What Are The Three Characteristics Of All Metals
Muz Play
Apr 02, 2025 · 6 min read
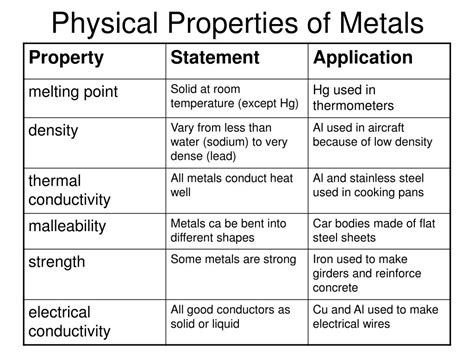
Table of Contents
What Are the Three Characteristics of All Metals? A Deep Dive into Metallic Properties
Metals. We interact with them daily, from the spoons in our kitchens to the skyscrapers that pierce the sky. But what fundamentally defines a metal? While a vast array of metals exist, showcasing diverse properties, three core characteristics unite them all: electrical conductivity, thermal conductivity, and malleability/ductility. This article will delve deep into each of these, exploring their underlying mechanisms and the exceptions that prove the rule.
1. Electrical Conductivity: The Free Electron Sea
One of the most defining features of metals is their exceptional ability to conduct electricity. Unlike insulators, where electrons are tightly bound to their atoms, metals possess a unique electronic structure that allows for the free flow of charge. This is best understood through the concept of the "sea of electrons".
The Sea of Electrons Model
In a metallic solid, the outermost valence electrons of the constituent atoms are not localized to any single atom. Instead, they are delocalized, forming a "sea" of mobile electrons that are free to move throughout the entire metallic lattice. This sea of electrons acts as a medium for charge transport. When an electric field is applied across a metal, these delocalized electrons readily respond to the field, moving in a directed manner, thus constituting an electric current.
Factors Affecting Electrical Conductivity
While all metals conduct electricity, the degree of conductivity varies considerably. Several factors influence this:
-
Temperature: Electrical conductivity generally decreases with increasing temperature. As temperature rises, the metal atoms vibrate more vigorously, disrupting the flow of electrons and increasing resistance.
-
Purity: Impurities in a metal act as scattering centers for electrons, reducing conductivity. Highly pure metals generally exhibit higher conductivity.
-
Crystal Structure: The arrangement of atoms in the metal's crystal lattice influences electron mobility. A well-ordered lattice facilitates electron flow, while defects in the lattice can hinder it.
-
Presence of Alloys: Alloys, mixtures of two or more metals, can exhibit conductivity properties that differ from their constituent elements. The addition of alloying elements can either increase or decrease conductivity depending on the specific elements and their concentration.
Superconductivity: An Extreme Example
At extremely low temperatures, certain metals exhibit a remarkable phenomenon called superconductivity. In this state, the electrical resistance drops to virtually zero, allowing for the flow of current without any energy loss. This property has significant implications for various technologies, including medical imaging (MRI) and high-speed trains (Maglev).
2. Thermal Conductivity: Transferring Heat Energy
Just as metals readily conduct electricity, they also efficiently conduct heat. This thermal conductivity is directly linked to the same "sea of electrons" responsible for electrical conductivity.
Mechanism of Heat Transfer
When one part of a metal is heated, the increased kinetic energy of the atoms and the free electrons is transferred through the metal lattice via collisions. The mobile electrons, in particular, play a crucial role in rapidly distributing this thermal energy throughout the material. This efficient heat transfer is what allows metals to feel cold to the touch (they quickly draw heat away from your hand) and makes them ideal for applications requiring efficient heat dissipation, such as cooking utensils and heat sinks in electronic devices.
Factors Influencing Thermal Conductivity
Similar to electrical conductivity, several factors affect a metal's thermal conductivity:
-
Temperature: Thermal conductivity generally decreases with increasing temperature, although the relationship is more complex than for electrical conductivity.
-
Purity: Impurities scatter phonons (lattice vibrations) and electrons, reducing thermal conductivity. Higher purity usually equates to higher thermal conductivity.
-
Crystal Structure: A well-ordered lattice facilitates efficient heat transfer, whereas defects can impede it.
-
Alloys: The thermal conductivity of alloys can be significantly different from that of their constituent elements.
Thermal Conductivity and Electrical Conductivity: The Wiedemann-Franz Law
The close relationship between thermal and electrical conductivity in metals is quantified by the Wiedemann-Franz law. This law states that the ratio of thermal conductivity to electrical conductivity is approximately proportional to the absolute temperature. This strong correlation reflects the shared mechanism of electron movement responsible for both properties.
3. Malleability and Ductility: Shaping Metals
Unlike brittle materials that fracture easily under stress, metals exhibit remarkable malleability and ductility. These properties refer to a metal's ability to be deformed without fracturing.
Malleability: Shaping into Sheets
Malleability describes a metal's ability to be deformed into thin sheets by hammering or rolling. This is possible because the metallic bonds are relatively non-directional, allowing the metal atoms to slide past each other under applied pressure without breaking the overall structure. The "sea of electrons" helps maintain cohesion even during significant deformation.
Ductility: Drawing into Wires
Ductility, on the other hand, refers to a metal's ability to be drawn into wires. This process involves pulling the metal through a die, reducing its cross-sectional area and increasing its length. Again, the non-directional nature of metallic bonding and the mobility of electrons allow for this extensive deformation without fracturing.
Factors Affecting Malleability and Ductility
Several factors influence a metal's malleability and ductility:
-
Temperature: Higher temperatures generally enhance malleability and ductility. Increased thermal energy allows atoms to move more readily, facilitating deformation.
-
Crystal Structure: The crystal structure significantly impacts a metal's ability to deform. Face-centered cubic (FCC) metals, like gold and copper, are typically highly malleable and ductile. Body-centered cubic (BCC) metals show varying degrees of malleability and ductility.
-
Grain Size: The size of the individual crystallites (grains) in the metal affects its mechanical properties. Smaller grains generally lead to improved strength and ductility.
-
Impurities: Impurities can hinder the movement of dislocations (crystal imperfections) responsible for plastic deformation, thereby reducing malleability and ductility.
Exceptions to the Rule: Brittle Metals
While most metals exhibit good malleability and ductility, some exceptions exist. Certain metals, particularly those with high-strength covalent bonding characteristics, can exhibit brittle behavior. This is often related to their crystal structure and the presence of impurities. For instance, some metals in specific crystalline forms or under specific conditions might be brittle, highlighting the complexity of material properties.
Conclusion: The Interplay of Metallic Properties
The three fundamental characteristics – electrical conductivity, thermal conductivity, and malleability/ductility – arise from the unique electronic structure of metals: the "sea of electrons." While these properties are generally exhibited by all metals, the degree to which they are expressed varies considerably depending on factors such as temperature, purity, crystal structure, and alloying. Understanding these fundamental properties and the factors that influence them is essential for selecting appropriate materials for diverse engineering and technological applications. The continued exploration of these properties remains crucial for advancing materials science and engineering, leading to the development of new materials with tailored properties for emerging technologies. Furthermore, the exceptions to these rules provide opportunities for deeper investigation and refinement of our understanding of metallic bonding and behavior.
Latest Posts
Latest Posts
-
What Is The Dielectric Constant Of Water
Apr 03, 2025
-
Draw A Phasor Diagram Showing Generator Voltage At Time 0
Apr 03, 2025
-
What Is A Wet Mount Slide
Apr 03, 2025
-
Examples Of Stoichiometry In Real Life
Apr 03, 2025
-
Why Is Water Necessary For Biological Organisms
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Are The Three Characteristics Of All Metals . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
