What Is A Primary Standard Chemistry
Muz Play
Apr 02, 2025 · 8 min read
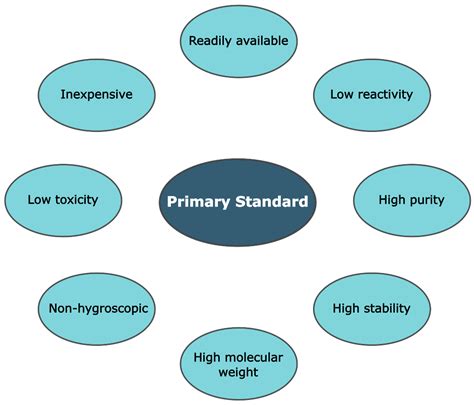
Table of Contents
What is a Primary Standard in Chemistry? A Comprehensive Guide
In the meticulous world of analytical chemistry, accuracy is paramount. Precise measurements are the bedrock of reliable experiments and meaningful results. This is where the concept of a primary standard becomes crucial. Understanding what constitutes a primary standard and its vital role in quantitative analysis is essential for any chemist, student, or researcher. This comprehensive guide will delve into the intricacies of primary standards, exploring their properties, selection criteria, applications, and limitations.
Understanding the Foundation: What is a Primary Standard?
A primary standard in chemistry is a highly purified compound used as a reference material in titrations and other quantitative chemical analyses. It serves as a benchmark against which the concentrations of other solutions (called secondary standards) are determined. Unlike secondary standards, primary standards don't require standardization against another substance. Their purity is so high that their exact concentration can be calculated directly from their mass and molar mass. This makes them indispensable in ensuring the accuracy and reliability of analytical measurements.
Think of a primary standard as the gold standard of chemical measurements. Just as a kilogram standard defines the unit of mass, a primary standard defines the precise concentration of a solution used in a quantitative analysis. The accuracy of the entire analytical process hinges on the purity and stability of the chosen primary standard.
Key Characteristics of a Primary Standard: The Defining Properties
Several critical characteristics distinguish a primary standard from other reagents. A substance qualifies as a primary standard only if it meets stringent criteria. These key properties include:
1. High Purity: The Cornerstone of Accuracy
The most crucial characteristic of a primary standard is its high purity. Impurities directly affect the accuracy of the analysis. The purity should ideally be above 99.9%, with the exact percentage known and documented. Any impurities present must be quantified and considered in calculations. This stringent purity requirement ensures that the mass used directly translates to the number of moles, without significant error introduced by contaminants.
2. Stability: Resisting Degradation
A primary standard must be stable under normal storage conditions. This means it shouldn't decompose, absorb moisture (hygroscopic), react with atmospheric gases (like CO2 or O2), or change its composition over time. Stability ensures that the concentration remains consistent throughout its lifespan, maintaining the integrity of the analysis. Deliquescent substances, which absorb moisture from the air to the point of dissolving, are entirely unsuitable.
3. High Molar Mass: Minimizing Weighing Errors
A high molar mass minimizes the impact of weighing errors on the overall accuracy. Small errors in weighing a substance with a high molar mass will have a smaller relative impact on the final concentration calculation compared to a substance with a low molar mass. This is because a larger mass is needed to achieve a specific molar quantity, thus proportionally reducing the impact of any weighing error.
4. Easy to Dry and Handle: Practicality in Use
The primary standard should be easily dried to a constant weight without decomposition. This is crucial because any residual water can significantly affect the calculated concentration. Furthermore, it should be non-hygroscopic to avoid moisture uptake during weighing. Ease of handling ensures that the weighing process itself does not introduce errors, such as spillage or loss of material.
5. Readily Available: Accessibility for Widespread Use
While high purity is paramount, the primary standard should also be readily available commercially at a reasonable cost. Availability ensures that researchers and analysts can consistently access this essential material without facing significant logistical or financial constraints. Accessibility fosters reproducibility and allows for widespread adoption of analytical methods.
6. Reacts Rapidly and Stoichiometrically: Efficient and Predictable Reactions
In titrations, the primary standard should react rapidly and stoichiometrically with the analyte. Rapid reactions ensure a quick and efficient analysis. Stoichiometric reactions guarantee a predictable and easily calculable mole ratio between the primary standard and the analyte. This ensures a clear and direct relationship between the mass of the primary standard and the concentration of the unknown solution.
Examples of Commonly Used Primary Standards: Practical Applications
Several compounds effectively serve as primary standards, each tailored to specific analytical needs. Here are some common examples:
-
Potassium Hydrogen Phthalate (KHP): A widely used primary standard for acid-base titrations. Its high purity, stability, and ease of handling make it ideal for standardizing strong bases like sodium hydroxide (NaOH). The reaction with a base is well-defined and proceeds readily.
-
Sodium Carbonate (Na₂CO₃): Another excellent choice for acid-base titrations, particularly for standardizing strong acids like hydrochloric acid (HCl). It exhibits high purity and stability when properly dried.
-
Potassium Dichromate (K₂Cr₂O₇): Frequently used in redox titrations. Its high purity, stability, and predictable reaction with reducing agents make it a reliable primary standard for standardizing solutions containing iron(II) or other reducing agents. Its intense orange color can be used to visually determine the endpoint of the titration.
-
Sodium Chloride (NaCl): Used for the standardization of silver nitrate (AgNO₃) solutions in argentometric titrations (precipitation titrations). Its high purity and relatively low cost make it a preferred choice.
-
Sulfamic Acid (NH₂SO₃H): A solid acid often used in acid-base titrations. It is less hygroscopic than many other solid acids, offering improved stability and easier handling.
-
Benzoic Acid (C₇H₆O₂): Another solid acid used as a primary standard. It is relatively non-hygroscopic and possesses high stability, making it suitable for standardizing strong bases.
Preparing a Primary Standard Solution: A Step-by-Step Guide
The preparation of a primary standard solution requires meticulous attention to detail to minimize errors and ensure accuracy. Here’s a general procedure:
-
Drying: Carefully dry the primary standard to a constant weight in a desiccator or oven at a suitable temperature, depending on the specific compound's properties. Avoid overheating, which could cause decomposition.
-
Weighing: Accurately weigh the required mass of the dried primary standard using an analytical balance. Record the exact weight with appropriate significant figures. The required mass depends on the desired concentration and volume of the standard solution.
-
Dissolution: Carefully dissolve the weighed primary standard in a suitable solvent, usually distilled or deionized water. Ensure complete dissolution before proceeding further. Use a volumetric flask to ensure accurate dilution.
-
Dilution: Quantitatively transfer the dissolved primary standard into a volumetric flask of the desired volume. Rinse the weighing vessel several times with solvent, adding the rinsings to the flask to ensure all the primary standard is transferred. Fill the flask up to the calibration mark, ensuring the meniscus is precisely at the mark.
-
Mixing: Stopper the flask and thoroughly mix the solution by inverting it several times to ensure homogeneity.
Primary Standards vs. Secondary Standards: Key Differences
It’s crucial to differentiate between primary and secondary standards. A secondary standard is a solution whose concentration has been determined by comparison to a primary standard. The process of using a primary standard to determine the concentration of a secondary standard is known as standardization. Secondary standards are more convenient for routine analysis as they are typically already in solution form. However, their accuracy directly depends on the accuracy of the primary standard used in their preparation.
| Feature | Primary Standard | Secondary Standard |
|---|---|---|
| Purity | Very high (≥99.9%) | Lower purity |
| Stability | Highly stable under normal storage conditions | May be less stable; requires frequent standardization |
| Standardization | Not required; concentration calculated directly | Requires standardization against a primary standard |
| Availability | May be less readily available | Often more readily available |
| Cost | Usually more expensive | Typically less expensive |
Limitations of Primary Standards: Addressing Potential Challenges
While primary standards offer high accuracy, certain limitations exist:
-
Cost: Some primary standards, especially those with extremely high purity requirements, can be expensive. This can be a limiting factor, particularly for large-scale analyses.
-
Availability: While many primary standards are readily available, some specialized or less common compounds may be difficult to source, potentially impacting the feasibility of specific analyses.
-
Hygroscopy: Even some commonly used primary standards can be slightly hygroscopic, requiring meticulous handling and storage to minimize errors from moisture uptake.
-
Reactivity: Some primary standards might react slowly with certain analytes, potentially increasing the time required for analysis and introducing errors if the reaction is not complete.
-
Toxicity: Certain primary standards possess toxicity, requiring careful handling and disposal procedures to ensure the safety of personnel and the environment.
Conclusion: The Indispensable Role of Primary Standards in Analytical Chemistry
Primary standards are foundational to accurate and reliable quantitative analysis. Their high purity, stability, and other defining characteristics make them indispensable for standardizing solutions and ensuring the overall accuracy of chemical measurements. Understanding their properties, selection criteria, preparation methods, and limitations is crucial for any chemist striving for precision and reproducibility in their work. While limitations exist, the value of primary standards in maintaining the integrity of analytical chemistry remains irrefutable. The careful selection and precise use of primary standards are fundamental to achieving accurate and reliable results in various chemical applications, from routine laboratory work to advanced scientific research.
Latest Posts
Latest Posts
-
Light Amplification By The Stimulated Emission Of Radiation
Apr 03, 2025
-
Octet Rule Violation Vs Wrong Electron Total
Apr 03, 2025
-
What Are The Three Points Of Cell Theory
Apr 03, 2025
-
How Many Atoms Are In A Simple Cubic Unit Cell
Apr 03, 2025
-
Boiling Point Is A Chemical Property
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is A Primary Standard Chemistry . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
