Horizontal Columns On The Periodic Table Are Called
Muz Play
Apr 02, 2025 · 7 min read
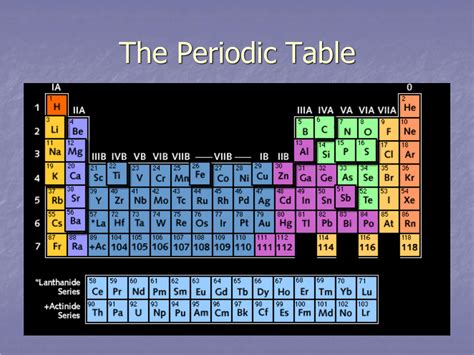
Table of Contents
Horizontal Columns on the Periodic Table are Called: Periods – A Deep Dive into Periodic Trends
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic number and recurring chemical properties. Understanding its structure is crucial to grasping the behavior of matter. While many focus on the vertical columns, or groups, the horizontal rows, known as periods, are equally important in understanding periodic trends and the properties of elements. This comprehensive guide delves deep into the significance of periods on the periodic table, exploring their relationship to electron configuration, atomic size, ionization energy, and electronegativity.
What are Periods in the Periodic Table?
The horizontal rows of the periodic table are called periods. Each period corresponds to a principal energy level (n) in an atom's electron shell. The number of the period indicates the highest principal energy level occupied by electrons in the ground state of the atoms in that period. For instance, Period 1 contains elements with electrons only in the n=1 energy level, Period 2 elements have electrons in the n=1 and n=2 energy levels, and so on.
This seemingly simple concept underpins a wealth of predictable trends in elemental properties. As we move across a period, the number of protons and electrons increases, leading to systematic changes in atomic radius, ionization energy, and electronegativity.
Number of Elements in Each Period
It's important to note that the number of elements in each period is not constant. The first period contains only two elements, hydrogen (H) and helium (He), because the first electron shell can hold a maximum of two electrons. The second and third periods each contain eight elements, reflecting the filling of the s and p orbitals in the second and third energy levels. Subsequent periods have more elements because of the inclusion of d and f orbitals, which can accommodate more electrons.
- Period 1: 2 elements (Hydrogen and Helium)
- Period 2: 8 elements (Lithium to Neon)
- Period 3: 8 elements (Sodium to Argon)
- Period 4: 18 elements (Potassium to Krypton)
- Period 5: 18 elements (Rubidium to Xenon)
- Period 6: 32 elements (Cesium to Radon)
- Period 7: 32 elements (Francium to Oganesson) (Incomplete)
Periodic Trends Across a Period
The systematic changes in elemental properties across a period are some of the most significant aspects of the periodic table's organization. These trends are directly linked to the increasing nuclear charge and the addition of electrons to the same principal energy level.
1. Atomic Radius: A Shrinking Trend
As we traverse a period from left to right, the atomic radius generally decreases. This is because the number of protons in the nucleus increases, resulting in a stronger positive charge pulling the electrons closer to the nucleus. While additional electrons are added to the same energy level, the increased nuclear charge outweighs the electron-electron repulsion, causing the overall atomic size to contract.
The noble gases at the end of each period, despite having a larger electron cloud due to electron-electron repulsion, have a smaller atomic radius compared to their preceding elements because the increased nuclear charge dominates the effect. Exceptions might occur in transition elements due to the filling of inner d-orbitals which have a less effective shielding effect.
2. Ionization Energy: An Increasing Trend
Ionization energy is the energy required to remove an electron from a gaseous atom. As we move across a period, ionization energy generally increases. This is a direct consequence of the increasing nuclear charge. The stronger attraction between the nucleus and the electrons makes it more difficult to remove an electron, hence requiring more energy.
Again, exceptions exist, particularly in the transition metals. The slight increase in shielding by inner d-electrons can sometimes lead to smaller increases in ionization energy than expected.
3. Electronegativity: A Growing Attraction
Electronegativity measures the ability of an atom to attract electrons in a chemical bond. Similar to ionization energy, electronegativity generally increases across a period from left to right. The increasing nuclear charge makes the atom more effective at attracting shared electrons in a covalent bond.
The noble gases are generally excluded from electronegativity trends as they rarely form chemical bonds.
4. Metallic Character: A Waning Influence
The metallic character of elements refers to their tendency to lose electrons and form positive ions. As we move across a period, metallic character generally decreases. This is because the increasing nuclear charge makes it more difficult for the atom to lose electrons, leading to a shift towards non-metallic behavior. Elements on the far left of the periodic table tend to be highly metallic, while those on the far right (excluding noble gases) are non-metallic.
5. Electron Affinity: A Complex Trend
Electron affinity is the energy change that occurs when an atom gains an electron. While generally exhibiting an increase across a period, the trend isn't as straightforward as ionization energy or electronegativity. The addition of an electron can sometimes lead to electron-electron repulsion, which offsets the attractive force of the nucleus, resulting in less negative or even positive electron affinities in certain cases.
The Significance of Periods and Predicting Properties
Understanding the trends associated with periods is crucial for predicting the properties of elements. Knowing the period an element belongs to provides significant information about its atomic radius, ionization energy, electronegativity, and metallic character. This information is invaluable in understanding chemical reactivity and bonding behavior.
For example, knowing that elements in Period 3 have electrons filling the third principal energy level allows us to predict that they will have a larger atomic radius than those in Period 2, but a smaller radius than elements in Period 4. Similarly, we can expect elements in Period 3 to have higher ionization energies than those in Period 2, reflecting the stronger nuclear attraction.
Periods and Chemical Reactivity
The position of an element within a period significantly influences its chemical reactivity. Elements on the far left (alkali metals) readily lose one electron to achieve a stable electron configuration, making them highly reactive. Elements on the far right (halogens) readily gain one electron to achieve a stable configuration, making them also highly reactive. The noble gases, however, are unreactive due to their stable electron configurations.
The transition metals within a period exhibit varying reactivity depending on factors such as the energy required to remove electrons and the stability of the resulting ions. The reactivity of elements within a period helps us predict the types of chemical bonds they will form (ionic, covalent, metallic) and the nature of their compounds.
Beyond the Basic Trends: Exceptions and Nuances
While the general trends discussed above are valuable for understanding the periodic table, it's crucial to acknowledge exceptions and nuances. The simple models used to explain these trends are approximations, and the actual behavior of elements can be influenced by complex interactions between electrons and the nucleus.
Transition metals, for instance, show less consistent trends due to the filling of inner d-orbitals. The poor shielding effect of d-electrons can lead to variations in ionization energies and atomic radii compared to what might be expected based on simple trends. Similarly, electron-electron repulsion and other factors can lead to deviations from the predicted trends for electron affinity.
Conclusion: Periods – Key to Understanding Elemental Behavior
The horizontal rows, or periods, of the periodic table are far more than just a convenient way to organize elements. They represent a fundamental aspect of atomic structure, directly reflecting the filling of principal energy levels and leading to predictable trends in elemental properties. Understanding these trends – in atomic radius, ionization energy, electronegativity, and metallic character – is crucial to comprehending the chemical behavior of elements and predicting their reactivity. While exceptions and complexities exist, the overarching principles associated with periods provide a powerful framework for interpreting the behavior of matter and the organization of the periodic table itself. This detailed understanding empowers further exploration of more complex chemical concepts and phenomena.
Latest Posts
Latest Posts
-
Cell Envelope Of Gram Negative Bacteria
Apr 03, 2025
-
What Chemical Element Has The Highest Electron Affinity
Apr 03, 2025
-
Range And Kernel Of A Linear Transformation
Apr 03, 2025
-
The Chemistry Of Living Organisms Is Called Chemistry
Apr 03, 2025
-
Finding Domain Of A Composite Function
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Horizontal Columns On The Periodic Table Are Called . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
