Location Of Metals On The Periodic Table
Muz Play
Apr 02, 2025 · 6 min read
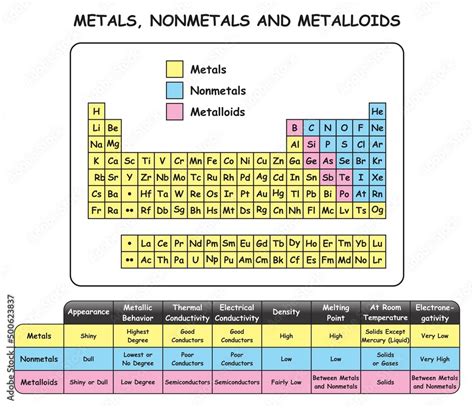
Table of Contents
The Location of Metals on the Periodic Table: A Comprehensive Guide
The periodic table is a cornerstone of chemistry, organizing elements based on their atomic structure and properties. Understanding the location of metals on this table is crucial for comprehending their chemical behavior and applications. This comprehensive guide explores the systematic arrangement of metals, delving into their properties, groups, and exceptions.
Understanding the Periodic Table's Organization
The periodic table's arrangement is no accident; it reflects the underlying structure of atoms. Elements are ordered by increasing atomic number (the number of protons in the nucleus). This arrangement reveals recurring patterns in properties, famously known as the periodic law. These properties are directly tied to the arrangement of electrons in the atom's electron shells. It's the outermost electrons, the valence electrons, that largely dictate an element's chemical reactivity and whether it behaves as a metal, nonmetal, or metalloid.
Key Areas of Metal Location
Metals predominantly occupy the left side and middle of the periodic table. A clear diagonal line running from Boron (B) to Astatine (At) roughly separates metals from nonmetals. Elements along this line, called metalloids or semimetals, exhibit properties intermediate between metals and nonmetals.
Major Groups of Metals:
-
Alkali Metals (Group 1): These highly reactive metals are located in the first column, excluding hydrogen. Their single valence electron makes them readily lose it to form +1 ions. They are soft, silvery-white, and have low densities. Examples include Lithium (Li), Sodium (Na), and Potassium (K).
-
Alkaline Earth Metals (Group 2): Located in the second column, these metals are also highly reactive, but less so than alkali metals. They have two valence electrons, readily forming +2 ions. They are harder, denser, and have higher melting points than alkali metals. Examples include Beryllium (Be), Magnesium (Mg), and Calcium (Ca).
-
Transition Metals (Groups 3-12): This large block in the middle of the table contains elements with partially filled d orbitals. This allows for variable oxidation states, leading to diverse chemical behavior and numerous complex ions. Many are known for their strength, durability, and catalytic properties. Examples include Iron (Fe), Copper (Cu), and Zinc (Zn).
-
Inner Transition Metals (Lanthanides and Actinides): These elements are placed separately at the bottom of the table to maintain the table's compactness. The lanthanides fill the 4f orbitals, while the actinides fill the 5f orbitals. Many are radioactive.
-
Post-Transition Metals: These metals, found in Groups 13-15, are located to the right of the transition metals. They have properties that transition toward the metalloids, exhibiting lower melting and boiling points, and some show varying oxidation states, though typically less diverse than the transition metals. Examples include Aluminum (Al), Tin (Sn), and Lead (Pb).
Properties of Metals and Their Location
The location of a metal on the periodic table directly correlates with its properties. These properties stem from the metallic bonding characteristic of metals.
Key Metallic Properties:
-
High Electrical Conductivity: Metals readily conduct electricity due to the delocalized electrons in their metallic bonding structure. This conductivity generally decreases as you move across a period (left to right) and increases down a group. Alkali metals are exceptionally good conductors.
-
High Thermal Conductivity: Metals efficiently transfer heat, owing to the mobility of the delocalized electrons. This property, like electrical conductivity, is generally higher down a group and lower across a period.
-
Malleability and Ductility: Metals can be easily hammered into thin sheets (malleability) and drawn into wires (ductility) without breaking. These properties result from the ability of metal atoms to slide past one another without disrupting the metallic bonding.
-
Metallic Luster: Metals typically have a shiny, reflective surface. This is due to the interaction of light with the delocalized electrons in the metallic structure.
-
High Melting and Boiling Points: The strong metallic bonds necessitate considerable energy to overcome, resulting in generally high melting and boiling points. However, exceptions exist, particularly among alkali metals which possess comparatively low melting and boiling points due to weaker metallic bonds.
-
Low Ionization Energies: Metals tend to have low ionization energies, meaning they readily lose electrons to form positive ions (cations). This tendency is most pronounced for alkali metals, with the lowest ionization energies across the entire periodic table.
Exceptions and Anomalies
While the general trend of metal location is clear, some exceptions exist:
-
Hydrogen: Although placed in Group 1, hydrogen behaves more like a nonmetal under standard conditions. However, under high pressure, it can exhibit metallic properties.
-
Metalloids (Semimetals): These elements, positioned along the metalloid staircase, possess properties intermediate between metals and nonmetals. Their behavior can vary significantly depending on the conditions.
-
Some Post-Transition Metals: Exhibiting properties more similar to metalloids as the group progresses. For instance, while Aluminum is a clear metal, elements lower in its group (such as Gallium and Thallium) show increasingly non-metallic characteristics.
Applications of Metals and Their Periodic Table Position
The properties of metals, directly related to their location on the periodic table, lead to their widespread applications.
-
Alkali Metals: Used in batteries (lithium-ion batteries) and as reducing agents (sodium and potassium). Their high reactivity necessitates handling precautions.
-
Alkaline Earth Metals: Magnesium alloys are used in aircraft construction due to their lightweight and strength. Calcium is crucial in biological systems.
-
Transition Metals: Iron is essential in steel production; Copper in electrical wiring and plumbing; Platinum and Palladium serve as catalysts in numerous chemical processes. The diverse oxidation states and complex ion formations of transition metals make them versatile in many applications.
-
Post-Transition Metals: Aluminum is widely used in packaging, construction, and transportation due to its lightweight, strength, and corrosion resistance. Tin and lead have historically been used in alloys like solder.
Advanced Concepts and Future Directions
The study of metal location and properties extends into more advanced areas of chemistry, including:
-
Alloying: Combining metals to create materials with enhanced properties, such as strength, corrosion resistance, and specific electrical conductivity. Alloy composition and properties are heavily influenced by the constituent metals' positions and properties on the periodic table.
-
Solid-State Chemistry: Investigating the crystalline structures and electronic properties of metals and metal compounds, which is crucial for materials science and semiconductor technology.
-
Catalysis: Understanding how the electronic structure and oxidation states of metals influence their catalytic activity, a critical aspect of chemical engineering and industrial processes.
-
Nanomaterials: Creating and studying metal nanoparticles and nanostructures, leading to advanced materials with unique properties. The nanoscale manipulation of metal properties directly relates to their fundamental characteristics, determined by their location on the periodic table.
Conclusion:
The periodic table offers a powerful framework for understanding the properties and applications of metals. The systematic arrangement reveals patterns in their chemical behavior, allowing us to predict properties and design new materials. By understanding the location of metals and the interplay of their properties, chemists and material scientists continue to develop innovative technologies with transformative potential. The study of metals and their placement on the periodic table remains a dynamic and vital field, constantly evolving with new discoveries and applications.
Latest Posts
Latest Posts
-
Where Is The Energy Stored In Glucose
Apr 03, 2025
-
Light Amplification By The Stimulated Emission Of Radiation
Apr 03, 2025
-
Octet Rule Violation Vs Wrong Electron Total
Apr 03, 2025
-
What Are The Three Points Of Cell Theory
Apr 03, 2025
-
How Many Atoms Are In A Simple Cubic Unit Cell
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Location Of Metals On The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
