What Makes Something A Good Leaving Group
Muz Play
Apr 03, 2025 · 5 min read
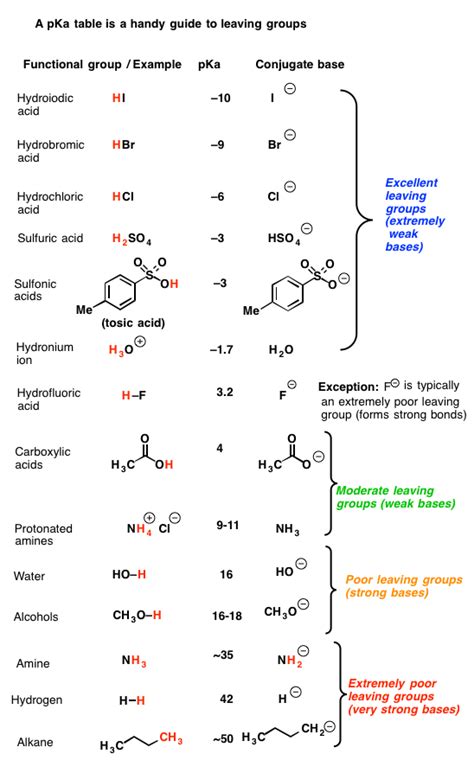
Table of Contents
What Makes a Good Leaving Group? A Deep Dive into Organic Chemistry
Leaving groups are fundamental to many organic reactions, particularly substitution and elimination reactions. Understanding what constitutes a good leaving group is crucial for predicting reaction outcomes and designing synthetic strategies. This in-depth exploration delves into the key factors that determine a leaving group's effectiveness, providing a comprehensive understanding for both students and seasoned chemists.
The Essence of a Leaving Group
A leaving group (LG) is an atom or group of atoms that departs from a molecule, taking with it a pair of electrons. This departure creates a reactive intermediate, often a carbocation or carbanion, which then reacts further. The stability of the leaving group significantly impacts the rate and feasibility of the reaction. A good leaving group readily accepts the electron pair, stabilizing the negative charge it acquires after leaving. Conversely, a poor leaving group resists accepting the electrons, hindering the reaction's progression.
Key Factors Determining Leaving Group Ability
Several key factors influence how effectively a group can act as a leaving group:
-
Stability of the leaving group: The most crucial factor. A stable leaving group readily accepts the electron pair and disperses the negative charge through resonance or inductive effects. This stability translates directly to a faster reaction rate.
-
Basicity: There's an inverse relationship between leaving group ability and basicity. Weak bases make excellent leaving groups. Strong bases, on the other hand, strongly hold onto their electrons and are reluctant to leave. This is because strong bases readily accept protons, making them poor at accepting electron pairs and stabilizing the negative charge.
-
Size and Polarizability: Larger atoms with more diffuse electron clouds are generally better leaving groups. These larger atoms can better accommodate the negative charge, making them more stable after departure. Polarizability enhances this effect.
-
Solvent Effects: The solvent plays a crucial role. Polar protic solvents are better at stabilizing the negatively charged leaving group, facilitating its departure. Conversely, aprotic solvents might hinder the process.
Examples of Good and Poor Leaving Groups
Let's examine some specific examples to illustrate the principles discussed above:
Excellent Leaving Groups
-
Iodide (I⁻): Iodide is a large, weakly basic anion. Its large size allows for effective dispersal of the negative charge, making it a highly stable leaving group.
-
Bromide (Br⁻): Similar to iodide, bromide is a relatively large and weakly basic anion. Its stability makes it a good leaving group, although slightly less effective than iodide.
-
Chloride (Cl⁻): Smaller than bromide and iodide, chloride is still a reasonably good leaving group. However, its smaller size results in less effective charge dispersal compared to the larger halides.
-
Tosylate (TsO⁻): Tosylates are excellent leaving groups due to resonance stabilization. The negative charge can be delocalized across the sulfonate group, significantly enhancing its stability. This resonance stabilization counteracts the basicity, effectively improving its leaving group ability.
-
Triflate (OTf⁻): Triflates, like tosylates, possess significant resonance stabilization, making them exceptionally good leaving groups. The electron-withdrawing fluorine atoms further enhance their stability.
Poor Leaving Groups
-
Hydroxide (OH⁻): Hydroxide is a strong base and poorly stabilizes the negative charge. It is a very poor leaving group in most reactions.
-
Alkoxide (RO⁻): Alkoxides are strong bases, making them unfavorable leaving groups. Their basic nature prevents them from readily accepting the electron pair and stabilizing the resultant negative charge.
-
Amide (NH₂⁻): Amides are extremely strong bases and exceptionally poor leaving groups. They strongly resist accepting the electron pair, significantly impeding reaction progress.
-
Hydride (H⁻): Hydride is a very strong base and a remarkably poor leaving group. Its high basicity and the small size of the hydrogen atom hinder the dispersal of the negative charge, making it exceptionally unstable as a leaving group.
The Impact of Leaving Group Ability on Reaction Rates
The quality of the leaving group directly influences the reaction rate. Reactions involving excellent leaving groups proceed much faster than those involving poor leaving groups. Consider a simple SN1 reaction: the rate-determining step is the formation of the carbocation intermediate, which is directly influenced by the leaving group's ability to depart. A good leaving group facilitates carbocation formation, speeding up the entire process.
Leaving Group Ability in Different Reaction Mechanisms
The importance of the leaving group varies depending on the reaction mechanism:
SN1 Reactions (Unimolecular Nucleophilic Substitution)
In SN1 reactions, the leaving group departs first, forming a carbocation intermediate. A good leaving group is crucial because it determines the rate-limiting step. Better leaving groups lead to faster reaction rates.
SN2 Reactions (Bimolecular Nucleophilic Substitution)
In SN2 reactions, the nucleophile attacks the substrate simultaneously as the leaving group departs. While a good leaving group is still beneficial, its effect on the rate is less pronounced compared to SN1 reactions. The strength of the nucleophile often plays a more significant role.
Elimination Reactions (E1 and E2)
Leaving group ability is paramount in elimination reactions. In E1 reactions, similar to SN1, the leaving group departs first, leading to a carbocation intermediate. In E2 reactions, the base abstracts a proton while the leaving group departs simultaneously. In both cases, a good leaving group significantly enhances the reaction rate.
Enhancing Leaving Group Ability: Protecting Groups and Activation
Sometimes, naturally occurring functional groups are poor leaving groups. In such cases, protecting groups and activation strategies are employed to transform them into better leaving groups. For instance, converting an alcohol (-OH) into a tosylate (-OTs) significantly improves its leaving group ability. This conversion effectively transforms a poor leaving group into an excellent one, enabling the desired reaction to proceed efficiently.
Conclusion: A Holistic View of Leaving Groups
The effectiveness of a leaving group is a multifaceted concept. It's not merely a matter of whether a group leaves or not, but how readily it departs. Factors like stability (influenced by basicity, size, and resonance), solvent effects, and the reaction mechanism all play crucial roles. A deep understanding of these factors is crucial for predicting reaction outcomes, designing effective synthetic pathways, and enhancing the efficiency of organic reactions. By considering these factors, chemists can strategically manipulate leaving groups to achieve their synthetic goals, leading to faster and more efficient chemical transformations. Continuous research further expands our knowledge of leaving group behavior, enriching our understanding of organic reaction mechanisms and synthetic strategies.
Latest Posts
Latest Posts
-
Two Bones That Form The Nasal Septum
Apr 04, 2025
-
How To Determine The Age Of A Fossil
Apr 04, 2025
-
The Nucleus Of An Atom Consists Of
Apr 04, 2025
-
What Happens To The Electrons In Metallic Bonding
Apr 04, 2025
-
How To Know Which Orbital A Lone Pair Is In
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Makes Something A Good Leaving Group . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
