What Is The Mass Of Neutron In Amu
Muz Play
Apr 05, 2025 · 5 min read
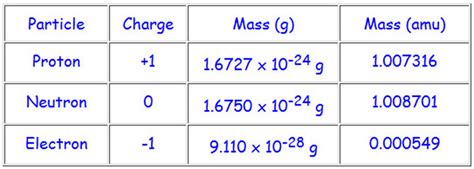
Table of Contents
What is the Mass of a Neutron in amu? A Deep Dive into Neutron Properties
The neutron, a fundamental subatomic particle, plays a crucial role in the structure and behavior of atomic nuclei. Understanding its properties, particularly its mass, is fundamental to comprehending nuclear physics, chemistry, and various applications across scientific disciplines. This comprehensive article delves into the mass of a neutron, exploring its measurement, significance, and applications.
Understanding Atomic Mass Units (amu)
Before diving into the neutron's mass, it's crucial to understand the unit of measurement: the atomic mass unit (amu), also known as the dalton (Da). One amu is defined as 1/12 the mass of a single, unbound, neutral atom of carbon-12. This standard provides a consistent and convenient way to express the masses of atoms and subatomic particles, avoiding the use of extremely small numbers. The amu is closely related to the unified atomic mass unit (u), with 1 amu ≈ 1 u.
The Mass of a Neutron in amu: Precise Measurement
The mass of a neutron isn't a simple, easily obtained number. It requires sophisticated experimental techniques to accurately determine. Through advanced methods like mass spectrometry and nuclear reactions, scientists have determined the neutron mass to a high degree of precision. The currently accepted value is approximately 1.00866491588(49) amu.
This seemingly precise number carries significance. The parentheses around "(49)" indicate the uncertainty in the last two digits. This uncertainty reflects the inherent limitations of measurement and the complexity of determining the mass of a single neutron. Despite the small uncertainty, this precision is crucial for accurate calculations in nuclear physics and related fields.
Methods for Determining Neutron Mass
Several methods contribute to this precise measurement. One common method involves analyzing nuclear reactions, specifically those involving neutron capture or emission. By meticulously measuring the energies and momenta of the particles involved in the reaction, scientists can infer the mass of the neutron through conservation laws.
Mass spectrometry, another crucial technique, measures the mass-to-charge ratio of ions. Although it directly measures ions and not free neutrons, sophisticated methods employing neutron-induced reactions can indirectly determine the neutron mass with high precision. The advancement of these techniques constantly refines our understanding and increases the accuracy of the neutron's mass measurement.
Significance of Neutron Mass in Nuclear Physics
The precise mass of the neutron is not merely a numerical curiosity; it's a cornerstone of nuclear physics. This mass, along with the masses of protons and electrons, determines several key nuclear properties:
1. Nuclear Binding Energy
The mass of a nucleus is always slightly less than the sum of the masses of its constituent protons and neutrons. This "missing" mass is converted into energy, known as the nuclear binding energy, which holds the nucleus together. Einstein's famous equation, E=mc², directly relates this mass defect to the binding energy. The neutron's mass is a critical component in calculating this binding energy, essential for understanding nuclear stability and radioactive decay.
2. Nuclear Reactions and Stability
The neutron-to-proton ratio within a nucleus significantly influences its stability. Neutrons play a crucial role in overcoming the electrostatic repulsion between protons. Knowing the neutron mass allows physicists to predict the stability of different isotopes and model various nuclear reactions, such as fission and fusion.
3. Isotope Abundance and Properties
The mass of a neutron directly impacts the mass of various isotopes of an element. Isotopes are atoms of the same element with differing numbers of neutrons. The relative abundance of different isotopes within an element is determined by various factors, including the stability of the nucleus, which is directly related to the neutron's mass and its interaction with protons.
4. Nuclear Models and Theories
Accurate neutron mass measurements are vital for refining and testing theoretical models of the nucleus. These models attempt to describe the complex interactions between protons and neutrons within the nucleus, and accurate mass data provides crucial constraints and validation for these theoretical frameworks.
Applications Beyond Nuclear Physics
The significance of the neutron's mass extends beyond the realm of pure nuclear physics. Its precise determination has implications in:
1. Materials Science
Neutron scattering techniques, which utilize beams of neutrons, are widely used to probe the atomic structure and dynamics of materials. Understanding the neutron's mass is essential for interpreting scattering data and gaining insights into the properties of materials, including their crystalline structure, magnetic properties, and diffusion processes.
2. Chemistry
Neutron activation analysis is a powerful analytical technique that utilizes neutrons to induce radioactivity in samples. By measuring the emitted radiation, scientists can identify and quantify trace elements within materials. Again, the neutron's mass is implicit in the calculations used to interpret the results.
3. Medicine
Neutron radiation, particularly in the form of neutron therapy, is used in cancer treatment. While not directly using the mass value, understanding neutron interactions with biological tissue requires knowledge of its properties, including its mass.
4. Astrophysics and Cosmology
Neutron stars, incredibly dense objects formed from the collapsed cores of massive stars, are composed primarily of neutrons. Their properties, including their mass and density, are related to the fundamental properties of the neutron, providing crucial insights into stellar evolution and the structure of the universe.
Further Refinements and Future Research
The quest for ever-increasing precision in the neutron mass measurement continues. Ongoing research focuses on improving experimental techniques and reducing uncertainties further. These efforts contribute to a more nuanced understanding of nuclear physics and its many applications. The development of more sophisticated detectors and advanced computational methods will undoubtedly lead to even more precise measurements in the future.
Conclusion: The Neutron's Mass – A Cornerstone of Scientific Understanding
The mass of a neutron in amu, a seemingly small detail, underpins a vast landscape of scientific knowledge and technological applications. From understanding the fundamental structure of matter to developing advanced materials and medical therapies, the precise determination of the neutron's mass has profound implications. The ongoing refinement of its measured value continues to push the boundaries of our understanding of the universe and the physical laws that govern it. The pursuit of ever-greater accuracy in this seemingly simple measurement underscores the fundamental importance of precise measurement in advancing scientific understanding and technological innovation. The continued exploration of neutron properties will undoubtedly uncover further insights into the intricate world of nuclear physics and its far-reaching consequences.
Latest Posts
Latest Posts
-
How To Find Center Of Data
Apr 06, 2025
-
Type Of Molecule With An Uneven Distribution Of Electrons
Apr 06, 2025
-
Differential Rate Law Vs Integrated Rate Law
Apr 06, 2025
-
How Do You Test An Equation For Symmetry
Apr 06, 2025
-
Circuit Diagram With 3 Resistors In Series
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about What Is The Mass Of Neutron In Amu . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
