Oxidation State Of O In H2o
Muz Play
Apr 03, 2025 · 6 min read
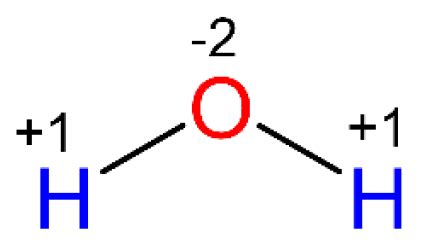
Table of Contents
The Oxidation State of Oxygen in Water (H₂O): A Deep Dive
The seemingly simple molecule of water, H₂O, holds a fascinating complexity when we delve into the oxidation state of its constituent atoms. While often taken for granted, understanding the oxidation state of oxygen in water is fundamental to comprehending a vast array of chemical reactions and processes. This article provides a comprehensive exploration of this topic, delving into the definition of oxidation states, the rules for assigning them, and the specific case of oxygen in water, along with its implications in various chemical contexts.
Understanding Oxidation States
Oxidation state, also known as oxidation number, is a crucial concept in chemistry that represents the hypothetical charge an atom would have if all bonds to atoms of different elements were 100% ionic. It's a crucial tool for balancing redox reactions, predicting the reactivity of compounds, and understanding the electronic structure of molecules. While not a true physical charge, the oxidation state provides a valuable framework for analyzing electron transfer and predicting chemical behavior.
Key Considerations When Assigning Oxidation States:
- Free elements: The oxidation state of an atom in its elemental form is always zero. For example, the oxidation state of oxygen in O₂ is 0.
- Monatomic ions: The oxidation state of a monatomic ion is equal to its charge. For instance, the oxidation state of sodium in Na⁺ is +1, and the oxidation state of chloride in Cl⁻ is -1.
- Fluorine: Fluorine, being the most electronegative element, always has an oxidation state of -1 in its compounds.
- Hydrogen: Hydrogen typically has an oxidation state of +1 in its compounds, except in metal hydrides (e.g., NaH), where it has an oxidation state of -1.
- Oxygen: Oxygen usually has an oxidation state of -2 in its compounds, except in peroxides (e.g., H₂O₂), where it has an oxidation state of -1, and in superoxides (e.g., KO₂), where it has an oxidation state of -1/2.
- Sum of oxidation states: In a neutral molecule, the sum of the oxidation states of all atoms must equal zero. In a polyatomic ion, the sum of the oxidation states must equal the charge of the ion.
Determining the Oxidation State of Oxygen in Water
Applying these rules to water (H₂O), we can determine the oxidation state of oxygen. We know that hydrogen typically has an oxidation state of +1. Since water is a neutral molecule, the sum of the oxidation states of the two hydrogen atoms and the one oxygen atom must equal zero.
Let's represent the oxidation state of oxygen as 'x'. We can set up the equation as follows:
2(+1) + x = 0
Solving for x, we get:
x = -2
Therefore, the oxidation state of oxygen in water is -2.
Implications of Oxygen's Oxidation State in Water
The -2 oxidation state of oxygen in water has significant implications across various chemical domains:
1. Redox Reactions:
Understanding the oxidation state of oxygen is crucial for balancing and predicting redox reactions. Water can act as both an oxidizing and a reducing agent, depending on the reaction conditions and the other reactants involved. For example, in the reaction of water with a highly reactive metal like sodium, water acts as an oxidizing agent, reducing itself to hydrogen gas while oxidizing the sodium to sodium ions. Conversely, in certain electrochemical processes, water can be reduced to hydrogen gas, acting as an oxidizing agent. The oxidation state of oxygen serves as a vital reference point for tracking electron transfers in these redox reactions.
2. Acid-Base Chemistry:
The oxidation state of oxygen in water is indirectly relevant to acid-base chemistry. The ability of water to act as both a Brønsted-Lowry acid (donating a proton) and a Brønsted-Lowry base (accepting a proton) is related to the polar nature of the O-H bond, which in turn stems from the electronegativity difference between oxygen and hydrogen. This electronegativity difference is a consequence of oxygen's higher effective nuclear charge and its ability to achieve a stable -2 oxidation state through covalent bonding.
3. Environmental Chemistry:
The oxidation state of oxygen is paramount in understanding various environmental processes. For instance, the oxidation of organic matter in aquatic systems, often mediated by microorganisms, involves changes in the oxidation states of carbon and oxygen. Water's role in these redox reactions is central to nutrient cycling and the overall health of aquatic ecosystems. Monitoring the oxygen levels in water bodies, often expressed as dissolved oxygen (DO), is a crucial indicator of water quality and ecosystem health. The oxidation state of oxygen helps to track the progress and efficiency of these processes.
4. Geochemistry:
Water plays a vital role in geological processes, and the oxidation state of oxygen is key to understanding many of these. Weathering of rocks, for example, involves reactions between water and minerals, often leading to changes in the oxidation states of various elements. The formation of hydrated minerals and the dissolution of minerals in water are heavily influenced by the oxidation state of oxygen in the water molecules. Hydrothermal vents, where superheated water interacts with rocks deep within the Earth's crust, provide a striking example of the geochemical significance of the oxygen’s oxidation state in water.
5. Biochemistry:
Oxygen's oxidation state is vital in various biochemical reactions. Respiration, the process by which organisms extract energy from nutrients, relies on the reduction of oxygen to water. This process involves a change in oxygen's oxidation state from 0 (in O₂) to -2 (in H₂O), releasing energy that fuels cellular processes. The precise control and regulation of this electron transfer are essential for life. Furthermore, many enzyme-catalyzed reactions involve water as a reactant or product, and the oxidation state of oxygen plays a role in the reaction mechanisms.
Exceptions and Advanced Considerations
While -2 is the most common oxidation state for oxygen, it's crucial to acknowledge exceptions:
- Peroxides (e.g., H₂O₂): In peroxides, oxygen exhibits an oxidation state of -1. The oxygen-oxygen single bond in peroxides results in each oxygen atom sharing only one electron with the other oxygen atom. Therefore, each oxygen atom "owns" seven electrons in total (six from its own valence shell plus one shared electron from the O-O bond), leaving it with one less electron than required for a full octet, resulting in an oxidation state of -1.
- Superoxides (e.g., KO₂): In superoxides, the oxidation state of oxygen is -1/2. This arises from the presence of the superoxide ion (O₂⁻), where the unpaired electron is delocalized over both oxygen atoms.
Understanding these exceptions requires a deeper understanding of molecular orbital theory and the concept of fractional oxidation states.
Conclusion
The oxidation state of oxygen in water, -2, is a seemingly simple yet fundamentally important concept that underpins a wide range of chemical phenomena. From redox reactions to biochemistry and geochemistry, the oxidation state of oxygen serves as a crucial tool for understanding electron transfer, chemical reactivity, and the intricate processes that shape our world. While the -2 oxidation state is predominant, recognizing the exceptions, such as peroxides and superoxides, further enriches our understanding of oxygen's versatile chemical behavior. Mastering this concept is essential for anyone pursuing a deeper understanding of chemistry and its diverse applications. Further research into the related fields of redox chemistry, electrochemistry, and environmental chemistry will reveal a deeper appreciation for the significance of this seemingly simple number. The oxidation state of oxygen in water isn't just a number; it's a window into the intricate workings of the chemical world.
Latest Posts
Latest Posts
-
What Is Steric Hindrance In Organic Chemistry
Apr 04, 2025
-
Why Are Seeds An Evolutionary Advantage For Seed Plants
Apr 04, 2025
-
Do Cations Lose Or Gain Electrons
Apr 04, 2025
-
Most Likely Cations And Anions On The Periodic Table
Apr 04, 2025
-
What Is Feedback Inhibition In Biology
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Oxidation State Of O In H2o . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
