What Is Steric Hindrance In Organic Chemistry
Muz Play
Apr 04, 2025 · 6 min read
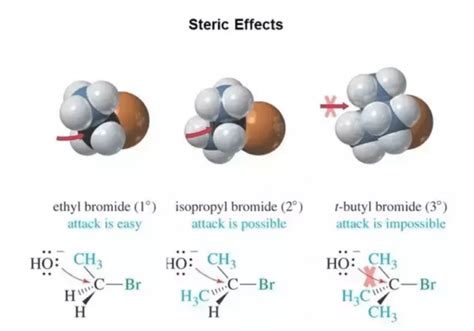
Table of Contents
What is Steric Hindrance in Organic Chemistry? A Deep Dive
Steric hindrance, a cornerstone concept in organic chemistry, profoundly influences the reactivity, stability, and overall behavior of molecules. Understanding steric hindrance is crucial for predicting reaction outcomes, designing synthetic strategies, and interpreting experimental observations. This comprehensive guide will delve into the intricacies of steric hindrance, exploring its definition, causes, consequences, and applications across various organic chemical reactions and concepts.
Defining Steric Hindrance: The Space Crunch in Molecules
Steric hindrance, also known as steric effect, refers to the reduction in reactivity or rate of a reaction caused by the physical bulk of neighboring atoms or groups. Essentially, it's a spatial constraint – large substituents prevent other atoms or molecules from approaching a reactive site, thereby hindering or slowing down the reaction. Think of it as a crowded dance floor: the more people (substituents) there are, the harder it becomes for others to move and interact.
Key Players in Steric Hindrance:
- Substituents: These are the atoms or groups attached to a central atom (often carbon) that cause steric hindrance. Bulky substituents like tert-butyl (t-Bu), phenyl (Ph), and isopropyl (i-Pr) are notorious for their steric bulk.
- Reactive Site: This is the atom or group in the molecule that participates in the chemical reaction. Steric hindrance impedes access to this site.
- Reactant: The molecule or atom that interacts with the reactive site. The size and shape of the reactant play a crucial role in determining the degree of steric hindrance.
Causes of Steric Hindrance: The Bulky Neighbors
The primary cause of steric hindrance is the size and shape of substituents surrounding the reactive site. Large substituents occupy significant space, creating a physical barrier that impedes the approach of reactants.
Factors influencing steric hindrance:
- Size of Substituents: Larger substituents naturally create more steric hindrance. The van der Waals radii of the atoms involved provide a good measure of their spatial requirements.
- Shape of Substituents: The shape of substituents also matters. Linear substituents might have less steric hindrance compared to branched ones, even if they have similar molecular weights. For example, a n-propyl group will exert less steric hindrance than an iso-propyl group.
- Conformational Effects: The specific three-dimensional arrangement (conformation) of a molecule can significantly influence the extent of steric hindrance. Certain conformations might minimize steric interactions, while others exacerbate them.
- Proximity of Substituents: The closer the bulky substituents are to the reactive site, the greater the steric hindrance.
Consequences of Steric Hindrance: Reactivity and Stability
Steric hindrance manifests in several observable ways:
- Reduced Reaction Rates: The most prominent consequence is a decrease in the rate of a reaction. The bulky substituents physically block the approach of reactants, reducing the frequency of successful collisions leading to reaction.
- Altered Reaction Pathways: Steric hindrance can alter the preferred reaction pathway. A reaction might favor a less sterically hindered pathway even if it's less thermodynamically favorable.
- Changes in Reaction Selectivity: In reactions with multiple possible sites of reaction, steric hindrance can direct the reaction to the less hindered site. This is highly valuable in organic synthesis for achieving regioselectivity.
- Increased Stability of Certain Conformations: Steric hindrance can stabilize certain conformations by minimizing repulsive interactions between substituents. This is often observed in cyclic molecules.
- Influence on Physical Properties: Steric hindrance can affect the melting point, boiling point, and solubility of a compound. Bulky substituents can hinder close packing in the solid or liquid state, influencing intermolecular interactions.
Examples of Steric Hindrance in Action
Steric hindrance plays a crucial role in numerous organic chemical reactions and concepts:
1. SN1 vs. SN2 Reactions:
- SN2 reactions: These are bimolecular nucleophilic substitution reactions where the nucleophile attacks the carbon atom from the backside simultaneously with the departure of the leaving group. Bulky substituents around the carbon atom hinder this backside attack, drastically slowing down the reaction rate. Therefore, SN2 reactions are significantly affected by steric hindrance.
- SN1 reactions: These are unimolecular nucleophilic substitution reactions where the leaving group departs first, forming a carbocation intermediate. The stability of the carbocation intermediate is crucial in SN1 reactions. Bulky substituents can stabilize the carbocation by hyperconjugation, but excessive steric hindrance can destabilize the carbocation by making it difficult for the solvent molecules to solvate it.
2. Electrophilic Aromatic Substitution:
In electrophilic aromatic substitution reactions, the incoming electrophile attacks the aromatic ring. Bulky substituents on the ring can hinder this attack, affecting both the rate and the regioselectivity of the reaction. Ortho and para positions are more affected than the meta position because they are directly adjacent to the substituent.
3. Esterification and Amide Formation:
These reactions involve the attack of a nucleophile (alcohol or amine) on a carbonyl group. Bulky substituents around the carbonyl group can significantly hinder the nucleophilic attack, slowing down the reaction rate.
4. Grignard Reactions:
Grignard reagents are organomagnesium compounds that react with carbonyl groups. Bulky substituents on the carbonyl group can hinder the addition of the Grignard reagent, affecting the reaction yield and selectivity.
5. Conformational Analysis:
Steric hindrance plays a crucial role in determining the preferred conformation of molecules. For instance, in butane, the anti-conformation is more stable than the gauche conformation due to less steric interaction between the methyl groups. Similarly, cyclohexane adopts a chair conformation to minimize steric interactions between axial substituents.
Overcoming Steric Hindrance: Strategies in Organic Synthesis
Organic chemists often encounter steric hindrance as a challenge in their synthetic endeavors. Several strategies can be employed to mitigate or overcome these obstacles:
- Choosing appropriate reaction conditions: Using higher temperatures, longer reaction times, or employing stronger nucleophiles or electrophiles can sometimes overcome steric hindrance.
- Using different solvents: The choice of solvent can affect the solvation of reactants and intermediates, influencing steric effects.
- Employing catalysts: Catalysts can activate the reactants or stabilize intermediates, facilitating reactions that would otherwise be hampered by steric hindrance.
- Protecting group strategies: Protecting groups can temporarily mask reactive sites, preventing unwanted reactions and allowing for selective transformations.
- Designing alternative synthetic routes: Sometimes, circumventing steric hindrance involves developing an entirely new synthetic route that avoids the sterically hindered steps.
Conclusion: The Pervasive Influence of Steric Hindrance
Steric hindrance is an omnipresent factor in organic chemistry, exerting a profound influence on the reactivity, selectivity, and stability of molecules. Understanding the principles of steric hindrance is vital for designing efficient synthetic strategies, predicting reaction outcomes, and interpreting experimental results. By comprehending the causes and consequences of steric hindrance, organic chemists can effectively navigate the complexities of molecular interactions and unlock new possibilities in the synthesis of novel compounds and materials. The exploration of steric hindrance continues to be a vibrant area of research, pushing the boundaries of our understanding of molecular behavior and shaping the future of organic chemistry.
Latest Posts
Latest Posts
-
Why Is Oil Necessary When Using The 100x Objective
Apr 04, 2025
-
Where Does Reduction Occur In An Electrolytic Cell
Apr 04, 2025
-
Analytical Balance Vs Top Loading Balance
Apr 04, 2025
-
Are Hydrogen Bonds Weak Or Strong
Apr 04, 2025
-
Is Brittle A Metal Nonmetal Or Metalloid
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is Steric Hindrance In Organic Chemistry . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
