Does Electron Withdrawing Groups Increase Acidity
Muz Play
Apr 03, 2025 · 6 min read
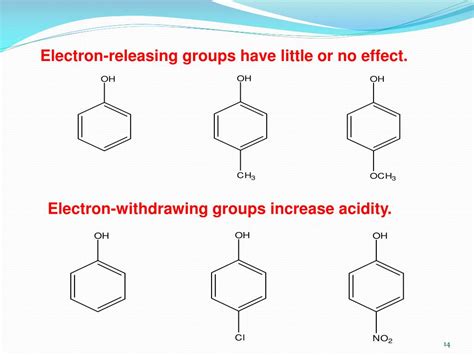
Table of Contents
Does Electron Withdrawing Groups Increase Acidity? A Deep Dive into Organic Chemistry
The acidity of a molecule, its propensity to donate a proton (H⁺), is a fundamental concept in organic chemistry. Understanding the factors that influence acidity is crucial for predicting reaction outcomes and designing new molecules with desired properties. One of the most significant factors affecting acidity is the presence of electron withdrawing groups (EWGs). This article will delve into the intricate relationship between EWGs and acidity, exploring the mechanisms behind their influence and providing numerous examples to solidify your understanding.
Understanding Acidity and the Role of Conjugate Bases
Before diving into the effect of EWGs, let's establish a solid foundation. Acidity is measured by the pKa value, a logarithmic scale indicating the strength of an acid. A lower pKa value signifies a stronger acid. The acidity of a molecule is intrinsically linked to the stability of its conjugate base – the species formed after the acid donates a proton. The more stable the conjugate base, the stronger the acid.
This stability is largely determined by several factors, including:
- Resonance Stabilization: The delocalization of electrons through resonance structures can significantly stabilize the conjugate base, thereby increasing the acidity of the parent acid.
- Inductive Effects: Electron-withdrawing or electron-donating groups attached to the molecule influence the electron density around the acidic proton.
- Hybridization: The hybridization of the atom bearing the acidic proton also affects acidity. More s-character leads to greater acidity.
- Solvent Effects: The solvent in which the acid is dissolved plays a critical role in influencing its acidity.
The Impact of Electron Withdrawing Groups (EWGs)
Electron withdrawing groups (EWGs) are atoms or functional groups that pull electron density away from a molecule. This electron-withdrawing effect plays a crucial role in enhancing the acidity of a compound. By withdrawing electron density, EWGs stabilize the negative charge on the conjugate base, making the acid more likely to donate a proton.
How EWGs Stabilize Conjugate Bases:
The primary mechanism by which EWGs increase acidity is through the stabilization of the negative charge on the conjugate base. This stabilization can occur through:
- Inductive Effect: EWGs, through their electronegativity, pull electron density away from the negatively charged atom in the conjugate base, reducing the overall charge density and making the conjugate base more stable. This effect is strongest when the EWG is close to the acidic proton.
- Resonance Effect: Some EWGs can participate in resonance, delocalizing the negative charge over multiple atoms. This delocalization significantly stabilizes the conjugate base, leading to increased acidity.
Examples of Electron Withdrawing Groups:
Several functional groups act as effective EWGs:
- Nitro group (-NO₂): A very strong EWG due to its high electronegativity and resonance effects.
- Cyano group (-CN): Another powerful EWG, possessing both inductive and resonance effects.
- Carboxyl group (-COOH): Exhibits both inductive and resonance effects, though less potent than -NO₂ or -CN.
- Haloalkyls (-F, -Cl, -Br, -I): Halogens are EWGs due to their electronegativity; their inductive effect on acidity is dependent on their proximity to the acidic proton and their electronegativity. Fluorine is the most powerful inductive EWG amongst the halogens.
- Sulfone group (-SO₂R): A strong EWG owing to the presence of two highly electronegative oxygen atoms.
- Carbonyl groups (C=O): The carbonyl group's electron-withdrawing nature stems from the highly electronegative oxygen atom.
Illustrative Examples: Comparing Acidity with and without EWGs
Let's consider some specific examples to illustrate the impact of EWGs on acidity:
1. Carboxylic Acids:
Compare acetic acid (CH₃COOH) and trifluoroacetic acid (CF₃COOH). The trifluoromethyl group (-CF₃) is a potent EWG. The presence of three highly electronegative fluorine atoms significantly withdraws electron density from the carboxyl group, stabilizing the conjugate base (trifluoroacetate ion) considerably more than the acetate ion. Consequently, trifluoroacetic acid is a much stronger acid than acetic acid.
2. Phenols:
Phenol (C₆H₅OH) is a weak acid. Introducing EWGs to the benzene ring enhances its acidity. For example, p-nitrophenol is significantly more acidic than phenol because the nitro group withdraws electron density from the phenoxide ion, stabilizing the negative charge. The p-position offers the most effective stabilization due to resonance.
3. Alcohols:
Alcohols are generally weak acids. The addition of EWGs close to the hydroxyl group increases acidity. For instance, an alcohol with a nearby electronegative atom will be more acidic than a simple alcohol.
4. β-Dicarbonyl Compounds:
β-dicarbonyl compounds, containing two carbonyl groups separated by a single methylene group, exhibit enhanced acidity compared to simple ketones or esters. This is due to the resonance stabilization of the conjugate base formed after deprotonation. The negative charge is delocalized across both carbonyl groups, leading to a significantly stabilized conjugate base.
Distance Dependence of Inductive Effects
The strength of the inductive effect exerted by an EWG diminishes with increasing distance from the acidic proton. The closer the EWG is to the acidic proton, the more pronounced its effect on acidity. For example, the acidity of chloroacetic acid (ClCH₂COOH) is greater than that of β-chloropropionic acid (ClCH₂CH₂COOH), demonstrating this distance dependence.
Synergistic Effects: Combination of EWGs
In many molecules, multiple EWGs are present. When this occurs, their effects are often synergistic, meaning the combined effect on acidity is greater than the sum of the individual effects. For instance, a molecule with both a nitro and a cyano group will be considerably more acidic than a molecule with only one of these groups.
Exceptions and Complicating Factors
While the general principle holds true that EWGs increase acidity, exceptions exist. Steric effects, hydrogen bonding, and other factors can sometimes counteract or overshadow the influence of EWGs. In such cases, a careful analysis of the specific molecular structure and its interactions with the environment is needed to fully understand the acidity.
Applications and Significance
The understanding of how EWGs affect acidity has significant implications across various fields:
- Drug Design: Many drugs contain acidic or basic functional groups. Manipulating acidity through the introduction of EWGs is crucial for optimizing drug properties, such as absorption, distribution, metabolism, and excretion (ADME).
- Materials Science: The acidity of materials influences their properties and reactivity. Tuning acidity through EWGs is important in designing materials with specific characteristics.
- Catalysis: Acid-base catalysis is a fundamental concept in chemistry. The acidity of catalysts can be modified by the incorporation of EWGs to optimize catalytic activity.
Conclusion: A Powerful Tool for Fine-Tuning Acidity
Electron withdrawing groups are powerful tools for manipulating the acidity of organic molecules. By understanding the mechanisms through which EWGs stabilize conjugate bases—primarily through inductive and resonance effects—we can predict and design molecules with specific acidity profiles. The distance dependence of these effects and potential synergistic interactions between multiple EWGs highlight the complexity and richness of this area of organic chemistry. This knowledge is invaluable in diverse scientific fields, ranging from drug development to materials science, underscoring the importance of understanding the profound relationship between EWGs and acidity. The examples provided in this article serve as a robust foundation for further exploration into this fascinating and vital area of chemistry. Remember to always consider the interplay of different factors – steric effects, hydrogen bonding, and solvent effects – for a comprehensive understanding of molecular acidity.
Latest Posts
Latest Posts
-
Why Are Seeds An Evolutionary Advantage For Seed Plants
Apr 04, 2025
-
Do Cations Lose Or Gain Electrons
Apr 04, 2025
-
Most Likely Cations And Anions On The Periodic Table
Apr 04, 2025
-
What Is Feedback Inhibition In Biology
Apr 04, 2025
-
What Is Group 2 In The Periodic Table
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Does Electron Withdrawing Groups Increase Acidity . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
