How Are Ionic And Covalent Bonding Similar
Muz Play
Apr 02, 2025 · 5 min read
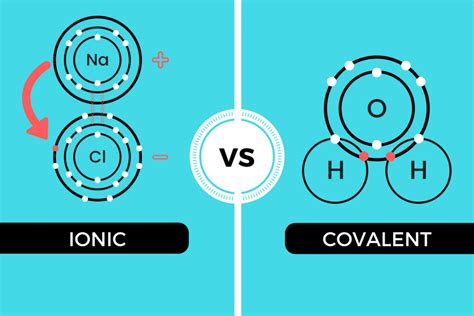
Table of Contents
How Are Ionic and Covalent Bonding Similar? Exploring the Overlapping Territories of Chemical Bonding
Chemical bonding, the driving force behind the formation of molecules and compounds, is a fundamental concept in chemistry. While distinct types of bonding exist, such as ionic and covalent bonding, it's crucial to understand their similarities alongside their differences. This detailed exploration delves into the shared characteristics of ionic and covalent bonds, highlighting the subtle nuances that connect these seemingly disparate bonding mechanisms.
The Foundation: Electrostatic Attraction
At their core, both ionic and covalent bonds are governed by the fundamental principle of electrostatic attraction. This means that the driving force behind the formation of both types of bonds is the attraction between oppositely charged particles.
-
Ionic bonds: Involve the transfer of electrons from one atom to another, creating ions with opposite charges (cations and anions). The electrostatic attraction between these oppositely charged ions holds the compound together. Think of it like magnets: a positive charge attracts a negative charge, creating a strong bond.
-
Covalent bonds: Involve the sharing of electrons between atoms. This shared electron pair creates a region of high electron density that attracts the positively charged nuclei of the bonded atoms. Again, this is electrostatic attraction – the positive nuclei are attracted to the negatively charged electron cloud.
This shared dependence on electrostatic attraction is a significant similarity, forming the bedrock of both bonding types. It's the underlying physics that unites them.
Electron Involvement: The Shared Currency of Bonding
Both ionic and covalent bonds fundamentally involve the valence electrons of the atoms participating in the bond. Valence electrons are the outermost electrons of an atom, and their behavior dictates the chemical reactivity of the element.
-
Ionic bonding: Electrons are transferred from the atom with lower electronegativity (the tendency to attract electrons) to the atom with higher electronegativity. This transfer leads to a stable electron configuration, often resembling a noble gas configuration.
-
Covalent bonding: Electrons are shared between atoms, also leading to a more stable electron configuration for each atom involved. The shared electrons contribute to the stability of both atoms.
The importance of valence electrons in both bonding types establishes another clear point of similarity. Both ionic and covalent bond formation aim for electron configurations that maximize stability, achieving a lower energy state for the overall system.
The Spectrum of Bonding: Polarity and Electronegativity
The line between purely ionic and purely covalent bonds is often blurred. The concept of electronegativity plays a crucial role in understanding this spectrum. Electronegativity measures an atom's ability to attract electrons within a chemical bond.
-
Ionic bonds: Typically form between atoms with a significant difference in electronegativity. One atom has a much stronger pull on the electrons than the other, leading to near-complete electron transfer. This results in a highly polar bond, where one end carries a strong negative charge and the other a strong positive charge.
-
Covalent bonds: Form between atoms with similar or relatively small differences in electronegativity. The electrons are shared more equally, leading to a nonpolar covalent bond if the electronegativity difference is negligible. However, even in covalent bonds, a difference in electronegativity can lead to polar covalent bonds, where one atom carries a slightly more negative charge than the other.
Therefore, while the degree varies, both ionic and covalent bonds can exhibit some degree of polarity. This overlapping characteristic demonstrates a continuum of bond types rather than strictly separate categories.
Formation of Crystalline Structures: Order and Arrangement
Both ionic and covalent bonding can lead to the formation of crystalline structures, although the nature of these structures differs significantly.
-
Ionic compounds: Often form crystalline lattices, three-dimensional structures with regular repeating patterns of positively and negatively charged ions. The strong electrostatic forces between ions create these highly ordered structures.
-
Covalent compounds: Can also form crystalline structures, though these are often less regular than ionic lattices. Network covalent solids, like diamond and quartz, represent covalent structures where atoms are connected in a continuous network. Many covalent compounds exist as discrete molecules, however, lacking the extensive three-dimensional structure of ionic compounds.
While the resulting crystal structures are different in many ways, the underlying principle – the attraction between component units leading to organized structures – is a similarity. Both bonding types drive the formation of macroscopic, ordered structures, even if the underlying forces and arrangements vary.
Properties of Compounds: A Comparative Look
The types of bonds in a compound significantly impact its physical and chemical properties. While the properties differ greatly, certain similarities emerge upon closer examination:
-
Melting and boiling points: Both ionic and covalent compounds can exhibit high melting and boiling points, although the underlying reasons differ. Ionic compounds have high melting points due to strong electrostatic attractions between ions. Covalent compounds with network structures (like diamond) also exhibit very high melting points because of the extensive network of strong covalent bonds. However, many simple covalent molecules have low melting and boiling points because of the weaker intermolecular forces.
-
Solubility: Both ionic and some covalent compounds can be soluble in certain solvents. Ionic compounds are often soluble in polar solvents (like water) due to ion-dipole interactions. Some polar covalent compounds also dissolve readily in polar solvents due to similar interactions. Nonpolar covalent compounds generally dissolve better in nonpolar solvents.
-
Electrical conductivity: Ionic compounds in molten state or dissolved in a solution conduct electricity because the ions are free to move and carry charge. However, most solid covalent compounds do not conduct electricity unless they are special semiconductors or involve conjugated pi systems that delocalize electrons.
These shared properties, while often manifested differently in magnitude or under different conditions, point towards a common origin – the fundamental interaction between atoms and their tendency toward stable configurations.
Conclusion: The Unity Beneath the Diversity
While ionic and covalent bonding represent distinct mechanisms for atomic interactions, a deeper examination reveals significant commonalities. Both rely on electrostatic attractions to hold atoms together, involve valence electrons in attaining stable configurations, can exhibit varying degrees of polarity, and can contribute to crystalline structure formation. Understanding these similarities, along with the crucial differences, offers a more comprehensive grasp of chemical bonding and the rich diversity of chemical compounds. The nuanced overlap between these bonding types highlights the interconnectedness of chemical principles and the beauty of their subtle variations. Recognizing both the commonalities and distinctions enables a more complete and sophisticated understanding of the fundamental forces shaping our physical world.
Latest Posts
Latest Posts
-
Where Is The Energy Stored In Glucose
Apr 03, 2025
-
Light Amplification By The Stimulated Emission Of Radiation
Apr 03, 2025
-
Octet Rule Violation Vs Wrong Electron Total
Apr 03, 2025
-
What Are The Three Points Of Cell Theory
Apr 03, 2025
-
How Many Atoms Are In A Simple Cubic Unit Cell
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Are Ionic And Covalent Bonding Similar . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
