How To Read A Solubility Curve
Muz Play
Apr 02, 2025 · 6 min read
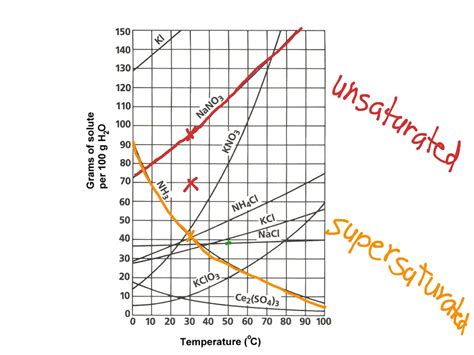
Table of Contents
How to Read a Solubility Curve: A Comprehensive Guide
Solubility curves are essential tools in chemistry, providing a visual representation of how the solubility of a substance changes with temperature. Understanding how to read and interpret these curves is crucial for various applications, from predicting the outcome of chemical reactions to designing effective purification processes. This comprehensive guide will equip you with the knowledge and skills to confidently navigate solubility curves and extract valuable information.
Understanding the Basics of Solubility
Before delving into the intricacies of solubility curves, let's establish a solid foundation. Solubility refers to the maximum amount of a solute that can dissolve in a given amount of solvent at a specific temperature and pressure. The solute is the substance being dissolved, and the solvent is the substance doing the dissolving (usually a liquid, most commonly water). Solubility is often expressed in grams of solute per 100 grams of solvent (g/100g).
A saturated solution is one where the maximum amount of solute has dissolved in the solvent at a given temperature. Any additional solute added will simply settle at the bottom, undissolved. An unsaturated solution contains less solute than the solvent can hold at that temperature, while a supersaturated solution contains more solute than it should theoretically hold at that temperature. Supersaturated solutions are unstable and tend to precipitate out excess solute if disturbed.
Deconstructing the Solubility Curve Graph
A solubility curve is a graphical representation of the relationship between temperature and solubility. The x-axis typically represents temperature (usually in degrees Celsius), while the y-axis represents solubility (typically in grams of solute per 100 grams of solvent). Each point on the curve represents the saturation point of the solute at that specific temperature.
Key Features of a Solubility Curve:
-
Positive Slope: Most solubility curves exhibit a positive slope, meaning that solubility increases with increasing temperature. This is because higher temperatures provide more kinetic energy to the solvent molecules, allowing them to more effectively break apart and surround the solute particles.
-
Negative Slope: Some substances, notably gases, show a negative slope. Their solubility decreases with increasing temperature. This is due to the increased kinetic energy of the gas molecules, allowing them to overcome the attractive forces holding them in solution and escape into the atmosphere.
-
Data Points: The curve itself is generated by plotting experimental data points, each representing the solubility of the solute at a particular temperature. These data points are meticulously obtained through laboratory experiments.
-
Solubility at a Specific Temperature: To find the solubility of a substance at a given temperature, locate the temperature on the x-axis, draw a vertical line upwards until it intersects the solubility curve, then draw a horizontal line to the y-axis to read the solubility value.
-
Comparison of Solubilities: Solubility curves allow for the comparison of the solubilities of different substances at the same temperature or the solubility of a single substance at different temperatures. This comparison is easily visualized by observing the relative positions of the curves.
Interpreting Solubility Curves: Practical Examples
Let's illustrate the interpretation of solubility curves with some practical examples. Imagine you have a solubility curve showing the solubility of potassium nitrate (KNO₃) and potassium chloride (KCl) in water.
Scenario 1: Determining Solubility at a Specific Temperature
Suppose you want to know the solubility of KNO₃ at 40°C. Locate 40°C on the x-axis, trace a vertical line until it intersects the KNO₃ curve, and then draw a horizontal line to the y-axis. The y-axis value represents the solubility of KNO₃ at 40°C in g/100g of water. For instance, if the value is 60 g/100g, it means 60 grams of KNO₃ can dissolve in 100 grams of water at 40°C.
Scenario 2: Comparing Solubilities at a Specific Temperature
Suppose you want to compare the solubilities of KNO₃ and KCl at 60°C. Find 60°C on the x-axis, draw vertical lines intersecting both curves, and then draw horizontal lines to the y-axis. The respective y-axis values will show the solubilities of each salt at 60°C. The higher value indicates the more soluble substance at that temperature.
Scenario 3: Determining the Temperature for a Specific Solubility
Let's say you have a solution containing 80 grams of KNO₃ in 100 grams of water, and you want to know at what temperature this solution becomes saturated. Locate 80 g/100g on the y-axis, draw a horizontal line until it intersects the KNO₃ curve, then draw a vertical line to the x-axis. The x-axis value gives the temperature at which the solution becomes saturated.
Scenario 4: Predicting Crystallization
If you cool a saturated solution of a substance, its solubility decreases. If the temperature drops below the solubility curve, the excess solute will precipitate out of the solution as crystals. This principle is used in crystallization, a common purification technique.
Advanced Applications of Solubility Curves
Solubility curves are not merely tools for simple solubility calculations. They have significant applications in various fields:
-
Recrystallization: Solubility curves are fundamental in recrystallization, a purification technique that involves dissolving a compound in a hot solvent, followed by cooling to precipitate out the purified compound. The choice of solvent and the cooling rate are guided by the solubility curve.
-
Chemical Engineering: In chemical engineering, solubility curves help design and optimize processes involving dissolution, crystallization, and precipitation. Accurate prediction of solubility is crucial for efficient process control and product yield.
-
Pharmaceutical Industry: Solubility is a critical factor in drug formulation and delivery. Solubility curves help determine the appropriate solvent and conditions for dissolving active pharmaceutical ingredients (APIs) and ensuring optimal drug absorption.
-
Environmental Science: Solubility curves are essential in understanding the environmental fate of pollutants. Knowing the solubility of a pollutant allows scientists to predict its mobility and bioavailability in soil and water systems.
-
Geochemistry: In geochemistry, solubility curves are used to understand the formation and dissolution of minerals in geological systems. They help predict the behavior of minerals under different temperature and pressure conditions.
Tips for Effectively Using Solubility Curves
-
Accurate Data: Ensure the solubility curve you are using is accurate and reliable. Use curves obtained from reputable sources or established experimental data.
-
Units: Pay close attention to the units used on the axes (temperature and solubility). Incorrect interpretation of units can lead to significant errors.
-
Interpolation: If you need to determine solubility at a temperature not explicitly shown on the curve, carefully interpolate between the known data points. Avoid extrapolation beyond the range of the given data.
-
Context: Always consider the context of the curve. The specific solute and solvent involved are crucial for accurate interpretation. Different solutes will have different solubility curves.
-
Limitations: Remember that solubility curves are typically generated under specific conditions (e.g., atmospheric pressure). Deviations from these conditions can affect solubility.
Conclusion
Solubility curves are invaluable tools providing a clear visual representation of the complex relationship between temperature and solubility. Mastery of reading and interpreting these curves is essential for anyone working in chemistry or related fields. By carefully analyzing the curve's features and applying the principles outlined in this guide, you can confidently extract critical information and apply it to a wide range of applications, from simple solubility calculations to advanced chemical processes. Remember to always check your data source for accuracy and consider the limitations of solubility curves when making predictions.
Latest Posts
Latest Posts
-
How Does Molecular Biology Support The Theory Of Evolution
Apr 03, 2025
-
Not A And Not B Truth Table
Apr 03, 2025
-
What Is The Density Of Glass
Apr 03, 2025
-
Oxidation State Of O In H2o
Apr 03, 2025
-
Does Electron Withdrawing Groups Increase Acidity
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How To Read A Solubility Curve . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
