Lock And Key Model Vs Induced Fit Model
Muz Play
Apr 02, 2025 · 6 min read
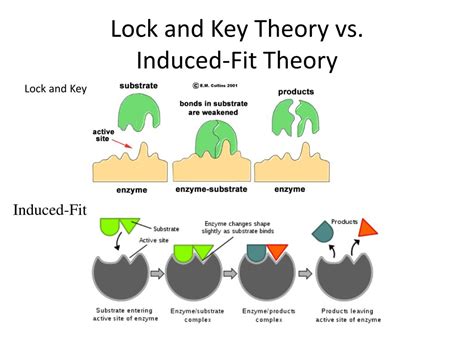
Table of Contents
Lock and Key vs. Induced Fit Model: A Deep Dive into Enzyme-Substrate Interactions
The interaction between enzymes and their substrates is fundamental to all biological processes. Two prominent models attempt to explain this intricate dance: the lock and key model and the induced fit model. While both models offer valuable insights, the induced fit model provides a more accurate and comprehensive understanding of enzyme-substrate interactions, especially when considering the nuances of enzyme specificity, catalytic efficiency, and regulatory mechanisms.
The Lock and Key Model: A Simple Analogy
Proposed by Emil Fischer in 1894, the lock and key model presents a simple and intuitive analogy. It suggests that the enzyme's active site possesses a rigid, pre-formed structure, perfectly complementary to the shape of its specific substrate. The substrate, like a key fitting into a lock, binds precisely to the active site, initiating the enzymatic reaction. This model elegantly explains enzyme specificity—only the correct substrate, with the matching shape, can fit and bind to the active site.
Strengths of the Lock and Key Model
- Simplicity: The model is easy to understand and visualize, making it a useful introductory concept in biochemistry.
- Specificity: It effectively explains the high degree of specificity observed in many enzyme-substrate interactions. Only substrates with the complementary shape can bind.
- Initial Understanding: It provided a foundational understanding of enzyme-substrate interactions before more detailed studies were conducted.
Limitations of the Lock and Key Model
The lock and key model, while a useful simplification, fails to account for several crucial aspects of enzyme-substrate interactions:
- Rigidity: It assumes a rigid enzyme structure, which is not always the case. Enzymes are dynamic molecules with flexible structures that can change shape.
- Transition State: It doesn't adequately address the formation of the transition state, a high-energy intermediate state in the reaction pathway. Enzyme catalysis involves stabilizing the transition state, a process not explained by a rigid lock and key interaction.
- Induced Conformational Changes: Experimental evidence shows that enzymes undergo significant conformational changes upon substrate binding, a phenomenon not incorporated into the lock and key model.
- Enzyme Regulation: It doesn't explain the allosteric regulation of enzymes, where binding of a molecule at a site other than the active site affects enzyme activity.
The Induced Fit Model: A More Dynamic Perspective
Daniel Koshland proposed the induced fit model in 1958, refining Fischer's original concept. This model acknowledges the dynamic nature of enzymes and incorporates conformational changes upon substrate binding. The active site is not a rigid, pre-formed structure but rather a flexible site that adapts its shape to accommodate the substrate. Substrate binding induces conformational changes in the enzyme, leading to a more precise fit and optimized catalysis.
The Mechanism of Induced Fit
The induced fit model describes a multi-step process:
- Initial Interaction: The substrate approaches the enzyme's active site, initiating weak interactions (e.g., hydrogen bonds, van der Waals forces). These interactions are not perfectly complementary; some conformational adjustments are necessary.
- Conformational Change: The weak initial interactions trigger conformational changes in the enzyme's active site. Amino acid residues rearrange themselves to create a more complementary shape for the substrate, enhancing binding affinity.
- Tight Binding: The substrate binds more tightly to the enzyme's active site, which now has a shape that's optimized for catalysis.
- Catalysis: The enzyme catalyzes the reaction, converting the substrate into products.
- Product Release: The products are released, and the enzyme returns to its original or a similar conformation, ready to catalyze another reaction.
Strengths of the Induced Fit Model
The induced fit model offers several advantages over the lock and key model:
- Flexibility: It accounts for the flexibility of enzymes and the dynamic nature of enzyme-substrate interactions.
- Transition State Stabilization: It explains how enzymes stabilize the transition state, a crucial step in accelerating reaction rates. The conformational changes optimize the active site for binding and stabilizing the transition state.
- Specificity and Regulation: It accommodates both high enzyme specificity and regulatory mechanisms, such as allosteric regulation. Conformational changes can be induced by allosteric effectors, influencing the enzyme's affinity for its substrate.
- Experimental Support: Extensive experimental evidence from techniques like X-ray crystallography and NMR spectroscopy supports the induced fit mechanism, revealing conformational changes upon substrate binding.
The Role of Conformational Changes
The conformational changes involved in induced fit are not merely passive adjustments; they are crucial for catalysis. These changes can involve:
- Optimizing substrate binding: Conformational changes bring catalytic residues closer to the substrate, enhancing interactions and promoting catalysis.
- Creating the optimal transition state: The induced fit optimizes the active site's shape and charge distribution to stabilize the transition state, thus lowering the activation energy.
- Excluding water molecules: Conformational changes can exclude water molecules from the active site, preventing unwanted side reactions.
- Substrate orientation: The induced fit ensures the substrate is oriented correctly for optimal interaction with catalytic residues.
Comparing the Two Models
| Feature | Lock and Key Model | Induced Fit Model |
|---|---|---|
| Enzyme Shape | Rigid, pre-formed active site | Flexible active site, changes upon substrate binding |
| Substrate Binding | Precise, immediate fit | Initial weak interactions, followed by conformational changes |
| Transition State | Does not explicitly address transition state stabilization | Explains transition state stabilization through conformational changes |
| Specificity | Explained by pre-formed complementary shape | Explained by induced fit and optimal substrate orientation |
| Regulation | Does not adequately explain allosteric regulation | Accommodates allosteric regulation through conformational changes |
| Experimental Support | Limited | Strong experimental evidence |
Beyond the Models: A More Nuanced View
While the induced fit model provides a significantly more accurate description than the lock and key model, it's crucial to recognize that enzyme-substrate interactions are incredibly complex. Recent research suggests that even the induced fit model is a simplification. The reality is likely a dynamic interplay between multiple factors, including:
- Conformational flexibility: Enzymes exhibit a range of conformational states, and substrate binding might select a specific conformation, rather than inducing a single, defined change.
- Dynamic interactions: The interaction between enzyme and substrate isn't a static "fit" but rather a continuous dynamic process involving multiple conformational changes.
- Environmental factors: The environment (pH, temperature, ionic strength) plays a critical role in influencing both enzyme and substrate conformation and consequently affecting the interaction.
Conclusion: Induced Fit - The Dominant Paradigm
The lock and key model serves as a valuable introductory concept, providing a basic understanding of enzyme specificity. However, the induced fit model offers a far more accurate and comprehensive representation of enzyme-substrate interactions. It successfully incorporates enzyme flexibility, transition state stabilization, and regulatory mechanisms, aligning more closely with experimental observations. While research continues to refine our understanding of this intricate process, the induced fit model remains the dominant paradigm in explaining the dynamic and complex relationship between enzymes and their substrates, a cornerstone of biochemical processes vital for life itself. Further research continues to elucidate the finer details of this dynamic interaction, revealing an ever-more intricate picture of enzyme catalysis. The ongoing investigation highlights the power of scientific models to evolve and adapt to accommodate new findings, leading to a deeper appreciation of the complexity of biological systems.
Latest Posts
Latest Posts
-
Image Formation In A Plane Mirror
Apr 03, 2025
-
Us Air Force Tongue And Quill
Apr 03, 2025
-
The Coefficients In A Chemical Equation Represent The
Apr 03, 2025
-
How To Calculate The Gradient Of A Stream
Apr 03, 2025
-
What Does Insoluble Mean In Chemistry
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Lock And Key Model Vs Induced Fit Model . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
