Which State Of Matter Has No Definite Volume Or Shape
Muz Play
Apr 02, 2025 · 6 min read
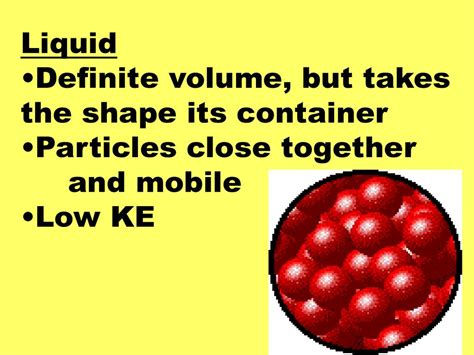
Table of Contents
Which State of Matter Has No Definite Volume or Shape? Understanding Gases
The answer to the question, "Which state of matter has no definite volume or shape?" is unequivocally gas. Unlike solids and liquids, gases are characterized by their lack of fixed volume and shape. They readily expand to fill any container they occupy and conform to its shape. Understanding this fundamental property of gases requires delving into the kinetic molecular theory and exploring the unique behavior of gaseous molecules. This article will comprehensively explore the properties of gases, comparing them to solids and liquids, and examining the factors influencing their behavior.
Defining the States of Matter: A Quick Review
Before we delve into the specifics of gases, let's briefly review the three fundamental states of matter: solid, liquid, and gas. Each state is defined by the arrangement and interaction of its constituent particles (atoms or molecules).
Solids: Fixed Shape and Volume
Solids possess a definite shape and volume. Their particles are tightly packed together in a highly ordered arrangement, held in place by strong intermolecular forces. This results in a rigid structure that resists changes in shape and volume. Examples of solids include ice, rocks, and metals.
Liquids: Definite Volume, Indefinite Shape
Liquids have a definite volume but an indefinite shape. Their particles are closer together than in gases but farther apart than in solids. They possess weaker intermolecular forces than solids, allowing them to flow and conform to the shape of their container while maintaining a constant volume. Examples include water, oil, and mercury.
Gases: Indefinite Shape and Volume
Gases exhibit neither a definite shape nor a definite volume. Their particles are widely dispersed and move freely, possessing negligible intermolecular forces. This allows them to expand and fill any container, taking on its shape and volume. Examples include air, oxygen, and helium.
The Kinetic Molecular Theory of Gases
The behavior of gases can be explained by the kinetic molecular theory (KMT). This theory postulates that:
-
Gases are composed of tiny particles (atoms or molecules) that are in constant, random motion. These particles are in continuous, chaotic movement, colliding with each other and the walls of their container.
-
The volume of the gas particles themselves is negligible compared to the volume of the container. This implies that the space occupied by the gas particles is insignificant relative to the overall volume of the gas.
-
There are no significant attractive or repulsive forces between gas particles. This means that the gas particles interact minimally with each other, primarily through collisions.
-
The average kinetic energy of gas particles is directly proportional to the absolute temperature (Kelvin scale). As the temperature increases, the average speed and kinetic energy of the gas particles increase.
-
Collisions between gas particles and the walls of the container are perfectly elastic. This means that no energy is lost during these collisions.
Why Gases Lack Definite Volume and Shape: A Deeper Dive
The lack of a definite volume and shape in gases directly stems from the principles of the kinetic molecular theory. Let's examine this in more detail:
-
Weak Intermolecular Forces: The negligible intermolecular forces in gases allow particles to move freely and independently. There's no cohesive force holding them together in a fixed arrangement, unlike in solids and liquids. This freedom of movement enables gases to expand to fill any available space.
-
High Kinetic Energy: The high kinetic energy of gas particles constantly pushes them against the walls of their container. This pressure exerted by the gas particles leads to the expansion of the gas to occupy the entire volume of the container.
-
Large Interparticle Distances: The vast distances between gas particles mean that the gas is highly compressible. By applying pressure, you can reduce the interparticle distances, thereby decreasing the volume of the gas.
Factors Affecting Gas Behavior
Several factors influence the behavior of gases, including:
Pressure (P)
Pressure is the force exerted by gas particles per unit area on the walls of their container. It's directly proportional to the number of collisions between gas particles and the container walls. Increased temperature or the number of gas particles will increase pressure.
Volume (V)
Volume is the space occupied by the gas. It's directly related to the amount of gas present and inversely related to the pressure. Increasing the volume will reduce the pressure, and vice-versa.
Temperature (T)
Temperature is a measure of the average kinetic energy of the gas particles. Higher temperatures lead to increased kinetic energy, resulting in faster particle movement and more frequent collisions, thus increasing pressure.
Amount of Gas (n)
The amount of gas, usually measured in moles (n), is directly proportional to the pressure. More gas particles mean more collisions and higher pressure, assuming the volume and temperature remain constant.
The Ideal Gas Law: Putting It All Together
The relationship between pressure, volume, temperature, and the amount of gas is summarized in the ideal gas law:
PV = nRT
Where:
- P = pressure
- V = volume
- n = amount of gas (moles)
- R = the ideal gas constant
- T = temperature (Kelvin)
The ideal gas law is a powerful tool for predicting and calculating the behavior of gases under various conditions. However, it's important to remember that it's a simplification and doesn't perfectly describe the behavior of all real gases, especially at high pressures or low temperatures where intermolecular forces become significant.
Real Gases vs. Ideal Gases
The ideal gas law assumes that gas particles have negligible volume and exert no intermolecular forces. While this is a good approximation for many gases under normal conditions, real gases deviate from ideal behavior, particularly at high pressures and low temperatures.
At high pressures, the volume of the gas particles becomes significant relative to the container volume, leading to a smaller volume than predicted by the ideal gas law. At low temperatures, intermolecular forces become stronger, causing particles to clump together, reducing the pressure.
Applications of Understanding Gaseous Properties
The understanding of gaseous properties and behavior is crucial in numerous fields, including:
-
Meteorology: Understanding atmospheric pressure, temperature, and gas composition is essential for weather forecasting.
-
Chemistry: Gaseous reactions and equilibrium are fundamental concepts in chemical processes.
-
Engineering: Gas dynamics are critical in designing engines, pipelines, and other systems involving gas flow.
-
Medicine: Understanding gas exchange in the lungs is crucial for respiratory health.
Conclusion
In summary, the state of matter that has no definite volume or shape is gas. This unique property stems from the kinetic molecular theory, which describes the constant, random motion of gas particles with negligible intermolecular forces and insignificant individual volume compared to the container. The behavior of gases is governed by factors like pressure, volume, temperature, and the amount of gas present, all elegantly encapsulated in the ideal gas law. While the ideal gas law provides a valuable simplification, it's important to acknowledge the deviations of real gases from ideal behavior under certain conditions. The understanding of gas properties and behavior has broad implications across many scientific and engineering disciplines.
Latest Posts
Latest Posts
-
A Magnifier Makes Things Appear Larger Because
Apr 03, 2025
-
What Is The Difference Between Intermolecular And Intramolecular Forces
Apr 03, 2025
-
Where Is The Energy Stored In Glucose
Apr 03, 2025
-
Light Amplification By The Stimulated Emission Of Radiation
Apr 03, 2025
-
Octet Rule Violation Vs Wrong Electron Total
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which State Of Matter Has No Definite Volume Or Shape . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
