In A Chemical Reaction Atoms Are
Muz Play
Apr 05, 2025 · 6 min read
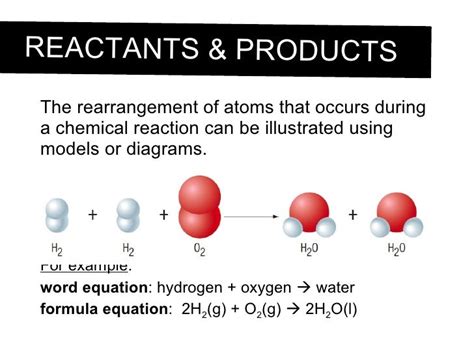
Table of Contents
In a Chemical Reaction, Atoms Are… The Unchanging Players
Chemical reactions are the fundamental processes that govern the world around us. From the rusting of iron to the photosynthesis in plants, everything is a manifestation of atoms rearranging themselves. But what exactly happens to the atoms during these transformations? The simple, yet profound, answer is: atoms are conserved. They are neither created nor destroyed; they simply rearrange themselves to form new molecules. This principle, known as the law of conservation of mass, is the cornerstone of our understanding of chemical reactions.
Understanding the Basics: Atoms and Molecules
Before diving into the complexities of chemical reactions, let's refresh our understanding of atoms and molecules. Atoms are the fundamental building blocks of matter, the smallest units that retain the chemical properties of an element. Each atom is composed of a nucleus containing protons and neutrons, surrounded by a cloud of orbiting electrons. The number of protons determines the element's identity (e.g., hydrogen has one proton, oxygen has eight).
Molecules are formed when two or more atoms chemically bond together. These bonds involve the sharing or transfer of electrons between atoms. The properties of a molecule are different from the properties of its constituent atoms. For example, two hydrogen atoms (highly reactive) combine to form a hydrogen molecule (H₂), a relatively unreactive gas. Similarly, two oxygen atoms combine to form oxygen gas (O₂), essential for respiration.
Chemical Reactions: A Dance of Atoms
A chemical reaction is essentially a process where molecules are broken down and reformed, resulting in the creation of new substances. This "breaking and reforming" involves the breaking of existing chemical bonds and the formation of new ones. Crucially, during this entire process, the atoms themselves remain unchanged. They are simply rearranged into different combinations.
Consider the classic example of the reaction between hydrogen and oxygen to form water:
2H₂ + O₂ → 2H₂O
This equation tells us that two molecules of hydrogen gas (H₂) react with one molecule of oxygen gas (O₂) to produce two molecules of water (H₂O). Let's analyze what happens at the atomic level:
- Before the reaction: We have four hydrogen atoms (2 x 2H) and two oxygen atoms (O₂).
- During the reaction: The bonds between the hydrogen atoms in H₂ and the oxygen atoms in O₂ are broken.
- After the reaction: The hydrogen and oxygen atoms rearrange themselves to form two molecules of water. Each water molecule consists of two hydrogen atoms bonded to one oxygen atom.
Notice that the total number of hydrogen atoms (four) and oxygen atoms (two) remains the same throughout the reaction. They are merely reorganized into a different arrangement. This exemplifies the law of conservation of mass.
Types of Chemical Reactions
Chemical reactions can be categorized into several types, each involving different types of atomic rearrangements:
- Synthesis (Combination) Reactions: Two or more substances combine to form a more complex substance. Example: 2Na + Cl₂ → 2NaCl (sodium and chlorine reacting to form sodium chloride).
- Decomposition Reactions: A single compound breaks down into two or more simpler substances. Example: 2H₂O → 2H₂ + O₂ (water decomposing into hydrogen and oxygen).
- Single Displacement (Replacement) Reactions: One element replaces another in a compound. Example: Zn + 2HCl → ZnCl₂ + H₂ (zinc replacing hydrogen in hydrochloric acid).
- Double Displacement (Metathesis) Reactions: Two compounds exchange ions to form two new compounds. Example: AgNO₃ + NaCl → AgCl + NaNO₃ (silver nitrate and sodium chloride reacting to form silver chloride and sodium nitrate).
- Combustion Reactions: A substance reacts rapidly with oxygen, usually producing heat and light. Example: CH₄ + 2O₂ → CO₂ + 2H₂O (methane burning in oxygen to produce carbon dioxide and water).
- Acid-Base Reactions (Neutralization Reactions): An acid reacts with a base to form salt and water. Example: HCl + NaOH → NaCl + H₂O (hydrochloric acid reacting with sodium hydroxide to form sodium chloride and water).
Visualizing Atomic Rearrangements
Understanding chemical reactions is significantly enhanced by visualizing the atomic rearrangements involved. While we cannot directly observe atoms moving and bonding, various models and representations can help us grasp this process:
- Chemical Equations: These use symbols and formulas to represent the reactants and products of a chemical reaction, providing a concise summary of the atomic rearrangements.
- Molecular Models: These physical or computer-generated models show the three-dimensional arrangement of atoms in molecules, offering a visual representation of the bonds that are broken and formed.
- Animations and Simulations: These dynamic tools allow us to "see" the atoms moving and rearranging themselves during a reaction, providing a more intuitive understanding.
The Role of Energy in Chemical Reactions
Chemical reactions are not simply about atomic rearrangement; they also involve energy changes. Some reactions release energy (exothermic reactions), while others require energy input to proceed (endothermic reactions). This energy is often manifested as heat, light, or other forms of energy. The energy changes are related to the strength of the chemical bonds involved. Breaking bonds generally requires energy input, while forming bonds usually releases energy.
Beyond Simple Reactions: Complex Chemical Processes
Many real-world chemical processes involve far more complex atomic rearrangements than the simple examples discussed above. These can include:
- Polymerization: The formation of large molecules (polymers) from smaller molecules (monomers). This process is crucial in the formation of plastics, proteins, and DNA.
- Enzyme Catalysis: Biological reactions are often catalyzed by enzymes, proteins that speed up the reaction rate by lowering the activation energy. The enzymes facilitate specific atomic rearrangements without being consumed in the process.
- Redox Reactions (Oxidation-Reduction Reactions): These involve the transfer of electrons between atoms, leading to changes in oxidation states. These reactions are fundamental to many processes, including respiration and corrosion.
Isotopes and Chemical Reactions
Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons. While isotopes have slightly different masses, they participate in chemical reactions in essentially the same way. The number of protons and electrons, which dictate chemical behavior, remain unchanged. Therefore, isotopes do not violate the law of conservation of mass in chemical reactions.
Applications of Understanding Atomic Rearrangements
The understanding of atomic rearrangements in chemical reactions is essential for numerous applications:
- Materials Science: Designing new materials with specific properties requires a deep understanding of how atoms interact and arrange themselves.
- Medicine: Drug development relies heavily on understanding chemical reactions within the body. Many drugs function by interacting with specific molecules, affecting atomic arrangements and biological processes.
- Environmental Science: Understanding chemical reactions is crucial for analyzing pollution, developing remediation strategies, and studying environmental processes.
- Industrial Chemistry: Chemical processes in industries like manufacturing, agriculture, and energy production depend on controlling and optimizing chemical reactions.
Conclusion: The Fundamental Nature of Chemical Change
In essence, chemical reactions are a dynamic dance of atoms. These submicroscopic particles, the fundamental building blocks of matter, are the unchanging players in this intricate choreography. They are neither created nor destroyed but simply rearrange themselves, breaking and forming bonds to create new substances with different properties. Understanding this fundamental principle is essential to comprehending the world around us and harnessing the power of chemical transformations for human benefit. From the simplest reactions to the most complex biological processes, the conservation of atoms remains the immutable law governing all chemical change. The constant rearrangement and recombination of these atoms create the diversity and dynamism of the chemical world. Further exploration of this fundamental concept will unlock even greater understanding and innovation across various scientific and technological fields.
Latest Posts
Latest Posts
-
Why Is Density An Intensive Property
Apr 06, 2025
-
Which Of The Following Bones Belong To The Axial Skeleton
Apr 06, 2025
-
What Are The Properties Of Solids
Apr 06, 2025
-
Trends In The Periodic Table Answer Key
Apr 06, 2025
-
How Do Intermolecular Forces Affect Viscosity
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about In A Chemical Reaction Atoms Are . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
