An Atom That Has Lost Or Gained An Electron
Muz Play
Apr 03, 2025 · 6 min read
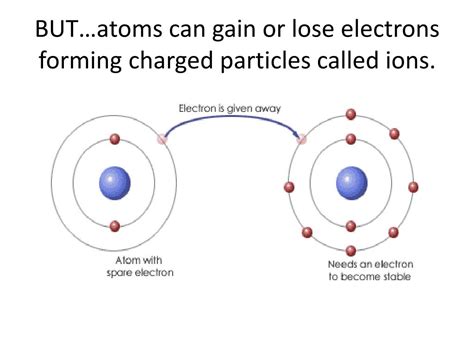
Table of Contents
An Atom That Has Lost or Gained an Electron: Understanding Ions and Their Significance
Atoms, the fundamental building blocks of matter, are typically depicted as neutral entities—possessing an equal number of protons (positively charged particles) and electrons (negatively charged particles). However, this neutrality is not always the case. Atoms can readily lose or gain electrons, transforming into charged particles known as ions. This seemingly simple process has profound implications, influencing the properties of matter, driving chemical reactions, and playing a critical role in numerous biological processes. Understanding the behavior of ions is therefore crucial for comprehending the world around us.
What Happens When an Atom Loses an Electron?
When an atom loses an electron, it loses a unit of negative charge. This results in a net positive charge, transforming the atom into a cation. The process of cation formation is often driven by the atom's desire to achieve a more stable electronic configuration, usually involving a full outer electron shell (octet rule). Elements with low electronegativity, meaning they have a weaker pull on electrons, tend to lose electrons more readily. These elements are typically found on the left side of the periodic table, belonging to groups 1 and 2 (alkali and alkaline earth metals).
The Formation of Cations: A Closer Look
Consider sodium (Na), a highly reactive alkali metal. Sodium has 11 electrons arranged in three shells: 2, 8, 1. The single electron in the outermost shell is loosely held and easily lost. When sodium loses this electron, it forms a sodium cation (Na⁺), with a stable configuration of 2, 8 electrons—matching the electron configuration of the noble gas neon (Ne). This stable configuration represents a lower energy state, making the process energetically favorable.
The loss of an electron significantly alters the properties of the atom. For instance, sodium metal is a soft, silvery-white solid that reacts violently with water. However, the Na⁺ ion, present in common table salt (NaCl), is much less reactive and exhibits vastly different chemical properties.
Factors Influencing Cation Formation
Several factors contribute to an atom's tendency to lose electrons and form cations:
- Ionization Energy: This is the energy required to remove an electron from an atom. Elements with lower ionization energies lose electrons more easily.
- Electronegativity: As mentioned earlier, atoms with lower electronegativity are more likely to lose electrons.
- Electrostatic Forces: The attraction between the positively charged nucleus and the negatively charged electrons influences the ease with which an electron can be removed.
What Happens When an Atom Gains an Electron?
Conversely, when an atom gains an electron, it acquires a unit of negative charge, forming an anion. This process is often driven by the atom's desire to fill its outermost electron shell, achieving a stable octet configuration. Elements with high electronegativity, meaning they have a strong attraction for electrons, tend to gain electrons more readily. These elements are typically found on the right side of the periodic table, particularly in groups 16 and 17 (chalcogens and halogens).
The Formation of Anions: A Closer Look
Consider chlorine (Cl), a highly reactive halogen. Chlorine has 17 electrons arranged in three shells: 2, 8, 7. Its outermost shell lacks one electron to achieve a stable octet. When chlorine gains an electron, it forms a chloride anion (Cl⁻), with a stable configuration of 2, 8, 8 electrons—matching the electron configuration of the noble gas argon (Ar). This stable configuration is energetically favorable.
Similar to cation formation, gaining an electron significantly alters the atom's properties. Chlorine gas (Cl₂) is a toxic, greenish-yellow gas, while the chloride anion (Cl⁻) is a constituent of numerous compounds, including table salt (NaCl), and is essential for many biological processes.
Factors Influencing Anion Formation
Several factors influence an atom's tendency to gain electrons and form anions:
- Electron Affinity: This is the energy change associated with adding an electron to a neutral atom. Elements with high electron affinities readily gain electrons.
- Electronegativity: As mentioned earlier, atoms with higher electronegativity are more likely to gain electrons.
- Electrostatic Forces: The attractive forces between the nucleus and the added electron play a crucial role in anion formation.
The Significance of Ions in Chemistry and Biology
The formation of ions has far-reaching consequences across various scientific disciplines:
Chemical Bonding:
Ions play a crucial role in forming ionic compounds, where the electrostatic attraction between oppositely charged ions holds them together. The formation of table salt (NaCl), for example, involves the transfer of an electron from sodium to chlorine, resulting in the formation of Na⁺ and Cl⁻ ions, which are then held together by strong electrostatic forces. Ionic compounds exhibit unique properties, such as high melting and boiling points, and often dissolve readily in water, forming electrically conductive solutions.
Biological Processes:
Ions are essential for numerous biological processes:
- Nerve Impulse Transmission: The movement of sodium (Na⁺) and potassium (K⁺) ions across cell membranes is crucial for nerve impulse transmission and muscle contraction.
- Enzyme Function: Many enzymes require specific ions as cofactors to function correctly.
- Osmosis and Water Balance: The concentration of ions inside and outside cells affects osmosis and water balance within organisms.
- Bone Structure: Calcium ions (Ca²⁺) are essential for building and maintaining strong bones.
Industrial Applications:
Ions find wide applications in various industries:
- Electroplating: The deposition of a thin layer of metal onto a surface using electric current relies on the movement of metal ions.
- Batteries: Batteries utilize the movement of ions to generate electricity.
- Corrosion Protection: The application of protective coatings often involves ion-based processes.
Identifying and Characterizing Ions
Identifying and characterizing ions often involves various techniques:
- Spectroscopy: Techniques like atomic emission spectroscopy and mass spectrometry can identify the presence and concentration of ions in a sample.
- Electrochemical Methods: Methods like potentiometry and voltammetry can measure the concentration and activity of ions in solution.
- Crystallography: X-ray crystallography can determine the arrangement of ions in crystalline solids.
Understanding Ionization States and Oxidation Numbers
The charge on an ion is often referred to as its oxidation state or oxidation number. This number indicates the number of electrons an atom has gained or lost. For instance, Na⁺ has an oxidation state of +1, indicating it has lost one electron, while Cl⁻ has an oxidation state of -1, indicating it has gained one electron. Understanding oxidation states is crucial for balancing chemical equations and predicting the reactivity of compounds.
Polyatomic Ions: A More Complex Scenario
While the examples above focused on monoatomic ions (ions consisting of a single atom), many ions are polyatomic, consisting of two or more atoms covalently bonded together and carrying a net charge. Examples include sulfate (SO₄²⁻), nitrate (NO₃⁻), and ammonium (NH₄⁺). These polyatomic ions behave similarly to monoatomic ions in terms of their electrostatic interactions but possess more complex internal structures and bonding.
Conclusion: The Ubiquity and Importance of Ions
The loss or gain of electrons by atoms, resulting in the formation of ions, is a fundamental process with profound implications. Ions drive chemical reactions, influence the properties of matter, and play crucial roles in biological systems and numerous industrial applications. Understanding the behavior of ions is therefore essential for advancements in chemistry, biology, materials science, and many other fields. Further exploration into the intricacies of ion formation, properties, and interactions will continue to unveil new insights and drive technological innovations. From the simple act of salt dissolving in water to the complex processes of nerve impulse transmission, the world around us is profoundly shaped by the behavior of these charged particles.
Latest Posts
Latest Posts
-
Two Bones That Form The Nasal Septum
Apr 04, 2025
-
How To Determine The Age Of A Fossil
Apr 04, 2025
-
The Nucleus Of An Atom Consists Of
Apr 04, 2025
-
What Happens To The Electrons In Metallic Bonding
Apr 04, 2025
-
How To Know Which Orbital A Lone Pair Is In
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about An Atom That Has Lost Or Gained An Electron . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
