What Is The Specific Heat Of Silver
Muz Play
Apr 02, 2025 · 7 min read
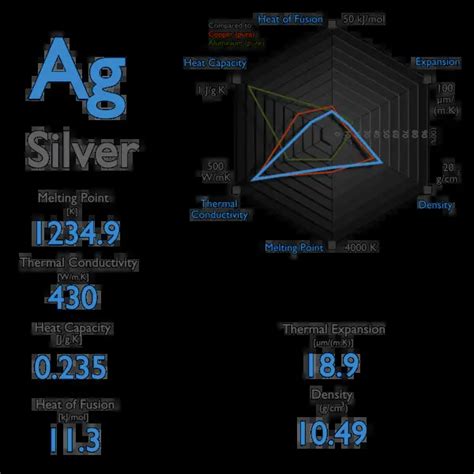
Table of Contents
What is the Specific Heat of Silver? A Deep Dive into Thermal Properties
Silver, a lustrous, white, precious metal known for its excellent conductivity, possesses a fascinating array of physical properties. Among these, its specific heat capacity plays a crucial role in various applications, from industrial processes to scientific research. This comprehensive article delves into the specific heat of silver, exploring its definition, measurement, factors influencing it, and its significance in diverse fields.
Understanding Specific Heat Capacity
Before diving into the specific heat of silver, let's establish a clear understanding of the concept itself. Specific heat capacity, often shortened to specific heat, is the amount of heat energy required to raise the temperature of one unit of mass of a substance by one degree Celsius (or one Kelvin). It's a crucial thermodynamic property that describes a substance's resistance to temperature change. A substance with a high specific heat requires a significant amount of energy to increase its temperature, while a substance with a low specific heat requires less.
The specific heat is typically expressed in units of Joules per kilogram-Kelvin (J/kg·K) or calories per gram-degree Celsius (cal/g·°C). These units reflect the energy input (Joules or calories) needed to change the temperature of a specific mass (kilogram or gram) by a single degree.
The Specific Heat of Silver: A Numerical Value
The specific heat of silver is approximately 235 J/kg·K or 0.056 cal/g·°C. This value signifies that it takes 235 Joules of heat energy to raise the temperature of 1 kilogram of silver by 1 Kelvin (or 1 degree Celsius). Compared to other metals, silver possesses a relatively high specific heat, indicating its ability to absorb significant heat energy without experiencing a large temperature increase.
It's important to note that the specific heat of silver, like any other material, can vary slightly depending on factors such as temperature, pressure, and the purity of the silver sample. The value provided above represents a typical approximation under standard conditions. Highly accurate measurements require precise control of these parameters.
Factors Affecting the Specific Heat of Silver
Several factors can influence the precise value obtained for the specific heat of silver:
1. Temperature:
The specific heat of silver is not constant across all temperatures. It exhibits a slight dependence on temperature, generally increasing slightly as the temperature rises. This variation is relatively small within a moderate temperature range but becomes more significant at extremely high or low temperatures. This temperature dependence stems from the complex interactions between the lattice vibrations (phonons) and electrons within the silver structure.
2. Pressure:
Pressure also plays a role, though the effect is often less pronounced than temperature. At higher pressures, the atoms are packed more closely, potentially influencing the vibrational modes and thus affecting the specific heat. However, for most practical applications, the pressure effect on the specific heat of silver can be considered negligible.
3. Purity:
The purity of the silver sample significantly impacts the specific heat measurement. The presence of impurities can alter the lattice structure and electron interactions, thereby affecting the heat capacity. Highly pure silver will exhibit a more precise and consistent specific heat value compared to alloys or silver containing impurities. This is a critical consideration in scientific experiments requiring high accuracy.
4. Phase:
The specific heat also varies depending on the phase of the silver. While silver is usually encountered in its solid phase at standard conditions, its specific heat will differ significantly in its liquid or gaseous phases. These phase transitions involve substantial changes in the atomic arrangement and intermolecular forces, leading to distinct specific heat values.
Measurement of Silver's Specific Heat
Determining the specific heat of silver involves employing calorimetric techniques. These methods involve measuring the heat absorbed or released by a silver sample as its temperature changes. Common calorimetry methods include:
1. Differential Scanning Calorimetry (DSC):
DSC measures the heat flow associated with phase transitions or other thermal events. By analyzing the heat flow as a silver sample undergoes a temperature change, its specific heat can be precisely determined.
2. Adiabatic Calorimetry:
Adiabatic calorimetry minimizes heat exchange between the sample and its surroundings. This approach enables precise measurement of the heat absorbed by a silver sample, enabling accurate calculation of its specific heat.
3. Drop Calorimetry:
Drop calorimetry involves heating a sample to a known temperature and then dropping it into a calorimeter. The heat transfer from the sample to the calorimeter is measured, providing data to calculate the specific heat.
The accuracy of these methods depends on factors such as instrument precision, sample preparation, and data analysis techniques. Advanced calorimetric techniques are required to obtain highly precise values for the specific heat of silver, accounting for the aforementioned influencing factors.
Applications Leveraging Silver's Specific Heat
The specific heat of silver, along with its excellent thermal and electrical conductivity, makes it suitable for various applications:
1. Electronics:
Silver's high thermal conductivity, coupled with its moderate specific heat, is crucial in heat dissipation in electronic components. Silver-based thermal interface materials efficiently transfer heat away from heat-generating components, preventing overheating and ensuring reliable operation.
2. Heat Exchangers:
Silver's high thermal conductivity and relatively high specific heat make it a suitable material for heat exchangers, although its cost often limits its use to specialized applications requiring exceptional thermal performance.
3. Industrial Processes:
In industrial processes involving heating and cooling, understanding silver's specific heat is crucial for precise temperature control and energy efficiency. Accurate calculations of heat transfer are essential for optimizing processes and minimizing energy waste.
4. Scientific Research:
The specific heat of silver serves as a fundamental parameter in various scientific investigations. Accurate knowledge of this property is essential for modeling thermal behavior in materials science, physics, and engineering simulations.
5. Medical Applications:
Silver's antimicrobial properties, combined with its thermal properties, might find applications in medical devices or treatments. However, research in this area is ongoing, and further exploration is needed to fully understand its potential.
Silver's Specific Heat in Comparison to Other Metals
Comparing the specific heat of silver to other common metals helps highlight its unique thermal properties:
-
Copper: Copper, another excellent conductor, has a specific heat of approximately 385 J/kg·K, higher than silver. This indicates that copper requires more energy to raise its temperature than silver.
-
Aluminum: Aluminum has a specific heat of around 900 J/kg·K, significantly higher than both silver and copper. This suggests aluminum can absorb a large amount of heat with a smaller temperature increase.
-
Gold: Gold has a specific heat of about 129 J/kg·K, lower than silver. This implies that gold heats up more quickly than silver for the same amount of energy input.
-
Iron: Iron's specific heat is approximately 450 J/kg·K, higher than silver.
This comparison underscores that while silver possesses excellent thermal conductivity, its specific heat is moderate compared to some other metals, offering a balance between rapid heat transfer and temperature stability.
Conclusion: The Significance of Silver's Specific Heat
The specific heat of silver, approximately 235 J/kg·K, is a critical parameter influencing its behavior in numerous applications. Its value reflects the amount of energy required to change its temperature, playing a crucial role in heat transfer processes and thermal management. Understanding the factors influencing this property, along with accurate measurement techniques, is crucial for harnessing silver's unique thermal characteristics in various technological and scientific advancements. Further research into the precise temperature and pressure dependence of silver's specific heat will continue to refine our understanding of this important material property and expand its potential applications. The ongoing advancements in calorimetry and materials science will undoubtedly lead to a more comprehensive and nuanced understanding of silver's thermal behavior in the future.
Latest Posts
Latest Posts
-
Image Formation In A Plane Mirror
Apr 03, 2025
-
Us Air Force Tongue And Quill
Apr 03, 2025
-
The Coefficients In A Chemical Equation Represent The
Apr 03, 2025
-
How To Calculate The Gradient Of A Stream
Apr 03, 2025
-
What Does Insoluble Mean In Chemistry
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Specific Heat Of Silver . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
