What Is The Formula Of A Hydronium Ion
Muz Play
Apr 03, 2025 · 5 min read
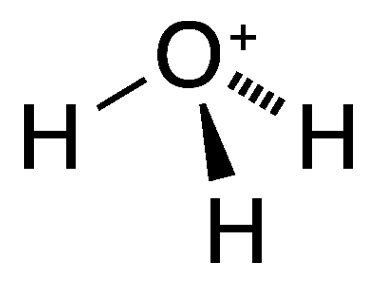
Table of Contents
What is the Formula of a Hydronium Ion? A Deep Dive into Acid-Base Chemistry
The hydronium ion, a fundamental concept in chemistry, plays a crucial role in understanding acid-base reactions and the behavior of acids in aqueous solutions. This article will explore the formula, structure, properties, and significance of the hydronium ion, delving into its role in various chemical processes and its impact on pH measurements. We will also examine common misconceptions and provide a clear, comprehensive understanding of this essential chemical species.
Understanding the Basics: Acids and Protons
Before diving into the hydronium ion itself, let's establish a foundational understanding of acids and their behavior in water. Acids are substances that donate protons (H⁺) – a hydrogen atom that has lost its electron, leaving only a positively charged proton. This proton donation is the defining characteristic of an acid. However, free protons, or bare H⁺ ions, do not exist independently in aqueous solutions. Instead, they are always associated with other molecules, most commonly water molecules.
The Formation of the Hydronium Ion: H₃O⁺
This is where the hydronium ion comes in. When an acid dissolves in water, it donates a proton to a water molecule (H₂O). This proton is accepted by the oxygen atom in the water molecule, forming a hydronium ion (H₃O⁺). The chemical equation representing this process is:
HA + H₂O ⇌ H₃O⁺ + A⁻
Where:
- HA represents the acid (e.g., HCl, acetic acid).
- H₂O represents the water molecule.
- H₃O⁺ represents the hydronium ion.
- A⁻ represents the conjugate base of the acid.
The reaction shown above is an equilibrium reaction. This means that the reaction doesn't proceed completely in one direction; instead, there is a dynamic balance between the reactants (HA and H₂O) and the products (H₃O⁺ and A⁻). The position of the equilibrium depends on the strength of the acid. Strong acids have a larger equilibrium constant (Ka) indicating a higher concentration of H₃O⁺ ions.
The Structure of the Hydronium Ion
The hydronium ion has a trigonal pyramidal structure. The central oxygen atom is bonded to three hydrogen atoms, with one hydrogen atom carrying a positive charge due to the extra proton. The oxygen atom is sp³ hybridized, and the bond angle is slightly less than 109.5° due to the lone pair of electrons on the oxygen atom. This lone pair exerts a repulsive force on the bonding pairs, slightly compressing the H-O-H angles.
Beyond H₃O⁺: The Reality of Hydration
While H₃O⁺ is the simplest representation of the hydrated proton, it's important to understand that the proton is further solvated by additional water molecules. In reality, the proton is highly mobile and rapidly exchanged between water molecules. Instead of a single H₃O⁺ ion, it's more accurate to consider a complex network of water molecules with a shared proton, sometimes represented as H₃O⁺(H₂O)ₙ, where 'n' indicates the number of water molecules surrounding the central hydronium ion. This number can vary depending on the concentration and conditions.
The Significance of the Hydronium Ion: pH and Acidity
The concentration of hydronium ions (H₃O⁺) in a solution is directly related to its acidity, a fundamental property that determines the solution’s pH. The pH scale, ranging from 0 to 14, indicates the concentration of H₃O⁺ ions:
- pH < 7: Acidic solution (higher concentration of H₃O⁺).
- pH = 7: Neutral solution (equal concentrations of H₃O⁺ and OH⁻).
- pH > 7: Basic solution (lower concentration of H₃O⁺).
The pH is calculated using the following formula:
pH = -log₁₀[H₃O⁺]
where [H₃O⁺] is the concentration of hydronium ions in moles per liter (mol/L). The lower the pH, the higher the concentration of hydronium ions and the stronger the acid.
Common Misconceptions about the Hydronium Ion
Several misconceptions surrounding the hydronium ion need clarification:
-
H⁺ vs. H₃O⁺: It's crucial to understand that free H⁺ ions do not exist in aqueous solutions. The representation H⁺ is often used as a shorthand for the hydronium ion, H₃O⁺, in many chemical equations for simplicity. However, understanding the true nature of the hydrated proton is essential for a complete understanding.
-
The size of the hydrated proton: The hydrated proton isn't a simple, single entity. The proton is rapidly exchanged between water molecules, creating a dynamic network. This makes it challenging to define a precise “size” for the hydrated proton.
The Hydronium Ion in Various Chemical Processes
The hydronium ion plays a vital role in numerous chemical processes, including:
-
Acid-base titrations: The concentration of H₃O⁺ is closely monitored during titrations to determine the equivalence point.
-
Electrochemistry: H₃O⁺ participates in redox reactions at electrodes, influencing the potential of electrochemical cells.
-
Catalysis: Many acid-catalyzed reactions involve H₃O⁺ as a catalyst. It acts as a proton donor, activating reactants and facilitating the reaction pathway.
-
Hydrolysis reactions: Hydronium ions are involved in the hydrolysis of various compounds, breaking down molecules by reacting them with water.
Advanced Concepts and Further Exploration
For those interested in a more in-depth understanding, further exploration could include:
-
Different degrees of hydration: Research on the exact structure and dynamics of the hydrated proton, exploring the different hydration shells and the number of water molecules associated with the proton.
-
Computational chemistry studies: Investigating the structures and energetics of hydrated protons using computational methods.
-
Isotope effects: Studying the effects of isotopic substitution (e.g., using deuterium instead of hydrogen) on the properties and reactions of the hydronium ion.
-
Hydronium ion in non-aqueous solvents: Understanding how the behaviour of acids and the formation of solvated protons change in solvents other than water.
Conclusion: A Cornerstone of Chemistry
The hydronium ion, despite its seemingly simple formula (H₃O⁺), is a cornerstone of acid-base chemistry and a crucial concept for understanding many chemical processes. Its correct representation and understanding, including the reality of hydration and its influence on pH, are vital for a thorough grasp of solution chemistry. This article aimed to provide a comprehensive overview, clarifying common misconceptions and highlighting the importance of the hydronium ion in various chemical contexts. By exploring this fundamental concept, we gain a deeper appreciation of the complexity and elegance of chemical reactions in aqueous solutions.
Latest Posts
Latest Posts
-
Cells Are Basic Unit Of Life
Apr 04, 2025
-
Two Bones That Form The Nasal Septum
Apr 04, 2025
-
How To Determine The Age Of A Fossil
Apr 04, 2025
-
The Nucleus Of An Atom Consists Of
Apr 04, 2025
-
What Happens To The Electrons In Metallic Bonding
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is The Formula Of A Hydronium Ion . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
