Does Co Have Dipole Dipole Forces
Muz Play
Apr 02, 2025 · 5 min read
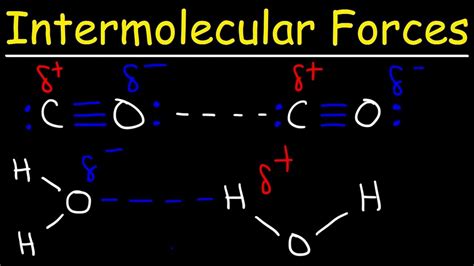
Table of Contents
Does CO Have Dipole-Dipole Forces? Understanding Intermolecular Forces in Carbon Monoxide
Carbon monoxide (CO), a simple yet crucial molecule, presents an interesting case study when exploring intermolecular forces. While seemingly straightforward, understanding the presence and strength of dipole-dipole forces in CO requires a closer look at its molecular structure and the nature of these forces. This article delves into the intricacies of CO's intermolecular interactions, examining its polarity, the role of dipole-dipole forces, and comparing them to other forces at play.
Understanding Dipole-Dipole Forces
Before investigating CO specifically, let's establish a clear understanding of dipole-dipole forces. These forces arise from the interaction between polar molecules. A polar molecule possesses a permanent dipole moment, meaning there's an uneven distribution of electron density, creating a slightly positive end (δ+) and a slightly negative end (δ-). Dipole-dipole forces occur when the positive end of one polar molecule is attracted to the negative end of another. These forces are stronger than London dispersion forces (present in all molecules) but weaker than hydrogen bonds.
Key characteristics of dipole-dipole forces:
- Polarity is essential: The existence of a permanent dipole moment is the prerequisite for dipole-dipole interactions.
- Strength varies: The strength of dipole-dipole forces depends on the magnitude of the dipole moment. Larger dipole moments lead to stronger forces.
- Directional: These forces are directional, meaning the interaction is strongest when the molecules are aligned with their positive and negative ends facing each other.
- Temperature dependence: The effectiveness of dipole-dipole forces is affected by temperature. Higher temperatures increase molecular kinetic energy, disrupting the alignment of dipoles and weakening the interactions.
The Polarity of Carbon Monoxide (CO)
The central question regarding CO's intermolecular forces hinges on its polarity. To determine this, we need to analyze its molecular structure and the electronegativity difference between carbon and oxygen.
Electronegativity: Oxygen is significantly more electronegative than carbon. Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. The higher electronegativity of oxygen means it attracts the shared electrons in the CO bond more strongly than carbon.
Molecular Geometry: CO is a linear molecule. This linear geometry plays a crucial role in determining the overall polarity.
Resultant Dipole Moment: Due to the difference in electronegativity and the linear geometry, the oxygen atom carries a partial negative charge (δ-), while the carbon atom carries a partial positive charge (δ+). This creates a permanent dipole moment, making CO a polar molecule.
The Presence of Dipole-Dipole Forces in CO
Given that CO is a polar molecule with a permanent dipole moment, it does exhibit dipole-dipole forces as one of its intermolecular interactions. These forces contribute to the overall attractive forces between CO molecules in the liquid and solid phases. The positive carbon end of one CO molecule attracts the negative oxygen end of another, leading to an attractive interaction.
Comparing Dipole-Dipole Forces with Other Intermolecular Forces in CO
While dipole-dipole forces are present in CO, it's important to consider that other intermolecular forces are also at play.
London Dispersion Forces (LDFs): These forces are present in all molecules, including CO. LDFs arise from temporary fluctuations in electron distribution, creating instantaneous dipoles. While individually weak, the cumulative effect of LDFs can be significant, especially in larger molecules. In the case of CO, LDFs are present alongside dipole-dipole forces.
Relative Strength: In CO, the dipole-dipole forces are significantly stronger than the LDFs due to the presence of the permanent dipole moment. However, both contribute to the overall cohesive forces within a sample of CO.
The Significance of Dipole-Dipole Forces in CO's Properties
The presence of dipole-dipole forces, along with LDFs, significantly influences CO's physical properties:
-
Boiling Point: The relatively higher boiling point of CO compared to nonpolar molecules of similar molecular weight is partially attributed to the presence of dipole-dipole forces. These forces require more energy to overcome during the phase transition from liquid to gas.
-
Solubility: The polarity of CO affects its solubility in various solvents. CO is more soluble in polar solvents than in nonpolar solvents due to the dipole-dipole interactions between CO and the solvent molecules.
-
Phase Transitions: The interplay of dipole-dipole forces and LDFs dictates the temperatures at which CO undergoes phase transitions (solid to liquid, liquid to gas).
Addressing Common Misconceptions
Some might argue that the dipole moment of CO is relatively small compared to other polar molecules. While true, even a small dipole moment is sufficient to create dipole-dipole forces. The key takeaway is the presence of the permanent dipole, not necessarily its magnitude. The combined effects of dipole-dipole forces and LDFs collectively contribute to the intermolecular interactions in CO.
Conclusion: A Comprehensive Look at CO's Intermolecular Forces
In conclusion, carbon monoxide (CO) does indeed possess dipole-dipole forces as a result of its polar nature due to the electronegativity difference between carbon and oxygen atoms and its linear molecular geometry creating a permanent dipole moment. Although these dipole-dipole forces are complemented by London dispersion forces, they significantly influence CO's physical and chemical properties, affecting its boiling point, solubility, and phase transitions. Understanding the interplay of these forces is key to grasping the behavior of this important molecule. Further studies exploring the precise contribution of each force to CO's macroscopic properties would provide a more comprehensive picture of its behavior under different conditions and environments. This intricate interplay of intermolecular forces highlights the complexity and fascinating nature of molecular interactions, even within seemingly simple molecules like CO.
Latest Posts
Latest Posts
-
Interpreting Data Absorption Spectra And Photosynthetic Pigments
Apr 03, 2025
-
Where Are The Cell Bodies For The Sensory Neurons Located
Apr 03, 2025
-
Difference Between Amorphous Solid And Crystalline Solid
Apr 03, 2025
-
Alkali Metals Alkaline Earth Metals Halogens And Noble Gases
Apr 03, 2025
-
What Determines Primary Structure Of A Protein
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Does Co Have Dipole Dipole Forces . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
