What Is The Equilibrium Constant Expression For The Given System
Muz Play
Apr 05, 2025 · 6 min read
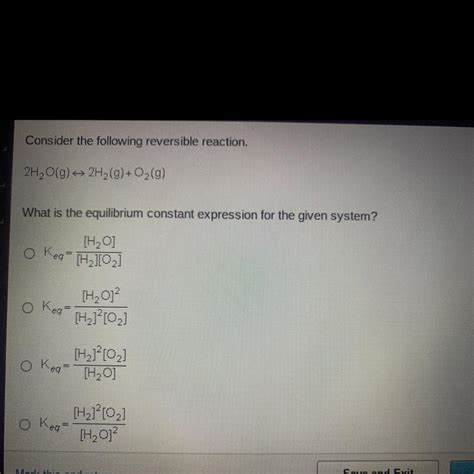
Table of Contents
What is the Equilibrium Constant Expression for the Given System? A Deep Dive into Equilibrium Chemistry
Understanding equilibrium constants is fundamental to mastering chemical reactions. This comprehensive guide delves into the intricacies of deriving and interpreting equilibrium constant expressions, providing a thorough understanding of this crucial concept in chemistry. We'll explore different types of equilibrium constants, their applications, and how to use them to predict the direction and extent of a reaction.
What is Chemical Equilibrium?
Before diving into equilibrium constant expressions, let's establish a clear understanding of chemical equilibrium itself. Chemical equilibrium is the state where the rates of the forward and reverse reactions are equal, resulting in no net change in the concentrations of reactants and products over time. This doesn't mean the reaction has stopped; rather, the forward and reverse reactions continue at the same rate, maintaining a dynamic balance.
This state of equilibrium is affected by several factors including temperature, pressure (for gaseous reactions), and concentration. Changes to these factors can shift the equilibrium position, favoring either the reactants or the products. This is governed by Le Chatelier's principle, which states that a system at equilibrium will adjust to counteract any stress applied to it.
The Equilibrium Constant (Kc): A Quantitative Measure of Equilibrium
The equilibrium constant, often denoted as Kc (for concentration), provides a quantitative measure of the relative amounts of reactants and products at equilibrium. It's a ratio of the concentrations of products to reactants, each raised to the power of its stoichiometric coefficient in the balanced chemical equation.
For a general reversible reaction:
aA + bB ⇌ cC + dD
The equilibrium constant expression is:
Kc = ([C]<sup>c</sup>[D]<sup>d</sup>) / ([A]<sup>a</sup>[B]<sup>b</sup>)
where:
- [A], [B], [C], and [D] represent the equilibrium concentrations of reactants A, B and products C, D respectively.
- a, b, c, and d are the stoichiometric coefficients from the balanced chemical equation.
Important Considerations:
- Pure solids and liquids: The concentrations of pure solids and liquids are considered constant and are not included in the equilibrium constant expression. Their activity is considered to be 1.
- Gases: For gas-phase reactions, partial pressures can be used instead of concentrations, leading to the equilibrium constant Kp. The relationship between Kc and Kp is given by: Kp = Kc(RT)<sup>Δn</sup>, where R is the ideal gas constant, T is the temperature in Kelvin, and Δn is the change in the number of moles of gas (moles of gaseous products - moles of gaseous reactants).
- Units: The units of Kc depend on the stoichiometry of the reaction. It's unitless if the number of moles of reactants and products are equal.
Deriving Equilibrium Constant Expressions: Step-by-Step Examples
Let's illustrate the derivation of equilibrium constant expressions with several examples, showcasing different scenarios and complexities:
Example 1: A Simple Reversible Reaction
Consider the reversible reaction:
N<sub>2</sub>(g) + 3H<sub>2</sub>(g) ⇌ 2NH<sub>3</sub>(g)
The equilibrium constant expression is:
Kc = ([NH<sub>3</sub>]<sup>2</sup>) / ([N<sub>2</sub>][H<sub>2</sub>]<sup>3</sup>)
Example 2: Incorporating Pure Solids and Liquids
Consider the reaction:
CaCO<sub>3</sub>(s) ⇌ CaO(s) + CO<sub>2</sub>(g)
Since CaCO<sub>3</sub> and CaO are pure solids, their concentrations are constant and not included in the expression. Therefore, the equilibrium constant expression is simply:
Kc = [CO<sub>2</sub>]
Example 3: A More Complex Reaction
Consider the reaction:
2SO<sub>2</sub>(g) + O<sub>2</sub>(g) ⇌ 2SO<sub>3</sub>(g)
The equilibrium constant expression is:
Kc = ([SO<sub>3</sub>]<sup>2</sup>) / ([SO<sub>2</sub>]<sup>2</sup>[O<sub>2</sub>])
Example 4: Using Partial Pressures (Kp)
For the Haber-Bosch process (Example 1), the Kp expression would be:
Kp = (P<sub>NH3</sub><sup>2</sup>) / (P<sub>N2</sub>P<sub>H2</sub><sup>3</sup>)
Interpreting the Magnitude of the Equilibrium Constant
The magnitude of the equilibrium constant provides valuable insights into the position of equilibrium and the relative amounts of reactants and products:
-
Kc >> 1: The equilibrium lies far to the right, indicating that the reaction strongly favors the formation of products. The concentration of products at equilibrium is significantly higher than that of reactants.
-
Kc ≈ 1: The equilibrium lies near the middle, indicating that the concentrations of reactants and products are comparable at equilibrium.
-
Kc << 1: The equilibrium lies far to the left, indicating that the reaction strongly favors the formation of reactants. The concentration of reactants at equilibrium is significantly higher than that of products.
Applications of Equilibrium Constants
Equilibrium constants have widespread applications across various fields:
-
Predicting the direction of a reaction: By comparing the reaction quotient (Q) to the equilibrium constant (Kc), one can predict whether a reaction will proceed in the forward or reverse direction to reach equilibrium. If Q < Kc, the reaction will proceed to the right (towards products); if Q > Kc, the reaction will proceed to the left (towards reactants); if Q = Kc, the system is at equilibrium.
-
Calculating equilibrium concentrations: Knowing the initial concentrations and the equilibrium constant allows the calculation of the equilibrium concentrations of reactants and products. This often involves solving algebraic equations.
-
Understanding reaction spontaneity: While not a direct measure of spontaneity, the equilibrium constant provides insights into the feasibility of a reaction under specific conditions. A large Kc suggests a more favorable reaction.
-
Industrial processes: The equilibrium constant is crucial in optimizing industrial processes, such as the Haber-Bosch process for ammonia synthesis, by determining the optimal conditions (temperature, pressure, etc.) to maximize product yield.
-
Environmental science: Equilibrium constants are used to understand and model various environmental processes, such as acid-base equilibria in water systems or the solubility of minerals.
-
Biochemical reactions: Equilibrium constants play a vital role in understanding biochemical reactions, such as enzyme-catalyzed reactions and the binding of ligands to proteins.
Factors Affecting the Equilibrium Constant
While the equilibrium constant is a constant at a given temperature, it does change with changes in temperature. The effect of temperature on Kc depends on the enthalpy change (ΔH) of the reaction:
-
Exothermic reactions (ΔH < 0): Increasing the temperature shifts the equilibrium to the left (favoring reactants), decreasing Kc.
-
Endothermic reactions (ΔH > 0): Increasing the temperature shifts the equilibrium to the right (favoring products), increasing Kc.
Pressure changes affect the equilibrium constant only for reactions involving gases. An increase in pressure favors the side with fewer gas molecules, while a decrease in pressure favors the side with more gas molecules. However, it's important to remember that Kc itself does not change unless the temperature changes; instead the equilibrium concentrations change to maintain the same Kc value.
Conclusion
The equilibrium constant expression is a powerful tool for understanding and quantifying chemical equilibrium. By mastering its derivation and interpretation, we gain crucial insights into the direction, extent, and factors affecting chemical reactions. Its applications extend far beyond the classroom, shaping various scientific and industrial processes. This deep dive into equilibrium chemistry provides a solid foundation for further exploration of this fascinating and important area of study. Remember to always carefully balance your chemical equations before deriving your equilibrium constant expression. Accurate stoichiometry is paramount for accurate calculations.
Latest Posts
Latest Posts
-
How To Find Average Molecular Speed
Apr 05, 2025
-
How To Divide Monomials And Polynomials
Apr 05, 2025
-
Give The Iupac Name Of The Branched Alkane
Apr 05, 2025
-
Consider This Step In A Radical Reaction
Apr 05, 2025
-
Cold Temperatures Slow Down The Growth Of Microorganisms
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about What Is The Equilibrium Constant Expression For The Given System . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
