What Is The Electron Configuration Of Neon
Muz Play
Apr 02, 2025 · 6 min read
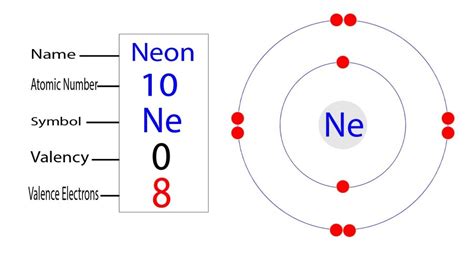
Table of Contents
What is the Electron Configuration of Neon? A Deep Dive into Atomic Structure
Neon, the vibrant gas that illuminates our signs and adds a glow to our everyday lives, holds a fascinating secret within its seemingly simple atomic structure: its electron configuration. Understanding this configuration unlocks a deeper appreciation for neon's properties and its place within the periodic table. This comprehensive guide will delve into the electron configuration of neon, exploring its implications for chemical behavior, spectroscopic properties, and its role in various applications.
Understanding Electron Configurations
Before we dive into the specifics of neon's configuration, let's establish a foundational understanding of what electron configuration means. An electron configuration is a symbolic notation that describes the arrangement of electrons in the various energy levels and sublevels within an atom. This arrangement dictates an atom's chemical properties, its reactivity, and its interaction with other atoms. It's crucial to remember that electrons occupy specific orbitals within these energy levels, following the principles of quantum mechanics.
Energy Levels and Sublevels
Electrons don't just randomly float around the nucleus. They exist in distinct energy levels, often visualized as shells surrounding the nucleus. Each energy level has a maximum number of electrons it can hold, determined by the formula 2n², where 'n' is the principal quantum number representing the energy level (n=1, 2, 3, etc.).
Within each energy level are sublevels, denoted by the letters s, p, d, and f. These sublevels represent different shapes and orientations of electron orbitals. The s sublevel can hold a maximum of 2 electrons, the p sublevel can hold up to 6, the d sublevel up to 10, and the f sublevel up to 14.
The Aufbau Principle and Hund's Rule
The filling of electrons into these orbitals follows specific rules:
-
The Aufbau Principle: Electrons first fill the lowest energy levels and sublevels before moving to higher ones. This is a fundamental principle guiding electron configuration.
-
Hund's Rule: Within a sublevel, electrons will individually occupy each orbital before pairing up in the same orbital. This minimizes electron-electron repulsion and creates a more stable configuration.
-
Pauli Exclusion Principle: No two electrons in an atom can have the same set of four quantum numbers (principal quantum number, azimuthal quantum number, magnetic quantum number, and spin quantum number). This principle dictates that each orbital can hold a maximum of two electrons with opposite spins.
Neon's Electron Configuration: 1s²2s²2p⁶
Now, let's finally uncover the electron configuration of neon (Ne), an element with an atomic number of 10. This means it possesses 10 protons in its nucleus and, in its neutral state, 10 electrons surrounding it.
Applying the Aufbau principle and Hund's rule, we can determine the electron configuration as follows:
1s²2s²2p⁶
Let's break this down:
-
1s²: The first energy level (n=1) contains the s sublevel, which holds two electrons.
-
2s²: The second energy level (n=2) also contains an s sublevel, holding another two electrons.
-
2p⁶: The second energy level (n=2) also contains a p sublevel, which can hold up to six electrons. In neon, this sublevel is completely filled with six electrons.
The Significance of a Filled Valence Shell
The most significant aspect of neon's electron configuration is the completely filled 2s and 2p sublevels in its outermost shell, also known as the valence shell. Having a complete valence shell makes neon exceptionally stable and unreactive. This is because achieving a full valence shell (often referred to as a noble gas configuration) minimizes the atom's energy and makes it highly resistant to forming chemical bonds with other atoms. This inertness is a defining characteristic of noble gases, including neon.
Neon's Spectroscopic Properties and Applications
Neon's complete valence shell directly influences its spectroscopic properties. When excited by an electrical discharge, neon's electrons jump to higher energy levels. When these excited electrons return to their ground state, they release energy in the form of photons, producing the characteristic reddish-orange glow associated with neon lights. The specific wavelengths of light emitted are a direct consequence of the energy differences between the electron energy levels, determined by its electron configuration.
This unique spectroscopic property underpins many of neon's applications, including:
-
Neon lighting: The most well-known application, leveraging neon's vibrant glow for signage and decorative lighting.
-
Laser technology: Neon's specific energy level transitions are exploited in certain types of lasers.
-
Helium-neon lasers: These lasers, using a mixture of helium and neon, produce highly coherent and monochromatic light used in various scientific instruments and applications.
-
Cryogenics: Though less prevalent than helium, neon can be used as a cryogenic refrigerant, particularly at temperatures slightly above those achieved using liquid helium.
Comparison with Other Elements
Comparing neon's electron configuration to neighboring elements on the periodic table provides further insight into its unique chemical behavior. For example, fluorine (atomic number 9) has one less electron than neon and hence one electron less in the 2p sublevel. This electron deficiency makes fluorine extremely reactive, readily accepting an electron to achieve the stable neon configuration. Conversely, sodium (atomic number 11) has one more electron than neon. This extra electron resides in the next principal energy level (3s), making sodium willing to lose this electron and gain a noble gas configuration similar to neon. This highlights how the electron configuration dictates an element's reactivity and its position within the periodic table.
Advanced Concepts: Orbital Diagrams and Quantum Numbers
A more detailed representation of neon's electron configuration involves orbital diagrams and quantum numbers. An orbital diagram shows each individual orbital within a sublevel and the arrangement of electrons within them. For neon, this would show two electrons in the 1s orbital, two electrons in the 2s orbital, and two electrons each in the three 2p orbitals (2px, 2py, 2pz), with each orbital containing a maximum of two electrons with opposite spins.
Each electron is also described by a set of four quantum numbers:
-
Principal Quantum Number (n): Represents the energy level. For neon's electrons, n = 1 or 2.
-
Azimuthal Quantum Number (l): Represents the sublevel (s, p, d, f). For neon, l = 0 (s) or 1 (p).
-
Magnetic Quantum Number (ml): Represents the specific orbital within a sublevel. For neon's p electrons, ml = -1, 0, or +1.
-
Spin Quantum Number (ms): Represents the electron spin (+1/2 or -1/2).
Conclusion: Neon's Simple Configuration, Profound Implications
Neon's electron configuration, although seemingly simple at first glance, is fundamental to understanding its properties and applications. The completely filled valence shell dictates its inert nature, its spectroscopic properties, and consequently, its numerous uses in lighting, laser technology, and even cryogenics. By understanding neon's electron configuration, we gain a deeper appreciation for the intricate interplay between atomic structure and macroscopic properties, demonstrating the predictive power of quantum mechanics in explaining the behavior of matter. This knowledge underpins our understanding of chemistry and its applications in various fields of science and technology. From the vibrant glow of neon signs to the precision of laser technology, neon’s simple yet profound electron configuration serves as a testament to the power of fundamental scientific principles.
Latest Posts
Latest Posts
-
Where Is The Energy Stored In Glucose
Apr 03, 2025
-
Light Amplification By The Stimulated Emission Of Radiation
Apr 03, 2025
-
Octet Rule Violation Vs Wrong Electron Total
Apr 03, 2025
-
What Are The Three Points Of Cell Theory
Apr 03, 2025
-
How Many Atoms Are In A Simple Cubic Unit Cell
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Electron Configuration Of Neon . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
