Is Magnesium A Metal Nonmetal Or Metalloid
Muz Play
Apr 02, 2025 · 5 min read
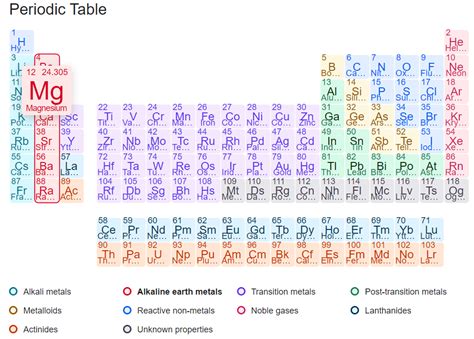
Table of Contents
Is Magnesium a Metal, Nonmetal, or Metalloid? A Comprehensive Exploration
Magnesium, a ubiquitous element found in numerous biological and industrial applications, often sparks curiosity regarding its classification within the periodic table's elemental categories. Is it a metal, nonmetal, or metalloid? The answer, decisively, is metal. However, understanding why it's classified as a metal requires a deeper dive into its properties and behavior. This comprehensive exploration will unravel the intricacies of magnesium's classification, examining its physical and chemical characteristics, comparing it to metalloids and nonmetals, and highlighting its practical applications.
Understanding the Elemental Categories
Before delving into the specifics of magnesium, let's briefly review the characteristics that define metals, nonmetals, and metalloids. This foundational understanding is crucial for accurately classifying any element.
Metals: A Definition
Metals are generally characterized by their:
- High electrical conductivity: Metals readily conduct electricity due to the presence of freely moving electrons in their structure. This allows for the easy flow of electrical current.
- High thermal conductivity: Similarly, metals efficiently transfer heat due to the mobility of their electrons. This property makes them ideal for applications requiring heat dissipation.
- Malleability and ductility: Metals can be easily shaped or drawn into wires without breaking. This property stems from the ability of metal atoms to slide past each other without disrupting the metallic bond.
- Metallic luster: Most metals possess a characteristic shine or luster, reflecting light effectively. This is a visual characteristic often associated with metals.
- High density: Metals generally have relatively high densities compared to nonmetals.
- Positive charge when ionized: Metals tend to lose electrons easily, forming positively charged ions (cations). This is a key factor in their chemical reactivity.
Nonmetals: Properties and Contrasts
Nonmetals, in stark contrast to metals, typically exhibit:
- Low electrical conductivity: Nonmetals are generally poor conductors of electricity.
- Low thermal conductivity: They also poorly conduct heat.
- Brittleness: Nonmetals are often brittle and lack the malleability and ductility of metals.
- Dull appearance: They lack the metallic luster observed in metals.
- Lower density: Nonmetals usually have lower densities than metals.
- Negative charge when ionized: Nonmetals tend to gain electrons, forming negatively charged ions (anions).
Metalloids: Bridging the Gap
Metalloids occupy a fascinating middle ground between metals and nonmetals, exhibiting properties of both categories. Their characteristics are often intermediate and can vary significantly depending on the specific metalloid and the conditions it's subjected to. Key features of metalloids include:
- Semiconductivity: Metalloids are semiconductors, meaning their electrical conductivity is intermediate between metals and nonmetals. Their conductivity can be altered by factors such as temperature and the presence of impurities.
- Variable properties: Their properties can vary greatly depending on the specific metalloid and environmental conditions.
- Brittle nature: Like many nonmetals, metalloids are typically brittle.
Magnesium: A Detailed Examination
Now that we have established the defining characteristics of the three elemental categories, let's scrutinize magnesium's properties to definitively place it within the classification system.
Magnesium's Metallic Characteristics
Magnesium overwhelmingly exhibits properties consistent with those of metals:
- Excellent electrical conductivity: Magnesium is a good conductor of electricity, a crucial property exploited in various applications.
- Good thermal conductivity: It efficiently conducts heat, making it useful in heat exchange systems.
- Malleable and ductile: Although less so than some other metals, magnesium can be shaped and drawn into wires.
- Metallic luster: Magnesium possesses a silvery-white metallic luster.
- Relatively high density: Its density is higher than that of many nonmetals.
- Formation of cations: Magnesium readily loses two electrons to form Mg²⁺ cations, a hallmark of metallic behavior. This ionic characteristic underpins its reactivity and its role in various chemical reactions.
Magnesium's Chemical Reactivity: Further Evidence of Metallic Nature
Magnesium's chemical reactivity further solidifies its classification as a metal. It readily reacts with:
- Oxygen: Magnesium reacts vigorously with oxygen, forming magnesium oxide (MgO), a reaction often demonstrated in introductory chemistry classes to illustrate the energy released in oxidation.
- Acids: Magnesium reacts with acids, releasing hydrogen gas. This reaction is a classic example of a metal displacement reaction.
- Halides: Magnesium reacts with halogens (such as chlorine and bromine) to form magnesium halides.
Comparing Magnesium to Metalloids and Nonmetals
The contrast between magnesium and metalloids or nonmetals is stark. Magnesium lacks the semiconducting properties characteristic of metalloids. Its high electrical and thermal conductivity, malleability, ductility, and metallic luster definitively distinguish it from nonmetals. Its reactivity with acids and halogens, forming ionic compounds, further reinforces its metallic nature.
Magnesium's Applications: A Testament to its Metallic Properties
The diverse applications of magnesium are a direct result of its metallic characteristics. Its lightness, strength (relative to its weight), and reactivity are exploited across various industries:
- Automotive industry: Magnesium alloys are used in automotive parts to reduce weight and improve fuel efficiency. This lightweight property is a key advantage of using magnesium in vehicles.
- Aerospace industry: Similarly, its lightweight and high strength-to-weight ratio make it valuable in aerospace applications. Weight reduction is critical in aircraft and spacecraft design.
- Electronics: Magnesium is used in electronic components, leveraging its electrical conductivity and ability to dissipate heat.
- Biomedical applications: Magnesium's biocompatibility makes it suitable for implants and other biomedical applications. Its role in biological processes also makes it important in healthcare.
- Pyrotechnics: Magnesium's highly exothermic reaction with oxygen makes it a key component in flares and fireworks, producing a bright white light.
Conclusion: Magnesium – Undeniably a Metal
In conclusion, there is no ambiguity in the classification of magnesium. Its physical and chemical properties definitively place it firmly within the metal category. Its high electrical and thermal conductivity, malleability, ductility, metallic luster, and characteristic chemical reactivity all align perfectly with the definition of a metal. The wide range of applications, from automotive engineering to biomedical devices, further underscores the significance of its metallic properties. While other elements might blur the lines between categories, magnesium's metallic identity is undeniable.
Latest Posts
Latest Posts
-
Energy Flow In Plants Concept Map
Apr 03, 2025
-
Chondrocytes Are To Cartilage As Osteocytes Are To
Apr 03, 2025
-
What Is The Dielectric Constant Of Water
Apr 03, 2025
-
Draw A Phasor Diagram Showing Generator Voltage At Time 0
Apr 03, 2025
-
What Is A Wet Mount Slide
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Is Magnesium A Metal Nonmetal Or Metalloid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
