What Does The Equivalence Point Of A Titration Indicate
Muz Play
Apr 05, 2025 · 6 min read
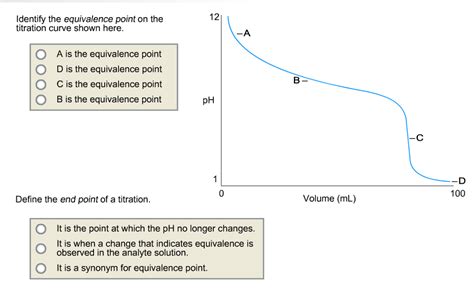
Table of Contents
What Does the Equivalence Point of a Titration Indicate?
Titration, a cornerstone technique in analytical chemistry, is a crucial method for determining the concentration of an unknown solution (analyte) using a solution of known concentration (titrant). The process involves the gradual addition of the titrant to the analyte until a reaction is complete, signaled by a change in color (using an indicator) or a significant change in pH. Understanding the equivalence point is fundamental to interpreting the results of a titration. This article will delve deeply into what the equivalence point indicates, its significance, how it differs from the endpoint, and the various factors that can affect its determination.
Understanding the Equivalence Point
The equivalence point in a titration is the point at which the amount of titrant added is stoichiometrically equivalent to the amount of analyte present. This means the moles of titrant have completely reacted with the moles of analyte according to the balanced chemical equation. At this point, the chemical reaction between the titrant and analyte is theoretically complete. It's a crucial theoretical point, representing the perfect neutralization or completion of the reaction. Importantly, the equivalence point isn't directly observable; it's a calculated point.
For example: In a strong acid-strong base titration, the equivalence point is reached when the number of moles of acid equals the number of moles of base. The solution at the equivalence point is neutral (pH 7), as all the acid and base have reacted to form water and salt. However, this is only true for strong acid-strong base titrations. In other titrations, the pH at the equivalence point can differ significantly from 7.
Different Types of Titrations and their Equivalence Points:
The nature of the equivalence point varies depending on the type of titration:
-
Strong Acid-Strong Base Titration: The equivalence point is at pH 7. The pH changes sharply near the equivalence point, making it relatively easy to detect.
-
Weak Acid-Strong Base Titration: The equivalence point is above pH 7 (basic). The pH change near the equivalence point is less sharp than in strong acid-strong base titrations. The resulting salt undergoes hydrolysis, leading to a basic solution.
-
Strong Acid-Weak Base Titration: The equivalence point is below pH 7 (acidic). Similar to weak acid-strong base titrations, the pH change is less sharp, and the resulting salt undergoes hydrolysis resulting in an acidic solution.
-
Weak Acid-Weak Base Titration: The equivalence point is near pH 7 but is difficult to detect due to the gradual change in pH. The pH change around the equivalence point is very gradual, making it challenging to pinpoint precisely. This makes these titrations less accurate.
The Difference Between Equivalence Point and Endpoint
While often used interchangeably, the equivalence point and the endpoint are distinct. The endpoint is the point at which the indicator changes color, signaling the apparent completion of the titration. The endpoint is an observable point, while the equivalence point is a calculated point. Ideally, the endpoint should be as close as possible to the equivalence point, but there's always a small difference, known as the titration error.
Factors Affecting the Difference Between Equivalence Point and Endpoint:
Several factors can influence the difference between the equivalence point and the endpoint:
-
Indicator choice: The pH range over which an indicator changes color must overlap with the steepest part of the titration curve near the equivalence point. A poorly chosen indicator can lead to a significant difference between the equivalence point and the endpoint.
-
Concentration of the analyte and titrant: Highly diluted solutions may show a less sharp change in pH near the equivalence point, making endpoint determination less precise.
-
Temperature: Temperature affects the ionization of the acid or base, influencing the pH at the equivalence point.
-
Impurities: The presence of impurities in the analyte or titrant can affect the reaction stoichiometry and the equivalence point.
-
Observer error: The subjective nature of observing the color change of the indicator can introduce human error in determining the endpoint.
Determining the Equivalence Point: Graphical and Mathematical Methods
The equivalence point is usually determined graphically using a titration curve. A titration curve is a plot of pH (or another suitable property) versus the volume of titrant added.
Graphical Method:
-
Constructing the Titration Curve: The data obtained during the titration (volume of titrant added and corresponding pH) are plotted on a graph.
-
Identifying the Equivalence Point: The equivalence point is identified as the point of steepest slope (the midpoint of the vertical portion) on the titration curve. For strong acid-strong base titrations, this point corresponds to a pH of 7. However, for other titrations, it may be at a different pH value. For weak acid-weak base titrations, the steepest part of the curve may not be easily identifiable, making the determination less accurate.
-
First Derivative Method: The first derivative of the titration curve can be used to pinpoint the equivalence point more precisely. The maximum value of the first derivative corresponds to the equivalence point.
-
Second Derivative Method: The second derivative of the titration curve can be even more precise; the point where the second derivative is zero (or crosses the x-axis) denotes the equivalence point.
Mathematical Method:
The equivalence point can also be determined mathematically using the stoichiometry of the reaction. This method requires knowing the balanced chemical equation and the concentration of the titrant.
-
Calculating Moles of Titrant: The moles of titrant added at the equivalence point are calculated using the volume and concentration of the titrant.
-
Using Stoichiometry: Using the stoichiometric ratio from the balanced chemical equation, the moles of analyte are determined from the moles of titrant at the equivalence point.
-
Calculating Concentration of Analyte: The concentration of the analyte is then calculated by dividing the moles of analyte by the initial volume of the analyte solution.
Significance of the Equivalence Point
The equivalence point holds paramount significance in titration analysis:
-
Accurate Concentration Determination: The primary purpose of a titration is to determine the concentration of an unknown solution. The equivalence point provides the basis for this calculation. An accurate determination of the equivalence point is crucial for obtaining an accurate concentration.
-
Understanding Reaction Stoichiometry: The equivalence point demonstrates the stoichiometric relationship between the titrant and the analyte. This provides insight into the chemical reaction taking place, reinforcing an understanding of fundamental chemistry principles.
-
Quality Control: Titration is widely used in quality control across various industries, including pharmaceuticals, food and beverage, and environmental monitoring. The equivalence point helps ensure the quality and purity of materials by providing accurate measurements of concentration.
-
Research and Development: Titration is instrumental in research and development, particularly in areas like drug discovery and materials science. Precise determination of concentrations using titration aids in understanding reaction kinetics and optimizing processes.
-
Environmental Monitoring: Titration plays a critical role in monitoring environmental pollutants. Accurate measurements of pollutants' concentrations, determined using titration at its equivalence point, are crucial for environmental protection and management.
Conclusion
The equivalence point in a titration represents the theoretical point at which the reaction between the titrant and analyte is stoichiometrically complete. It is a crucial parameter for determining the concentration of the unknown analyte. While it's not directly observable, it can be accurately determined using graphical or mathematical methods, typically by analyzing the titration curve. Understanding the equivalence point and its distinction from the endpoint is essential for accurate and reliable titration results, which have far-reaching applications in various fields of science and industry. Accurate determination of the equivalence point, therefore, is paramount for obtaining reliable and meaningful results in analytical chemistry.
Latest Posts
Latest Posts
-
How Was The First Industrial Revolution Different From The Second
Apr 06, 2025
-
Number Of Atoms In Face Centered Cubic
Apr 06, 2025
-
Two Reactions Between A Grignard Reagent And A Carbonyl Compound
Apr 06, 2025
-
The Hawthorne Studies Concluded That Worker Motivation
Apr 06, 2025
-
Formula For Kinetic Energy Of A Spring
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about What Does The Equivalence Point Of A Titration Indicate . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
