties of elements and their compounds.
Latest Posts
Latest Posts
-
What Is The Formula Of A Hydronium Ion
Apr 03, 2025
-
What Is Closed And Open System
Apr 03, 2025
-
What Does It Mean When We Say Water Is Adhesive
Apr 03, 2025
-
What Makes Something A Good Leaving Group
Apr 03, 2025
-
Cell Membrane And Transport Graphic Answer Key
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Graph Of Atomic Number Vs Atomic Radius . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
