If The Value Of Cfse For Ni Is
Muz Play
Apr 04, 2025 · 7 min read
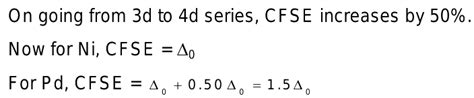
Table of Contents
If the Value of CFSE for Ni is... Understanding Crystal Field Stabilization Energy
The crystal field stabilization energy (CFSE) is a crucial concept in coordination chemistry, providing insights into the stability and properties of transition metal complexes. Understanding CFSE is essential for predicting the geometry, magnetism, and reactivity of these compounds. This article delves deep into the calculation and implications of CFSE, particularly focusing on nickel (Ni) complexes and how varying CFSE values influence their characteristics.
What is Crystal Field Stabilization Energy (CFSE)?
CFSE quantifies the energy difference between the d-orbital splitting in a transition metal ion's electronic configuration in a ligand field compared to the free ion. Ligands, being negatively charged or having lone pairs, repel the d-electrons of the central metal ion. This repulsion isn't uniform; it's dependent on the geometry of the complex. In an octahedral complex, for instance, the d-orbitals split into two sets: the lower-energy t<sub>2g</sub> orbitals (d<sub>xy</sub>, d<sub>xz</sub>, d<sub>yz</sub>) and the higher-energy e<sub>g</sub> orbitals (d<sub>x²−y²</sub>, d<sub>z²</sub>).
The energy difference between these sets is denoted as Δ<sub>o</sub> (octahedral splitting energy), while in a tetrahedral complex, it's Δ<sub>t</sub> (tetrahedral splitting energy). The magnitude of Δ<sub>o</sub> or Δ<sub>t</sub> depends on several factors, including the nature of the ligand (strong field vs. weak field) and the metal ion itself.
The CFSE is calculated by considering the number of electrons in each set of d-orbitals and the energy difference between them. A negative CFSE indicates stabilization of the complex, while a positive value indicates destabilization (which is less common).
Calculating CFSE for Nickel Complexes
Nickel (Ni) is a transition metal with an electronic configuration of [Ar] 3d<sup>8</sup>. Its oxidation states vary, leading to a range of possible d-electron configurations in complexes. Let's analyze some common scenarios:
1. Octahedral Ni(II) Complexes:
Ni(II) has a d<sup>8</sup> configuration. The distribution of electrons in the t<sub>2g</sub> and e<sub>g</sub> orbitals depends on the nature of the ligands.
-
Weak Field Ligands: In weak field octahedral complexes, the pairing energy (P) is greater than Δ<sub>o</sub>. Electrons will occupy the orbitals individually before pairing occurs, following Hund's rule. This results in a configuration of t<sub>2g</sub><sup>6</sup> e<sub>g</sub><sup>2</sup>.
CFSE = [-0.4 * 6 + 0.6 * 2] Δ<sub>o</sub> = -1.2 Δ<sub>o</sub>
-
Strong Field Ligands: With strong field ligands, Δ<sub>o</sub> > P, leading to electron pairing in the t<sub>2g</sub> orbitals before occupying the e<sub>g</sub> orbitals. The configuration is t<sub>2g</sub><sup>6</sup> e<sub>g</sub><sup>2</sup>. However, the energy difference due to pairing will need to be considered. While the electron distribution is the same, the actual CFSE calculation will reflect the pairing energy.
CFSE = [-0.4 * 6 + 0.6 * 2] Δ<sub>o</sub> - xP (where x is the number of electron pairs formed).
2. Tetrahedral Ni(II) Complexes:
In tetrahedral complexes, the d-orbital splitting is different. The energy difference, Δ<sub>t</sub>, is typically smaller than Δ<sub>o</sub> (Δ<sub>t</sub> ≈ 4/9 Δ<sub>o</sub>). The splitting pattern also differs; the orbitals split into a lower energy doublet (e) and a higher energy triplet (t<sub>2</sub>).
The d<sup>8</sup> configuration in a tetrahedral field generally results in a configuration of e<sup>4</sup> t<sub>2</sub><sup>4</sup> (weak field) because the Δ<sub>t</sub> is relatively small compared to the pairing energy.
CFSE = [-0.6 * 4 + 0.4 * 4] Δ<sub>t</sub> = -0.8 Δ<sub>t</sub>
3. Square Planar Ni(II) Complexes:
Square planar complexes are often observed for d<sup>8</sup> metal ions like Ni(II). The d-orbital splitting is more complex, with different energy levels for the d<sub>x²−y²</sub>, d<sub>xy</sub>, d<sub>xz</sub>, and d<sub>yz</sub> orbitals. The CFSE calculation is more intricate, involving determining the appropriate energy levels based on the ligand field strength.
Factors Affecting CFSE for Nickel Complexes
Several factors influence the value of CFSE for nickel complexes:
-
Ligand Field Strength: The strength of the ligands significantly impacts Δ<sub>o</sub> and Δ<sub>t</sub>. Strong field ligands (e.g., CN<sup>-</sup>, CO) cause larger splitting, leading to higher CFSE (in absolute value), while weak field ligands (e.g., I<sup>-</sup>, Br<sup>-</sup>) cause smaller splitting. The spectrochemical series organizes ligands by their field strength.
-
Geometry of the Complex: The geometry of the complex dictates the pattern of d-orbital splitting and thus the CFSE. Octahedral, tetrahedral, and square planar complexes show different splitting patterns, resulting in varied CFSE values.
-
Oxidation State of Nickel: The oxidation state of the nickel ion influences the number of d-electrons and consequently the CFSE. For instance, Ni(III) (d<sup>7</sup>) will have a different CFSE compared to Ni(II) (d<sup>8</sup>).
-
Pairing Energy: The energy required to pair electrons in the same orbital (pairing energy, P) plays a role, especially in weak field complexes where Δ is smaller than P.
Implications of CFSE
The CFSE value has significant implications for the properties of nickel complexes:
-
Stability: A larger negative CFSE indicates greater stability. This explains why certain complexes are favored over others based on the ligand field and geometry.
-
Magnetic Properties: The electron configuration determined by the CFSE and the ligand field strength directly affects the magnetic properties of the complex. High spin complexes (weak field) will have more unpaired electrons and will be paramagnetic, while low spin complexes (strong field) will have fewer unpaired electrons or none, leading to diamagnetism or reduced paramagnetism.
-
Spectroscopic Properties: The d-orbital splitting, and hence the CFSE, is directly related to the absorption spectrum of the complex. The color of a complex is a consequence of the electronic transitions between the split d-orbitals. This allows for experimental determination of the magnitude of the crystal field splitting parameter.
-
Reactivity: The CFSE influences the reactivity of the complex by affecting the accessibility of the d-electrons for reactions. Complexes with large negative CFSE might show reduced reactivity compared to those with smaller CFSE values.
Experimental Determination of CFSE
While CFSE is a theoretical concept, its value can be experimentally determined or estimated through various spectroscopic techniques such as:
-
UV-Vis Spectroscopy: Measuring the absorption spectrum of the complex allows for the determination of the ligand field splitting energy (Δ<sub>o</sub> or Δ<sub>t</sub>). This energy difference can then be used to calculate the CFSE.
-
Magnetic Susceptibility Measurements: Measuring the magnetic susceptibility helps determine the number of unpaired electrons, which provides information about the electron configuration and subsequently the CFSE.
Conclusion
The crystal field stabilization energy (CFSE) is a powerful tool for understanding the stability, magnetic properties, spectroscopic properties, and reactivity of transition metal complexes, particularly those of nickel. By considering the ligand field strength, geometry of the complex, oxidation state of the metal ion, and pairing energy, we can accurately predict and understand the behavior of these vital compounds. The interplay between these factors is crucial in the field of coordination chemistry and provides a solid foundation for understanding the complex world of transition metal complexes. Further research into these interactions continues to refine our understanding and allow for the design of novel materials with specific properties. The accurate calculation and interpretation of CFSE remain indispensable for advancing our knowledge in this dynamic field.
Latest Posts
Latest Posts
-
What Is A Balance Point In Math
Apr 05, 2025
-
What Is The Equilibrium Constant Expression For The Given System
Apr 05, 2025
-
Number Of Valence Electrons In Alkali Metals
Apr 05, 2025
-
Are Lysosomes Only In Animal Cells
Apr 05, 2025
-
Which Describes An Object In Projectile Motion
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about If The Value Of Cfse For Ni Is . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
