Elements That Have Characteristics Of Both Metals And Nonmetals
Muz Play
Apr 02, 2025 · 6 min read
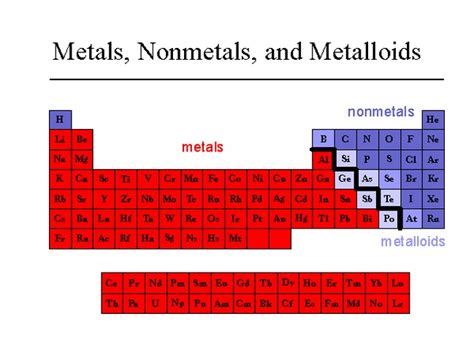
Table of Contents
Metalloids: Bridging the Gap Between Metals and Nonmetals
The periodic table, a cornerstone of chemistry, organizes elements based on their properties. While we neatly categorize elements into metals, nonmetals, and metalloids, the reality is often more nuanced. This article delves into the fascinating world of metalloids, elements that exhibit a fascinating blend of metallic and nonmetallic characteristics, making them uniquely valuable in various applications. Understanding their properties, behaviors, and uses is crucial for appreciating their significance in modern technology and beyond.
Defining Metalloids: A Balancing Act of Properties
Metalloids, also known as semimetals, occupy a diagonal band on the periodic table, separating the metals from the nonmetals. This strategic position reflects their intermediate nature. They don't perfectly fit into either category, possessing a unique combination of traits. This ambiguity, however, is precisely what makes them so interesting and technologically vital.
Here's a breakdown of the key characteristics that define metalloids:
A Blend of Metallic and Nonmetallic Properties:
-
Electrical Conductivity: Metalloids are semiconductors. This means their electrical conductivity falls between that of metals (excellent conductors) and nonmetals (insulators). Their conductivity is highly temperature-dependent, increasing with increasing temperature – a characteristic distinctly different from metals.
-
Thermal Conductivity: Similar to their electrical conductivity, their thermal conductivity sits in the middle ground, exhibiting lower values compared to metals but higher than most nonmetals.
-
Appearance: Metalloids can have a metallic luster, but they are often brittle and lack the malleability and ductility of typical metals.
-
Chemical Reactivity: Their chemical reactivity varies widely depending on the specific element and conditions. Some metalloids can act as oxidizing agents, while others can act as reducing agents. They often form amphoteric oxides, exhibiting both acidic and basic properties.
-
Other Properties: Metalloids may exhibit other properties that blur the line between metals and nonmetals. For example, their magnetic properties can differ significantly from either extreme.
Key Metalloids and Their Unique Traits: A Closer Look
Let's explore some of the most well-known metalloids, highlighting their individual properties and applications:
Boron (B): A Versatile Element
Boron, the first metalloid in the periodic table, is a hard, brittle, dark brown solid. Its high melting point and exceptional hardness are distinctly metallic, but its low electrical conductivity at room temperature and its ability to form covalent bonds are nonmetallic characteristics.
Applications: Boron finds its applications in:
-
Nuclear Reactor Control Rods: Boron's ability to absorb neutrons makes it vital for controlling nuclear reactions.
-
High-Strength Materials: Boron fibers are added to composite materials to enhance their strength and stiffness.
-
Semiconductors: Boron-doped silicon is used in semiconductor manufacturing.
-
Pharmaceuticals: Boron compounds have uses in medicinal applications.
Silicon (Si): The Foundation of Modern Electronics
Silicon, the second most abundant element in the Earth's crust, is arguably the most crucial metalloid. Its semiconducting properties are fundamental to the modern electronics industry.
Applications: Silicon's importance is evident in:
-
Semiconductors: The foundation of integrated circuits (ICs), microprocessors, and countless electronic devices. Silicon's ability to be doped with other elements (like boron or phosphorus) allows for precise control of its electrical conductivity.
-
Solar Cells: Silicon's ability to convert sunlight into electricity makes it crucial in photovoltaic technology.
-
Glass and Ceramics: Silicon dioxide (SiO2), commonly known as silica, is a major component of glass and various ceramic materials.
-
Silicones: Organosilicon polymers, called silicones, have found diverse applications in lubricants, sealants, and medical implants.
Germanium (Ge): A High-Performance Semiconductor
Germanium is a hard, grayish-white metalloid with excellent semiconducting properties, once crucial in transistors and other early electronic components. While silicon has largely replaced it in many applications, germanium retains its importance in niche areas.
Applications: Germanium continues to be used in:
-
Infrared Optics: Germanium's transparency to infrared light makes it vital in infrared detectors and lenses used in thermal imaging and optical fiber systems.
-
Fiber Optics: Germanium-doped silica fiber optics enhance light transmission.
-
Semiconductors: Despite being surpassed by silicon, germanium remains crucial in some high-performance transistors and integrated circuits.
Arsenic (As): A Toxic Metalloid with Limited Uses
Arsenic is a gray, crystalline metalloid known for its toxicity. However, it does have some limited industrial applications.
Applications: Arsenic's uses are mostly restricted to:
-
Doping Semiconductors: Small amounts of arsenic are used to dope silicon and gallium arsenide semiconductors to improve their properties.
-
Wood Preservatives: Historically used in wood preservatives (though this use is now largely phased out due to its toxicity).
-
Medicinal Uses: Some arsenic compounds are being investigated for medicinal properties, particularly in cancer treatment (though this remains highly specialized and carefully controlled).
Safety Note: Arsenic is highly toxic, and its use and handling must be strictly controlled.
Antimony (Sb): Applications in Alloys and Flame Retardants
Antimony is a silvery-white, brittle metalloid. Its main use is in alloying with other metals to improve their properties.
Applications: Antimony is utilized in:
-
Alloys: Improves the hardness and strength of lead-based alloys used in batteries and ammunition.
-
Flame Retardants: Antimony compounds are used as flame retardants in plastics and textiles.
-
Semiconductors: Similar to arsenic, it can be used in doping semiconductors.
Tellurium (Te): A Rare Metalloid with Specialized Uses
Tellurium is a rare, silvery-white metalloid. Its semiconducting properties and other unique traits have led to specialized applications.
Applications: Tellurium is used in:
-
Solar Cells: Used in cadmium telluride solar cells, although not as extensively as silicon.
-
Steel Alloys: Small quantities are used to improve the machinability of steel.
-
Rubber Vulcanization: Tellurium compounds accelerate the vulcanization process of rubber.
The Importance of Metalloids in Modern Technology
The unique properties of metalloids have been instrumental in shaping modern technology. From the silicon chips powering our computers and smartphones to the germanium in infrared detectors used in night vision, metalloids are essential components of numerous devices and systems. Their role in shaping our world is far-reaching and continues to evolve.
Semiconductors and Electronics:
Metalloids are the bedrock of the semiconductor industry. Their ability to control the flow of electricity, depending on temperature, voltage, or other factors, underpins modern electronics. Without metalloids, we wouldn't have the computers, smartphones, and other electronic gadgets that have become integral parts of our lives.
Energy Applications:
Metalloids play crucial roles in renewable energy technologies. Silicon-based solar cells are increasingly crucial in harnessing solar energy. The development of improved photovoltaic materials is an area of ongoing research and innovation, with metalloids continuing to be at the forefront.
Medical Applications:
While many metalloids are toxic, some compounds are finding uses in targeted drug delivery systems. Research into the medicinal applications of certain metalloids is an ongoing area, albeit with careful consideration of their toxicity.
Other Industries:
Metalloids are utilized in other diverse sectors, including:
-
Catalysis: Some metalloids and their compounds act as catalysts in various chemical reactions.
-
Glass and Ceramics: Silicon dioxide (silica) is a fundamental component of glass, while various other metalloid compounds enhance properties of different glass and ceramic formulations.
-
Coatings: Metalloids can be incorporated into coatings to enhance properties like wear resistance or corrosion resistance.
Conclusion: Metalloids—A Bridge Between Two Worlds
Metalloids stand out as a unique group of elements that bridge the gap between metals and nonmetals. Their intermediate properties, particularly their semiconducting capabilities, have revolutionized numerous technological fields. From the silicon chips driving modern electronics to the tellurium in solar cells, their contribution to our technological advancements is undeniable. Further research and innovations continue to unlock the full potential of these remarkable elements. As we delve deeper into the complexities of materials science and engineering, the role of metalloids will undoubtedly become increasingly important, fueling further innovation across various domains. The ongoing exploration of their properties and applications promises even greater technological advancements in the future.
Latest Posts
Latest Posts
-
Point Estimate Of The Population Standard Deviation
Apr 03, 2025
-
A Magnifier Makes Things Appear Larger Because
Apr 03, 2025
-
What Is The Difference Between Intermolecular And Intramolecular Forces
Apr 03, 2025
-
Where Is The Energy Stored In Glucose
Apr 03, 2025
-
Light Amplification By The Stimulated Emission Of Radiation
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Elements That Have Characteristics Of Both Metals And Nonmetals . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
