What Is The Difference Between Empirical Formula And Molecular Formula
Muz Play
Mar 30, 2025 · 6 min read
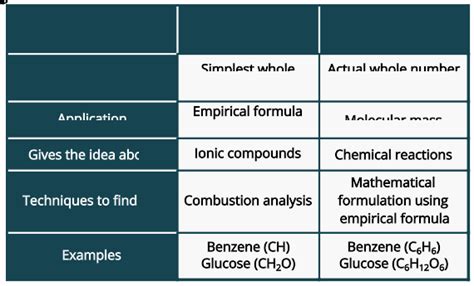
Table of Contents
- What Is The Difference Between Empirical Formula And Molecular Formula
- Table of Contents
- What's the Difference Between Empirical Formula and Molecular Formula? A Comprehensive Guide
- Defining Empirical Formula and Molecular Formula
- Empirical Formula: The Simplest Ratio
- Molecular Formula: The True Composition
- The Relationship Between Empirical and Molecular Formulas
- Determining Empirical Formulas: A Step-by-Step Guide
- Determining Molecular Formulas: Beyond the Empirical Formula
- Advanced Concepts and Applications
- Isomers: Same Formula, Different Structure
- Determining Empirical and Molecular Formulas from Experimental Data
- Applications in Different Fields
- Conclusion: A Foundation for Chemical Understanding
- Latest Posts
- Latest Posts
- Related Post
What's the Difference Between Empirical Formula and Molecular Formula? A Comprehensive Guide
Understanding the fundamental concepts of chemistry, such as empirical and molecular formulas, is crucial for anyone navigating the world of chemical compounds. While these terms might seem similar at first glance, they represent distinct aspects of a molecule's composition. This comprehensive guide will delve into the differences between empirical and molecular formulas, exploring their definitions, applications, and how to determine them. We'll also cover advanced concepts and provide practical examples to solidify your understanding.
Defining Empirical Formula and Molecular Formula
Let's start by clearly defining each term.
Empirical Formula: The Simplest Ratio
The empirical formula represents the simplest whole-number ratio of atoms of each element present in a compound. It shows the relative proportions of different elements, but not necessarily the actual number of atoms in a molecule. Think of it as a simplified blueprint. It doesn't tell you the exact number of bricks, but it shows the ratio of different types of bricks used in a building.
Key Characteristics of an Empirical Formula:
- Simplest ratio: Always expresses the ratio of atoms in their smallest whole-number form.
- Relative proportions: Indicates the relative abundance of each element in the compound.
- Not necessarily the true formula: Might not reflect the actual number of atoms in a molecule.
Molecular Formula: The True Composition
The molecular formula, on the other hand, represents the actual number of atoms of each element present in one molecule of a compound. This formula provides the complete picture of the molecule's composition. Returning to our building analogy, it's like having a complete architectural plan detailing the exact number of each type of brick.
Key Characteristics of a Molecular Formula:
- Actual number of atoms: Accurately reflects the number of each atom type in a single molecule.
- Complete composition: Gives a precise representation of the molecule's structure.
- Multiple possibilities: A single empirical formula can correspond to multiple molecular formulas.
The Relationship Between Empirical and Molecular Formulas
The connection between empirical and molecular formulas is straightforward: the molecular formula is always a whole-number multiple of the empirical formula. This means you can derive the empirical formula from the molecular formula, but you cannot always derive the molecular formula from the empirical formula alone. Additional information, such as the molar mass of the compound, is required.
Mathematical Representation:
Molecular Formula = n x Empirical Formula
Where 'n' is a whole number (1, 2, 3, and so on).
Determining Empirical Formulas: A Step-by-Step Guide
Determining the empirical formula involves several key steps:
-
Determine the mass of each element: This is usually given in the problem statement, or it can be calculated from experimental data, such as combustion analysis. Express the masses in grams.
-
Convert grams to moles: Use the molar mass of each element (found on the periodic table) to convert the mass of each element from grams to moles. Remember, moles = mass (grams) / molar mass (grams/mole).
-
Find the mole ratio: Divide the number of moles of each element by the smallest number of moles calculated in step 2. This will give you the mole ratio of the elements in the simplest form.
-
Convert to whole numbers: If the mole ratios obtained in step 3 are not whole numbers, multiply all the ratios by the smallest integer that will make them all whole numbers. This ensures the empirical formula is expressed in the simplest whole-number ratio.
-
Write the empirical formula: Use the whole-number ratios from step 4 as subscripts for each element's symbol in the formula.
Example:
Let's say a compound contains 75% carbon and 25% hydrogen by mass. To find its empirical formula:
-
Assume a 100g sample: This simplifies the calculations; 75g is carbon, and 25g is hydrogen.
-
Convert to moles:
- Moles of Carbon: 75g / 12.01 g/mol (molar mass of C) ≈ 6.24 moles
- Moles of Hydrogen: 25g / 1.01 g/mol (molar mass of H) ≈ 24.75 moles
-
Find the mole ratio: Divide by the smallest number of moles (6.24 moles):
- Carbon: 6.24 moles / 6.24 moles = 1
- Hydrogen: 24.75 moles / 6.24 moles ≈ 3.97 ≈ 4 (rounding to the nearest whole number is acceptable in this context)
-
Write the empirical formula: CH₄ (methane)
Determining Molecular Formulas: Beyond the Empirical Formula
To find the molecular formula, you need the empirical formula and the molar mass of the compound.
-
Calculate the empirical formula mass: Add up the molar masses of the atoms in the empirical formula.
-
Determine the whole number multiple (n): Divide the molar mass of the compound (given or experimentally determined) by the empirical formula mass. This gives you 'n'.
-
Determine the molecular formula: Multiply the subscripts in the empirical formula by 'n' to obtain the molecular formula.
Example:
Let's assume the empirical formula of a compound is CH₂O, and its molar mass is 180 g/mol.
-
Empirical formula mass: 12.01 g/mol (C) + 2(1.01 g/mol) (H) + 16.00 g/mol (O) = 30.03 g/mol
-
Determine n: 180 g/mol / 30.03 g/mol ≈ 6
-
Molecular formula: (CH₂O)₆ = C₆H₁₂O₆ (glucose)
Advanced Concepts and Applications
Isomers: Same Formula, Different Structure
Isomers are molecules with the same molecular formula but different structural arrangements. This highlights that the molecular formula alone doesn't fully define a compound's properties. For example, glucose, fructose, and galactose all have the molecular formula C₆H₁₂O₆, but they are distinct sugars with different properties.
Determining Empirical and Molecular Formulas from Experimental Data
Techniques such as combustion analysis (for organic compounds) and various spectroscopic methods provide crucial data for determining the composition of unknown compounds. These methods allow scientists to quantify the amounts of different elements present, which is then used to calculate the empirical and molecular formulas.
Applications in Different Fields
The determination of empirical and molecular formulas is fundamental across many scientific fields:
- Organic Chemistry: Essential for characterizing newly synthesized compounds and identifying unknown organic molecules.
- Biochemistry: Crucial for understanding the composition and properties of biomolecules like proteins, carbohydrates, and lipids.
- Material Science: Used for characterizing new materials and understanding their properties.
- Environmental Science: Used to analyze pollutants and understand their chemical composition.
Conclusion: A Foundation for Chemical Understanding
Understanding the difference between empirical and molecular formulas is crucial for comprehending the composition of chemical compounds. While the empirical formula provides a simplified ratio of elements, the molecular formula accurately reflects the actual number of atoms in a molecule. The ability to determine both formulas from experimental data is a fundamental skill in chemistry and is vital for advancements across a wide range of scientific disciplines. Mastering these concepts forms a solid foundation for further exploration of chemical principles and applications.
Latest Posts
Latest Posts
-
Counting Atoms In One Gram Answer Key
Apr 03, 2025
-
Is Coke A Base Or Acid
Apr 03, 2025
-
What Are The Horizontal Columns On The Periodic Table Called
Apr 03, 2025
-
A Person Who Is Heterozygous For The Cystic Fibrosis Allele
Apr 03, 2025
-
Derivation Of Van Der Waals Equation
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Difference Between Empirical Formula And Molecular Formula . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
