What Is The Relationship Between Energy And Frequency
Muz Play
Mar 30, 2025 · 6 min read
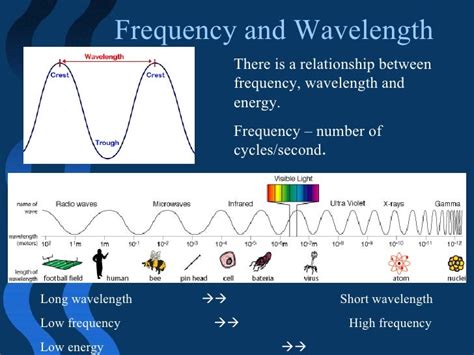
Table of Contents
- What Is The Relationship Between Energy And Frequency
- Table of Contents
- What is the Relationship Between Energy and Frequency?
- The Foundation: Planck's Equation
- Implications of Planck's Equation
- Exploring Different Forms of Energy and Frequency
- 1. Light and Electromagnetic Radiation:
- 2. Sound Waves:
- 3. Atomic and Molecular Vibrations:
- 4. Nuclear Processes:
- Applications of the Energy-Frequency Relationship
- 1. Spectroscopy:
- 2. Medical Imaging:
- 3. Telecommunications:
- 4. Materials Science:
- 5. Astrophysics:
- Beyond Planck's Equation: More Complex Relationships
- Conclusion: A Universal Principle
- Latest Posts
- Latest Posts
- Related Post
What is the Relationship Between Energy and Frequency?
The relationship between energy and frequency is fundamental to our understanding of the universe, spanning from the smallest subatomic particles to the largest cosmic structures. This relationship, elegantly expressed by Planck's equation, reveals a profound connection between the seemingly disparate concepts of energy and vibration. Understanding this connection is crucial in various fields, from quantum mechanics and astrophysics to electronics and materials science. This article delves deep into the intricacies of this relationship, exploring its implications and applications across diverse scientific disciplines.
The Foundation: Planck's Equation
The cornerstone of understanding the energy-frequency relationship lies in Planck's equation: E = hf, where:
- E represents energy (typically measured in Joules).
- h is Planck's constant, a fundamental physical constant approximately equal to 6.626 x 10<sup>-34</sup> Joule-seconds.
- f represents frequency (typically measured in Hertz, or cycles per second).
This simple yet powerful equation reveals a direct proportionality: energy is directly proportional to frequency. This means that as the frequency of a wave (be it light, sound, or any other form of electromagnetic radiation) increases, its energy increases proportionally. Conversely, a decrease in frequency results in a decrease in energy.
Implications of Planck's Equation
Planck's equation has far-reaching consequences, revolutionizing our understanding of the nature of energy and matter. Some key implications include:
-
Quantization of Energy: Planck's equation highlights the quantized nature of energy. Energy is not continuous but exists in discrete packets called quanta. The energy of a quantum is directly related to its frequency. This concept is central to quantum mechanics, explaining phenomena that classical physics could not account for.
-
The Particle-Wave Duality: Planck's equation contributes significantly to the understanding of the wave-particle duality of matter. Particles, like electrons and photons, exhibit both wave-like and particle-like properties. The frequency of their associated wave determines their energy.
-
Electromagnetic Spectrum: The electromagnetic spectrum, ranging from radio waves to gamma rays, is perfectly explained by Planck's equation. Higher-frequency electromagnetic radiation, such as gamma rays, carries significantly higher energy than lower-frequency radiation, like radio waves. This explains the differing effects of these types of radiation on matter.
Exploring Different Forms of Energy and Frequency
The energy-frequency relationship isn't limited to electromagnetic radiation. It extends to various other forms of energy and oscillatory phenomena:
1. Light and Electromagnetic Radiation:
Light, a form of electromagnetic radiation, demonstrates the energy-frequency relationship vividly. Visible light, a small portion of the electromagnetic spectrum, has frequencies corresponding to specific colors. Violet light, with its higher frequency, has higher energy than red light, with its lower frequency. This explains why ultraviolet (UV) light, with a higher frequency than visible light, can cause sunburns, while infrared (IR) light, with a lower frequency, provides warmth.
2. Sound Waves:
Sound waves, mechanical vibrations traveling through a medium, also exhibit this relationship. Higher-frequency sound waves, such as those perceived as high-pitched sounds, carry more energy than lower-frequency sound waves, perceived as low-pitched sounds. This explains why high-intensity sounds can be damaging to hearing; the higher energy associated with higher frequencies can cause physical damage to the ear's delicate structures.
3. Atomic and Molecular Vibrations:
Atoms and molecules possess vibrational energy. The frequency of these vibrations depends on the mass of the atoms and the strength of the bonds between them. Higher-frequency vibrations indicate higher energy. In spectroscopy, analyzing the absorption and emission of energy by molecules is used to determine the vibrational frequencies and consequently the energy levels of molecules, providing crucial insights into molecular structure and dynamics.
4. Nuclear Processes:
Nuclear processes, such as radioactive decay, also involve energy and frequency relationships. The energy released during radioactive decay is related to the frequency of the emitted radiation (alpha, beta, or gamma). High-energy gamma radiation, for instance, has a very high frequency. Understanding these energy-frequency relationships is crucial in nuclear physics, medical imaging (PET scans), and other applications.
Applications of the Energy-Frequency Relationship
The profound implications of the energy-frequency relationship have led to numerous applications across various scientific and technological fields:
1. Spectroscopy:
Spectroscopy, the study of the interaction between matter and electromagnetic radiation, heavily relies on the energy-frequency relationship. Different molecules and atoms absorb and emit electromagnetic radiation at specific frequencies, providing a unique "fingerprint" that can be used for identification and analysis. Techniques like infrared (IR) spectroscopy, nuclear magnetic resonance (NMR) spectroscopy, and ultraviolet-visible (UV-Vis) spectroscopy use this principle to analyze the composition and structure of materials.
2. Medical Imaging:
Medical imaging techniques like X-rays, CT scans, and PET scans utilize the energy-frequency relationship to create images of the internal structures of the body. X-rays, high-energy electromagnetic radiation, are used to penetrate soft tissues and create images of bones and denser structures. PET scans use the detection of gamma rays emitted from radioactive isotopes to visualize metabolic activity within the body.
3. Telecommunications:
Telecommunications rely heavily on the manipulation and transmission of electromagnetic waves. Different frequencies are assigned to various communication channels (radio, television, mobile phones), each carrying information encoded in the variations of the wave's amplitude and frequency. The energy of the signal is critical for ensuring reliable transmission over long distances.
4. Materials Science:
The energy-frequency relationship is crucial in materials science for understanding the properties of materials. The vibrational frequencies of atoms in a material influence its thermal, electrical, and mechanical properties. Techniques like Raman spectroscopy and inelastic neutron scattering are used to study these vibrational frequencies and understand material behavior.
5. Astrophysics:
In astrophysics, the energy-frequency relationship helps astronomers understand the processes occurring in stars and galaxies. The spectrum of light emitted by celestial objects provides information about their temperature, composition, and motion. Redshift, the stretching of light waves as objects move away from us, results in a decrease in frequency and energy, crucial for determining the expansion rate of the universe.
Beyond Planck's Equation: More Complex Relationships
While Planck's equation provides a fundamental understanding of the energy-frequency relationship, it is a simplification of more complex scenarios. In some cases, other factors influence the relationship:
-
Quantum Field Theory: Quantum field theory provides a more sophisticated framework for understanding the energy-frequency relationship, particularly when dealing with interacting particles and fields. The energy of a particle is not solely determined by its frequency but also by its interactions with other particles and fields.
-
Relativistic Effects: At high energies and velocities, relativistic effects become significant. Einstein's theory of special relativity modifies the energy-frequency relationship, introducing effects such as time dilation and length contraction.
-
Nonlinear Systems: In nonlinear systems, the relationship between energy and frequency is not always linear. The energy of a wave can depend on its amplitude and other factors in a complex manner.
Conclusion: A Universal Principle
The energy-frequency relationship, as epitomized by Planck's equation, is a universal principle with profound implications across various scientific disciplines. From understanding the behavior of subatomic particles to exploring the vastness of the cosmos, this relationship provides a fundamental framework for comprehending the nature of energy and its diverse manifestations. Continued research and advancements in our understanding of this relationship will undoubtedly lead to further innovations and breakthroughs across various fields of science and technology. The inherent link between energy and frequency underscores the interconnectedness of the universe and provides a powerful tool for exploring its complexities. Further research will only deepen our appreciation of this fundamental aspect of the physical world.
Latest Posts
Latest Posts
-
What Is Dielectric Constant Of Water
Apr 03, 2025
-
Cells Are The Basic Structural Units Of Living Organisms Explain
Apr 03, 2025
-
Not A Or B Truth Table
Apr 03, 2025
-
The Phylogenetic Classification Of Bacteria Is Based On
Apr 03, 2025
-
Which Amino Acid Has A Positively Charged R Group
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Relationship Between Energy And Frequency . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
